Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Granulocytic sarcoma with unusual localizations (breast and stomach) without bone marrow involvement after the allogeneic transplantation
Serife Solmaz1*; Gulen Gul2; Inci Alacacıoglu3; Fatih Demirkan3; Guner Hayri Ozsan3; Pinar Balci4; Sermin Ozkal5; Mehmet Ali Ozcan3
1 Associate Professor, Department of Hematology, Katip Celebi University, Izmir, Turkey.
2 Assistan Professor, Department of Pathology, Dokuz Eylul University, Izmir, Turkey.
3 Professor, Department of Hematology, Dokuz Eylul University Faculty of Medicine Hematology, Izmir, Turkey
4 Professor, Department of Radiology, Dokuz Eylul University Faculty of Radiology, Izmir, Turkey.
5 Professor, Department of Pathology, Dokuz Eylul University, Izmir, Turkey
*Corresponding Author: Serife Solmaz
Associate Professor, Department of Hematology, Katip
Celebi University, Izmir, Turkey
Email: solmazserife@yahoo.com
Received : Dec 02, 2021
Accepted : Jan 11, 2022
Published : Jan 18, 2022
Archived : www.jcimcr.org
Copyright : © Solmaz S (2022).
Abstract
Granulocytic sarcoma (GS) is a rare extramedullary solid tumor composed of immature myeloid cell that is associated with acute myeloid leukemia or myelodysplastic syndrome. Especially, the involvement of the breast as a pattern of relapse after allogeneic bone marrow transplantation (allo SCT) is extremely rare. We report the case of a 57-year-old woman who palpated a painless lump in the superior quadrant of her right breast. Her past medical history was a myelodysplastic syndrome (MDS) treated with bone marrow transplantation. Imaging work-up by mammogram and ultrasonography (US) showed solitary mass suspect of malignancy in breast. Trucut biopsy was performed from the suspect palpable mass of the right breast. Histologic findings were compatible with a granulocytic sarcoma in breast. While she was also evaluated for mass in the breast, also investigated by endoscopy for the symptoms of gastrointestinal system. The endoscopic biopsy showed diffuse neoplastic infiltration similar the pathology of the breast. GS of the breast is a rare extramedullary involvement of hematologic diseases. There are no the specific radyologic features in the presentation with GS of the breast. Our case also showed that careful histopathological review along with all panel of immunohistochemistry is extremely important for diagnose.
Keywords: myelodysplastic syndrome; granulocytic sarcoma; allogeneic bone marrow transplantation.
Citation: Solmaz S, Gul G, Alacacıoglu I, Demirkan F, Ozsan GH, et al. Granulocytic sarcoma with unusual localizations (breast and stomach) without bone marrow involvement after the allogeneic transplantation. J Clin Images Med Case Rep. 2022; 3(1): 1582.
Introduction
Granulocytic sarcoma is a rare extramedullary solid tumor composed of immature myeloid cell and is associated with acute myeloid leukemia or myelodysplastic syndrome [1].
It was first reported by Burns in 1811 and was described as chloroma by King in 1853 [2,3]. The common sites of presentation are lymph nodes, bone, soft tissue and skin [4,5]. The breast is an uncommon site for presentation of this tumour. Despite of its importance for the initial assessment of the disease, breast imaging also lacks specificity and GS can mimic several other tumors, including breast carcinoma and lymphoma [6]. However, there are instances where it has been reported without blood or bone marrow involvement [7]. Especially, the involvement of the breast as a pattern of relapse after bone marrow transplantation is extremely rare. We report case of high risk mylodysplastic syndrome (MDS) with relapse. 18 months after allo SCT as a granulocytic sarcoma in the right breast without bone marrow involvement. We also discuss the clinicopathologic features of granulocytic sarcoma in the breast and stomach after allo SCT.
Case presentation
A 56 year-old woman was applied to our center due to cytopenia, and the diagnosis of high risk myelodysplastic syndrome (MDS) was made in January 2016. At the time of diagnosis hemoglobin was 8,5 mg/dl, white blood cell count was 6 X 109 /l and platelet count was 93 X 109 /l in the peripheral blood count. Biochemial tests were normal. Blasts in the bone marrow aspirate were 12%. There was positive for cluster of differentiation (CD)34 (12%), for CD117 (20%) and for P53(30- 40%). Cytogenetic analysis showed normal karyotype. She was treated with decitabine 20 mg/kg/day intravenously (ıv) on days 1-5. She received 4 courses of decitabine treatment.
The patient underwent an alloSCT from her human leukocyte antigen (HLA)- matched brother after a conditioning regimen of busulfan (16 mg/kg) and cyclophoshomide (120 mg/kg) in June 2016. Graft- versus- host disease (GVHD) prophylaxis consisted of cyclosporine and methotrexate. Full donor chimerism was obtained on 30th day after transplantation. Acute skin GVHD disappeared with methylprednisolone. Chronic GVHD was not observed in the patient during follw-up period.
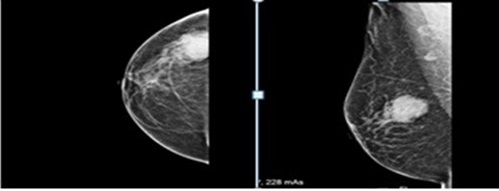
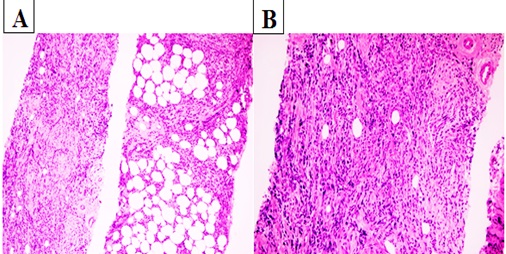
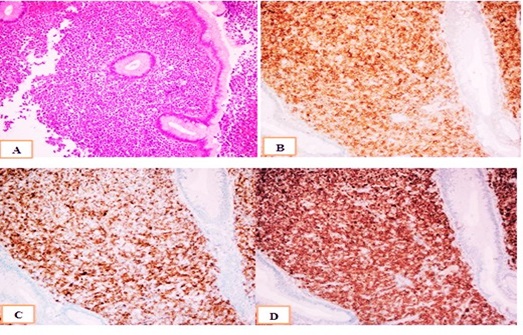
She was admitted with a palpable mass in the right breast that had been increasing in size for the past 3 months in December 2017. She had been a firm mass that was palpated on the right breast with 3.0 X 3.0 cm in diameter. There were no signs of retraction or skin abnormalities. Mammography planned to patient because of examined palpable mass. Mammography showed a single, irregular, poorly defined mass that was 2,5 X 3,5 X 4 cm in diameter, without calcification on the right breast with axillary lymph node that have 1,5 cm in diameter and thick cortexed (Figure 1). She has diagnosed as granulocytic sarcoma of the breast with a tru-cut biopsy of the right breast. The results of immunohistochemistry were positive for CD34, lysozyme, and myeloperoxidase (MPO), CD117, CD33 and were negative for CD3, CD20, CD30 and PAX5 (Figure 2). Her bone marrow aspiration and biopsy revealed no blast. Endoscopy has been planned due to abdominal pain and vomiting that persisting for 1 month. Endoscopy revealed a mass in the fundus area, 3,5 cm in size, from which a biopsy sample was taken. The gastroscopic biopsy showed diffuse neoplastic infiltration that the cluster of tumor cells expressed CD43, CD117, CD33,CD34 and MPO (Figure 3). GS was diagnosed in the stomach also. Later, she was treated with idarubicin at 12 mg/m2 /day ıv on days 1-3 and cytarabine ( ara-C) at 100 mg/m2 /day ıv on days 1-7. DLI was planned following chemotherapy.
Discussion
There are four clinical presentations of granulocytic sarcoma; 1) accompanied with AML; 2) could be the sign of AML; 3) could be associated with MDS transforming to leukemia and 4) solitary tumors [8]. Granulocytic sarcoma can ocur concomitantly or can percede AML in 2%-14% [9]. It has been known that extrameduller relaps can involve the soft tissue of the head and neck, bone, skin and central system.
However, they have reported to involve other sites such as the breast, nasopharynx, paranasal sinusoids, bladder, stomach, intestines, testis, perineum, plevral and peritoneal cavity [10]. Granulocytic sarcomas of the breast and stomach are uncommon neoplasms. Especially, The relapse with GS after bone marrow transplantation is extremely rare without bone marrow involvement.
The pathogenesis of GS after alloSCT still unexplained. The potential mechanism of GS without leukemic marrow involvement is that the graft versus leukemia (GVL) effect is particularly more potent in the bone marrow, but is thiner in the extrameduller sites. DLI could eradicate marrow remnant of tumor cells, but could not be effective in the extramedüller relaps [11]. However, DLI was suggested that the GVL effect developed from chronic GVHD is effective avoiding bone marrow relapse, but is not effective in extrameduller relapses.
The prognosis of patients with GS is extremely poor and the majority of patients died to disease within a short time. Management of GS after allo SCT is extremely difficult. There is no consensus on the treatment of GS as there have not been many randomized prospective trials. Previously, treatments for granulocytic sarcoma have included surgical, local radiotherapy and chemotherapy. However, allogeneic stem cell transplantation could be effective treatment for GS. Although local therapy can offer long-term survival, most patients develop systemic relapse. systemic or combined therapy should be considered in patients with good performance status [12]. However, DLI could be effect that showed in the literature and planed a second transplant in repeated relapse [13,14]. Our case was well tolerated systemic chemotherapy, later we planned DLI in the our patient because of good performance. The combine therapy modalities can be more toxic in patient that have made allo SCT, so we can be careful.
We found 18 patients (Table 1) with granulocytic sarcoma of following AML that have breast involvement in the literature. The age of presentation ranged from 28-71 years with a mean age of 45 years. The duration time of follow-up ranged from 2 to 96 months. Patients usually present with a mass. Our case presented 57 years old with a palpable mass in the right breast. Later, the abdominal pain and vomiting that had persisted for 1 month added to the patient’s complaints. The results of gastroscopy was found GS in stomach. Our case presented with unusual localizations (stomach and breast). The only 6.5% of myeloid sarcomas derive from the gastrointestinal tract [26]. These patients usually present with nonspesific symptoms including abdominal pain, vomiting or bleeding without bone marrow involvement ,that the same as in our case. The median time from SCT to GS relapse has been reported as 10 to 17 months [12,27,28]. However, our case was 57 year-old, and this time was 16 months after allo SCT.
Table 1: The cases of granulocytic sarcoma of the breast.
Author, Year |
Age of patient (years) |
Side of presentation |
Clinical follow up |
Eshghabadi et al15, 1986 |
45 |
R |
Alive,at 64 months |
Pettinato et al 16,1988 |
56 |
L |
Alive,at 11 months |
Jung et al 17,1998 |
28 |
R |
Dead, at 30 months |
Breccia et al 18,2000 |
71 |
L |
Alive, at 19 months |
Quintas- Cardoma et al 4, 2003 |
31 |
R |
Alive, at 37 months |
Shea et al 19, 2004 |
55 |
B |
Alive, at 24 months |
Valbuena et al 20,2005 |
31 |
R |
Alive, at 96 months |
D’Costa et al 21, 2007 |
45 |
L |
AML after 6 months |
Azim et al 7,2008 |
52 |
L |
Alive, at 12 months |
Chavez et al 22, 2009 |
29 |
R |
Dead, at 16 months |
Gündüz et al 23, 2012 |
30 |
R |
Dead, at 17 months |
Kim et al 6, 2013( 2 case) |
39 |
R |
Dead, at 2 months |
Nalva et al 1, 2015 |
33 |
B |
Alive, at 12 months |
Huang et al 24, 2015 |
58 |
L |
Alive, at 4 months |
Vieira et al 25, 2017 |
45 |
B |
Not evaluated |
The present case, 2018 |
57 |
R |
Alive, at 3 months |
Mammographic studies have shown that the lesions can have both regular and irregular borders and are non-calcified. However, GS is difficult to distinguish from other types of tumor using mammography or breast ultrasonography. In the present case; mammography showed a single, irregular, poorly defined mass that was 2,5 X 3,5 X 4 cm in diameter, without calcification on the right breast.
Hematoxylin and eosin staining can reveal a range of changes in morphology, causing the common misdiagnosis of GS. To approve the final diagnosis of GS, the immunohistochemical detection of MPO positive cells is useful. However, A panel of immunohistochemical markers comprising of MPO, CD34, CD43, CD117 and CD68 is positive in the majority of cases and 75% of cases are immunopositive for CD45 [20,22]. The results of immunohistochemistry were positive for CD34, lysozyme, MPO, CD117, CD33 in pathology preparations of the breast and the results of the gastroscopic biopsy showed diffüse neoplastic infiltration that the tumor cells expressed CD43, CD117, CD33,CD34 and MPO in our patient. In conclusion, it is difficult to make a clinical decision for the treatment of GS. The prognosis of patients who develop EM relapse after allo SCT remains poor. Immunophenotyping after biopsy is essential for differential diagnosis.
Conclusion
In conclusion, GS is malignant neoplasm of myeloid origin that could be in any area of the body. GS derived from the breast and stomach is relatively rare and tends to be misdiagnosed. The prognosis of granulocytic sarcoma remains poor. The patients with granulocytic sarcoma should be carefully evaluated with clinical and immunohistochemical results. However, it is necessary to perform a diagnostic search of any kind of mass that develops during the follow-up in patient with leukemia.
Declarations
Conflicts of interest: None
Consent for publication: The authors state that an informed and written consent was obtained from the patient prior to the publication.
Ethical approval: The authors declare that the article follows the Helsinki declaration guidelines and the manuscript has not been submitted to any other journal.
References
- Nalva A, Nath D, Suri V, Jamaluddin MA, Srivastava A. Myeloid sarcoma of the breast in an aleukemic patient: a rare entity in an uncommon location. Malaysian J Pathol. 2015; 37(1): 63-66.
- Neiman RS, Barcos M, Berard C, Bonner H, Mann R, Rydell RE, et al. Granulocytic sarcoma: a clinicopathologic study of 61 biopsied cases. Cancer. 1981; 48: 1426-37.
- Wiernik PH, Serpick AA. Granulocytic sarcoma (chloroma). Blood 1970; 35: 361-9.
- Quintas-Cardama A, Fraga M, Antunez J, Forteza J. Primary extramedullary myeloid tumor of the breast: a case report and review of the literature. Ann Hematol. 2003; 82(7): 431-4.
- Cassi E, Tosi A, De’ Paoli A, et al. Granulocytic sarcoma without evidence of acute leukemia: 2 cases with unusual localization(uterus and breast) and 1 case with bone localization. Haematologica. 1984; 69(4): 464-96.
- Kim JS, Hong WS, Jun SH, Jeong SH, Kang SY, Kim TH, et al. Granulocytic Sarcoma in Breast after Bone Marrow Transplantation. J Breast Cancer 2013; 16(1): 112-116.
- Azim HA Jr, Gigli F, Pruneri G, et al. Extramedullarymyeloid sarcoma of the breast. J Clin Oncol. 2008; 26(24): 4041-3.
- Shimuzi H, Saitoh T, Hatsumi N, Takada S, Yokohama A, Handa H, J et al. Clinical significance of granulocytic sarcoma in adult patients with acute myeloid leukemia. Cancer Sci. 2012; 103: 1513-17.
- Baer MR and Greer JP. Acute myeloid leukaemia in adults. In: Wintrobe’s Clinical Haematology. Greer JP, Foerster J, Rodgers GM, et al. 12th Edition. Lippincott Williams & Wilkins, Philadelphia, PA, pp1843 1888, 2009.
- Koc Y, Miller KB, Schenkein DP, et al. Extramedullary tumors of myeloid blasts in adults as a pattern of relapse followig allogeneic bone marrow transplantation. Cancer. 1999; 85: 608-615.
- Singhal S, Powles R, Kulkarni S, et al. Long-term follow-up of relapsed acute leukemia treated with ımmunotherapy after allogeneic transplantation: The inseparability of graft-versushost disease and graft versus leukemia, and the problem of extramedullary relapse. Leuk Lymphoma. 1999; 32: 505-512.
- Solh M, DeFor TE, Weisdorf DJ, Kaufman DS. Extramedullary relapse of acute myelogenous leukemia after allogeneic hematopoietic stem cell transplantation: better prognosis than systemic relapse. Biol Blood Marrow Transplant. 2012; 18: 106- 112.
- Kolb HJ. Graft versus leukemia effects of transplantation and donor lymphocytes. Blood. 2008; 112: 4371-4383.
- Yoshihara S, Ikegame K, Kaida K, Taniguchi K, Kato R, Inoue T, et al. Incidence of extramedullary relapse after haploidentical SCT for advanced AML/myelodysplastic syndrome. Bone Marrow Transplant. 2012; 47: 669-676.
- Eshghabadi M, Shojania AM, Carr I. Isolated granulocytic sarcoma: report of a case and review of the literature. J Clin Oncol. 1986; 4(6): 912-7.
- Pettinato G, De Chiara A, Insabato L, De Renzo A. Fine needle aspiration biopsy of a granulocytic sarcoma (chloroma) of the breast. Acta Cytol.1988; 32(1): 67-71.
- Jung SM, Kuo TT, Wu JH, Shih LY. Granulocytic sarcoma presenting as a giant breast tumor in a pregnant woman: a case report. Changgeng Yi Xue Za Zhi. 1998; 21(1): 97-102.
- Breccia M, Petti MC, Fraternali-Orcioni G, et al. Granulocytic sarcoma with breast and skin presentation: a report of a case successfully treated by local radiation and systemic chemotherapy. Acta Haematol. 2000; 104(1): 34-7.
- Shea B, Reddy V, Abbitt P, Benda R, Douglas V, Wingard J. Granulocytic sarcoma (chloroma) of the breast: a diagnostic dilemma and review of the literature. Breast J. 2004; 10(1): 48- 53.
- Valbuena JR, Admirand JH, Gualco G, Madeiros LJ. Myeloid sarcoma involving the breast. Arch Pathol Lab Med. 2005; 129(1): 32-8.
- D’Costa GF, Hastak MS, Patil YV. Granulocytic sarcoma of breast: an aleukemic presentation. Indian J Med Sci. 2007; 61(3): 152-5.
- Vela-Chávez TA, Arrecillas-Zamora MD, Quintero- Cuadra LY, Fend F. Granulocytic sarcoma of the breast without development of bone marrow involvement: a case report. Diagn Pathol. 2009; 4: 2.
- Gunduz E,Akay MO, Karagulle M, Ak IS, Isolated Granulocytic Sarcoma of the Breast after Allogeneic Stem Cell Transplantation: A Rare Involvement Also Detected by 18FDG-PET/CT. Turk J Hematol. 2014; 31: 88-91
- Huang XE, Lı YJ, Zhou XD. Granulocytic sarcoma of the breast: A case report. Oncology Letters, 2015; 10: 2447-2449
- Vieira VF, Vo Q, Jolinière JB, Khomsi F, Feki A, Hoogewoud HM. Granulocytic sarcoma presenting as a palpable Breast Lump. Case Report. 2017; 3: 1-6
- Pileri SA, Ascani S, Cox MC, Campidelli C, Bacci F, Piccioli M, et al. Myeloid sarcoma: Clinico-pathologic, phenotypic and cytogenetic analysis of 92 adult patients. Leukemia. 2000: 21; 340-350.
- Lee KH, Lee JH, Choi SJ, Lee JH, Kim S, Seol M, et al. Bone marrow vs extramedullary relapse of acute leukemia after allogeneic hematopoietic cell transplantation: risk factors and clinical course. Bone Marrow Transplant 2003; 32: 835-842.
- Cunningham I. Extramedullary sites of leukemia relapse after transplant. Leuk Lymphoma. 2006; 47: 1745-1767.