Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Perineural spread of melanoma to the central nervous system
George T Kannarkat*; Camilo Diaz Cruz
Department of Neurology, The Johns Hopkins Hospital, Baltimore, MD 21287, USA.
*Corresponding Author: George T Kannarkat
Department of Neurology, The Johns Hopkins Hospital,
Baltimore, MD 21287, USA.
Email: gkannarkat@gmail.com
Received : Oct 30, 2021
Accepted : Jan 07, 2022
Published : Jan 14, 2022
Archived : www.jcimcr.org
Copyright : © Kannarkat GT (2022).
Abstract
Leptomeningeal carcinomatosis is an increasingly recognized phenomenon for metastatic spread of a wide variety of carcinomas to the Central Nervous System (CNS). We report a case of a 79 year-old man with perineural spread of locally invasive melanoma to the CNS along the brachial nerves and plexus. This rarely reported mechanism of CNS spread of malignancy portends a poor prognosis and may be under-recognized because of its insidious nature and the vast number of possible differential diagnoses for neurologic symptoms in cancer patients.
Citation: Kannarkat GT, Cruz CD. Perineural spread of melanoma to the central nervous system. J Clin Images Med Case Rep. 2022; 3(1): 1567.
Introduction
Carcinomatous meningitis, also called leptomeningeal metastasis, is considered a rare event with an incidence of 5-8% and 15-20% in solid and hematological malignancies, respectively [1]. This phenomenon may be underrecognized in cancer patients since autopsy studies demonstrate an incidence of up to 20% [2]. Most commonly, tumors that invade the leptomeninges are of breast, lung, and melanoma origin [3]. CNS metastases have proposed to spread via hematogenous, direct invasion, choroid plexus, venous plexus, perineural, and iatrogenic routes [4]. Perineural routes of metastases are more often reported in head and neck cancers but can occur along spinal nerve roots [5]. Headache is the most common presenting symptom of carcinomatous meningitis with cerebrospinal fluid remarkable for lymphocytic pleocytosis, elevated protein level, low to normal glucose, and abnormal cytology [6-11]. Cytology is 80-95% sensitive but highly specific and is improved by higher volume and by using the initial fluid from a lumbar puncture [6]. MRI is only 76% sensitive for solid tumor carcinomatous meningitis demonstrating nodular enhancement but other features can include non-obstructive hydrocephalus [12]. Treatment can be with radiation and intrathecal or systemic chemotherapy but prognosis is poor [13]. Despite its rarity, recognizing leptomeningeal metastasis is important because it can inform and guide cancer treatment and prognosis.
Case presentation
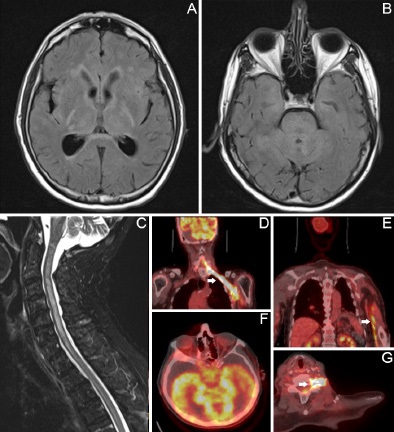
A 79-year-old man with metastatic melanoma was admitted to the hospital for one week of progressive confusion. His melanoma was initially diagnosed in his left shoulder and draining lymph nodes. Despite radiotherapy, resection, and immune checkpoint inhibitor therapy, he had further lymphatic and muscularspread associated with progressive loss of neuromuscular function in his left arm over several months. Upon evaluation by the neurology consultant in the hospital, he was sleepy but arousable to voice, disoriented, inattentive, unable to follow multistep commands, weak, numb, and hyporeflexic in his left upper extremity. Prior to evaluation, he had received highdose steroid for presumed paraneoplastic encephalitis. Brain and spine MRI revealed non-obstructive hydrocephalus with extensive T2-hyperintense, non-enhancing lesions in the bilateral mesial temporal lobes, diencephalon, cerebellum, brainstem, and central spinal cord (A-C). On cerebrospinal fluid analysis, he had normal opening pressure, 39 leukocytes/mL with a lymphocytic predominance, protein 88 mg/dl, glucose <2 mg/ dl, unrevealing infectious studies, and cytology showing rare, atypical cells. Autoimmune and paraneoplastic antibody panels from the CSF and serum were unrevealing. Whole-body PET/CT demonstrated increased radiotracer uptake along the nerves of the left arm into the spinal foramina as well as in the cerebellar and bilateral temporal cortices consistent with perineural spread of melanoma causing meningeal carcinomatosis (D-G). After discussion of these findings with his spouse, the patient was transitioned to comfort care and passed away within days.
Discussion
Perineural spread of melanoma is a recognized but rarely reported mechanism for tumor metastasis [14]. It has been reported to involve cranial nerves and is often associated with the desmoplastic variant [15]. However, perineural spread involving cervical nerves and the brachial plexus has been reported less frequently [16]. In a retrospective review of the Mayo Clinic database, Marek et al. identified patients with a history of melanoma and brachial plexus compromise treated between 1994 and 2017. Only two of them had Magnetic Resonance Imaging [MRI] and/or Positron Emission Tomography [PET] features consistent with perineural spread, and histopathological evidence of melanoma within nerves at the time of the symptomatic brachial plexopathy [17]. In one of them, the primary lesion was located in the posterior neck and the authors hypothesized that the melanoma spread along the lesser occipital nerve and/or the great auricular nerve to the cervical plexus with further extension to the phrenic, spinal accessory, and hypoglossal nerves and then to the brachial plexus. In the second case, the malignancy began on the angle of the right jaw, and the proposed spread was through a cranial nerve interconnection with the cervical plexus and eventually to the brachial plexus [16]. To our knowledge, there is only one published case of a neck melanoma with MRI, PET, and histological evidence of invasion along cervical nerves, extending proximally into the spinal cord [18]. In the case presented here, based on the clinical profile, imaging, and CSF studies, it was concluded that the patient had experienced perineural spread of melanoma along the brachial plexus into the meninges. After extensive infectious and autoimmune investigations were unrevealing, the CSF profile with undetectable glucose, high protein, and abnormal cells seen on cytology were strongly suggestive of direct leptomeningeal involvement. MRI findings were explained by leptomeningeal metastatic disease, particularly around the fourth ventricle and the cerebellum, non-obstructive hydrocephalus, and possibly compromised venous drainage of the spinal cord due to the pattern of central hyperintensity. Finally, several reasons have been proposed for potential underdiagnosis of perineural spread of melanoma resulting in brachial plexopathy. They include 1) the prolonged interval between the melanoma diagnosis and the onset of the neurologic symptoms, 2) development of the more frequent brain or lung metastases which may detract from evaluation of a brachial plexopathy, and 3) possible equivocal radiologic features on MRI and PET-CT that can be misdiagnosed as post-radiation or post-surgical changes.
Conclusion
Carcinomatous meningitis portends a poor prognosis but is often difficult to diagnose as CSF analysis and imaging have low sensitivity. We demonstrate a case of perineural spread of melanoma along the brachial plexus and spinal nerves into the CNS that is visualized on PET/CT. Since the differential diagnosis of neurologic symptoms in cancer patients is vast, consideration of leptomeningeal metastasis should be considered if an alternate explanation is not determined.
References
- Wang N, Bertalan M S, Brastianos P K. Leptomeningeal metastasis from systemic cancer: Review and update on management. Cancer. 2018; 124: 21–35.
- Le Rhun E, Taillibert S, Chamberlain M C. Carcinomatous meningitis: Leptomeningeal metastases in solid tumors. Surg. Neurol. Int. 2013; 4: S265-288.
- Clarke J L, Perez H R, Jacks L M, Panageas K S, Deangelis L M. Leptomeningeal metastases in the MRI era. Neurology. 2010; 74: 1449–1454.
- Arshad F, Wang L, Sy C, Avraham S, Avraham H K. Blood-brain barrier integrity and breast cancer metastasis to the brain. Pathol. Res. Int. 2010; 2011: 920509.
- Fowler B Z, Crocker I R, Johnstone P A S. Perineural spread of cutaneous malignancy to the brain. Cancer. 2005; 103: 2143–2153.
- Taillibert S, Chamberlain M C. Leptomeningeal metastasis. Handb. Clin. Neurol. 2018; 149: 169–204.
- Taillibert S. et al. Leptomeningeal metastases from solid malignancy: A review. J. Neurooncol. 2005; 75: 85–99.
- Nayar, G. et al. Leptomeningeal disease: Current diagnostic and therapeutic strategies. Oncotarget. 2017; 8: 73312–73328.
- Foo C T, Burrell L M, Johnson D F. An unusual presentation of carcinomatous meningitis. Oxf. Med. Case Rep. 2016; 2016: omw068.
- Chamberlain M C, Corey Bloom J. Leptomeningeal metastases: 111indium-DTPA CSF flow studies. Neurology. 1991; 41: 1765– 1769.
- Clarke J L. Leptomeningeal metastasis from systemic cancer. Contin. Minneap. Minn. 2012; 18: 328–342.
- Ko Y. et al. Association of MRI findings with clinical characteristics and prognosis in patients with leptomeningeal carcinomatosis from non-small cell lung cancer. J. Neurooncol. 2019; 143: 553–562.
- Anwar A, Gudlavalleti A, Ramadas P. Carcinomatous Meningitis. in StatPearls (StatPearls Publishing, 2021).
- Varey A H R. et al. Neurotropic melanoma: An analysis of the clinicopathological features, management strategies and survival outcomes for 671 patients treated at a tertiary referral center. Mod. Pathol. 2017; 30: 1538–1550.
- Desmoplastic, neural transforming and neurotropic melanoma: A review of 45 cases.
- Restrepo C E, et al. Perineural spread of malignant melanoma from the mandible to the brachial plexus: Case report. J. Neurosurg. 2015; 122: 784–790.
- Marek T, Laughlin R S, Howe B M, Spinner R J. Perineural Spread of Melanoma to the Brachial Plexus: Identifying the Anatomic Pathway (s). World Neurosurg. 2018; 111: e921–e926.
- Law W P, Pereira N, Vaska, K. Perineural spread of recurrent cutaneous melanoma along cervical nerves into the spinal cord. BJR Case Rep. 2017; 3: 20160122.