Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 2
Coinfection with Sars-Cov2 and Kodamaea ohmeri: A case report from Tunisia
Nawel Baccouchi1,2*; Latifa Mtibaa1; Fida Maatallah1; Mohamed Bejaoui1; Hana Souid1,2; Boutheina Jemli1
1 Laboratory of Parasitology Mycology, Military Hospital of Tunis, 1008 Monfleury, Tunis, Tunisia
2 Faculty of Sciences of Tunis, University of Tunis El Manar, Tunisia.
*Corresponding Author: Nawel Baccouchi
Laboratory of Parasitology Mycology, Military Hospital
of Tunis, 1008 Monfleury, Tunis, Tunisia.
Email: nawelbaccouchi@gmail.com
Received : Nov 23, 2021
Accepted : Jan 12, 2022
Published : Jan 19, 2022
Archived : www.jcimcr.org
Copyright : © Baccouchi N (2022).
Abstract
Introduction: Kodamaea ohmeri, previously known as Yamadazyma ohmeri and Pichia ohmeri, is ascomycetous yeast that belongs to the Saccharomycetaceae family. It is a rare clinical isolate that has recently become known to cause various human infections. Here, we report the first case of co-infection with Sars-Cov2 and Kodamaea ohmeri in Tunisian patient without underlying disease.
Methodology: We describe a patient with Covid-19 where Kodamaea ohmeri was isolated in sputum and protected tracheal samples. The identification was carried out by Vitek®2 YST ID card and confirmed by PCR sequencing. Susceptibility to antifungal was made by E-test.
Results: Kodamaea ohmeri was identified using phenotypic, biochemical, and molecular methods. The isolate was susceptible for amphotericin B, caspofungin, and intermediate sensitivity for Fluconazole.
Conclusion: Patients with severe Covid-19 are mostly colonized and co-infected with rare yeasts which are resistant to antifungals. This case incites to pay more attention to the emergence of this yeast.
Keywords: Kodamaea ohmeri; diagnosis; antifungal; molecular methods.
Citation: Baccouchi N, Mtibaa L, Maatallah F, Bejaoui M, Souid H, Jemli B. Coinfection with Sars-Cov2 and Kodamaea ohmeri: A case report from Tunisia. J Clin Images Med Case Rep. 2022; 3(1): 1587.
Introduction
Kodamaea ohmeri, previously known as Yamadazyma ohmeri and Pichia ohmeri, is an ascomycetous yeast that belongs to the Saccharomycetaceae family [4]. Kodamaea ohmeri is a rare clinical isolate that has recently become known to cause various human infections [9]. The first clinical isolation of K. ohmeri was made in 1984 from the pleural fluid of a patient from Java, but this isolate was regarded as a contaminant [5]. In Tunisia, the three firsts cases reported were resistant to antifungals which incite to pay more attention to the emergence of this yeast in human pathology [6]. In addition, this fungus is reported especially in patients with underlying immunosuppression such as hematologic or solid neoplasms use of immunosuppressive drug, type 2 diabetes mellitus and HIV seropositive [6]. However, its isolation in COVID-19 patients has not been reported in the literature. Here, we report the first case of co-infection with Sars-Cov2 and Kodamaea ohmeri in Tunisian patient without underlying disease.
Case description
A 62-year-old patient with no underlying disease was admitted to the emergency (day 0) for dyspnea and fever at 38°C. SpO2 in ambient air was at 87%. At arterial blood gas, the PaO2 was 65 mmHg and the SaO2 was 94%. The chest CT angiography showed an aspect of SARS-COV2 pneumonia with severe involvement estimated between 50 and 75%. In biological investigation, C-Reactive Protein (CRP) was 113 mg/L and D-dimers was 3176 ng/mL. The patient was put on oxygen therapy with a high concentration mask with antibiotic therapy based on Ceftriaxone and azithromycin and corticosteroid therapy as well as preventive anticoagulation with Enoxaparin 0.6 mL. On day +3, the SpO2 decreased under 90% and the PaO2 was 79 mmHg requiring the use of positive pressure ventilation alternating with ventilation by high concentration mask. On day +4, he was transferred to the intensive care unit. At admission, he was afebrile with stable hemodynamic constants. Blood culture, cytobacteriological and mycological examinations of urine and sputum, and protected tracheal samples are performed with stopping of azithromycin for treatment duration of 5 days and continuation of Ceftriaxone for 10 days.
Laboratory test results showed Creatinemia at 73 µmol/L, Urea at 5.6 mmol/L, CRP at 240 mg/L and Procalcitonin at 0, 29 µg/L. The buccal and nasal sites were colonized with Candida (C.) yeasts (C. albicans, C. parapsilosis, and C. famata). Mycological examination of the sputum and the protected tracheal sample revealed the presence of Kodamaea ohmeri yeasts. Thus, the patient was put on Voriconazole.
On day +7, he presented a respiratory distress with polypnea and an SpO2 of 60% requiring the tracheal intubation and the initiation of tazocillin and amiklin. In face of non improvement, the antibiotic therapy was changed on Day +11 to Imipeneme and vancomycin. His blood count showed moderate anemia (Hb = 11.6 g / dl), the D-dimer dropped to 1192 ng/mL with increased serum ferritin to 866 µmol/L. On day +16, the patient’s condition deteriorated rapidly leading to death from severe sepsis post COVID-19 on the same day.
Methods
Sputum and the protected tracheal samples were cultured on Sabouraud medium, and CHROM agar medium (Beckton Dickinson, Paris, France). The identification was based on the results of the chlamydosporulation test on Tween agar medium, Vitek® 2 compact (YST bio Merieux, France), and PCR sequencing of the Internally Transcribed Spacer (ITS). The primers used were Its1 (5’-TCCGTAGGTGAACCTGCGG-3’) and Its4 (5’-TCCTCCGCTTATTGATATGC-3’) as described by Mtibaa et al [6]. The analysis of the sequences was then done with the Blast tool. The antifungal susceptibility of the three isolates was determined by the guidelines of the Clinical and Laboratory Standards Institute (CLSI) by using RPMI 1640 medium and E test.
Results
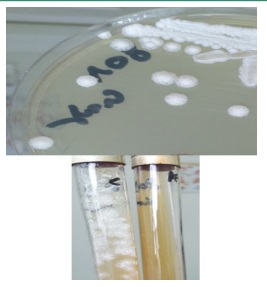
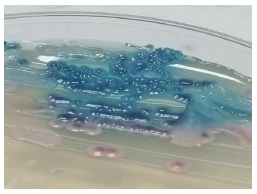
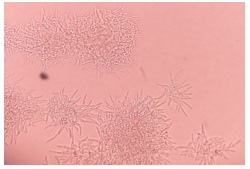
Colonies on Sabouraud medium were creamy and smooth after 48 hours of incubation at 37°C. After 96 hours, they became radiated with a central dome (Figure 1). The quantification was greater than 102 / mL of dilution. On CHROM agar medium, the isolates produced pink-colored colonies which changed to blue after 48 h of incubation (Figure 2). On Tween agar, the strain showed small spores with tapering pseudofilamentation after 24 hours at 27°C (Figure 3). The isolate was identified Kodamea (Pichia) ohmeri by Vitek system (Bio Mérieux) after 24 hours.
The molecular identification showed that sequences matched in 99% with previous K. ohmeri in Gene Bank. The strain was sensitive to amphotericin B (MIC = 0.47 ug/ml), to caspofungin (MIC = 0.25 ug/ml) with intermediate sensitivity to fluconazole (MIC = 4 ug/ml).
Discussion
Kodamaea (Pichia) ohmeri belongs to the Ascomycetae class and is rarely occurring yeast that can be isolated from environmental sources, such as seawater, fruits, and pools [10]. The first isolate of K.ohmeri was reported in 1984 from a pleural fluid sample but it was considered a contaminant. In the same year, fungemia in a 48-year-old diabetic patient with immunosuppression due to renal transplantation was reported, who subsequently died from the infection [9]. Since then, more infections with this yeast have been reported considering it a true clinical pathogen, especially in patients with underlying immunosuppression [9,6]. Here, we reported first case of co-infection with Sars-Cov2 and Kodamaea ohmeri in Tunisian patient without underlying disease.
Table 1 summarizes shows that the majority of case reports are from Asia (Korea, Turkey, India), 23/36, 64%. There are a few descriptions from North America 7/36, 20%, some from Europe 6/36, 16%, and Arabic region (Turkey 2; Lebanon 1; Qatar 1; Kuwait 1). The largest series reported in the same site included 38 patients from a tertiary care hospital in North India, with 78.9% of these cases corresponding to neonates in intensive care units. In that series, the attributed mortality from this infection was 31.8%, but there are other reports of mortality as high as 50% in the pediatric population.
In the literature, the age of patients ranged from new born to 84 years old, the mean age was 42.6 years, 64.1% were males. Among the patients infected with K. ohmeri, 60.6% had already received an antibiotherapy treatment with antibiotics. All patients with infection of kodemea ohmeri have one or more underlying conditions or risk factors. They including are immunocompromised due to malignant hematological or solid tumors, post-chemotherapy neutropenia, immunosuppressive treatments, diabetes, or chronic renal failure [2]. The Majority were catheter-related and catheter removal was key to healing. Pre-exposure to systemic antibiotics was also reported in many cases. Systemic infections due to K. ohmeri are rarely reported in immunocompetent subjects.
Table 1:Clinical characteristics of 36 cases of K. ohmeriinfections in the literature [4]. (Kanno et al 2017)
Age/Gender |
Background |
Antifungal |
Source |
Outcome |
Country |
Year |
48/F |
DM,renal transplantation, |
FLZ then AMB |
Fungemia |
Died |
USA |
1998 |
71/M |
DM, pacemaker, CVC, endocarditis |
AMB |
Fungemia |
Died |
Netherlands |
2000 |
42/M |
IV heroin use, HCV |
AMB and 5-FC |
Endocarditis |
Survived |
Portugal |
2002 |
76/M |
Prosthetic valve, PM |
FLZ, then AMB |
Endocarditis |
Survived |
USA |
2002 |
84/M |
Right maxillary sinus SCC, CVC |
FLZ, and then AMB |
Catheter |
Died |
Japan |
2002 |
59/M |
VP shunt infection, phlebitis |
AMB |
Fungemia |
Survived |
Korea |
2003 |
14/M |
ALL, neutropenia, CVC |
FLZ |
Fungemia |
Survived |
2004 |
|
8 month/M |
Encephalitis,intravenous, catheter |
FLZ |
Fungemia |
Died |
Turkey |
2005 |
10/M |
B cell-ALL, neutropenia, CVC |
AMB |
Fungemia |
Survived |
2005 |
|
Neonate/F |
Prematurity, necrotizing enterocolitis, intravenous catheter |
FLCZ, AMB then liposomal AMB |
Fungemia |
Survived |
USA |
2006 |
58/F |
CML, CVC, chemotherapy |
AMB |
Fungemia |
Survived |
Brazil |
2006 |
11/M |
Burkitt’s lymphoma, neutropenia, CVC |
FLZ |
Fungemia + phlebetis |
Died |
Korea |
2007 |
41/M |
Alcoholic ketoacidosis, TB, CVC |
FLZ |
Fungemia+ catheter |
Survived |
||
47/M |
DM, CRF, pneumonia, CVC |
CVC removal, FLZ then AMB |
Fungemia |
Died |
||
4/f |
Tetralogy of Fallot, CVC |
FLCZ+AMB |
Fungemia + catheter |
Died |
||
Neonate/F |
Prematurity, CVC |
- |
Fungemia |
Survived |
||
82/F |
DM, CRF, surgery |
AMB |
Fungemia |
Died |
Spain |
2007 |
5months /M |
Short gut syndrome, hepatic insufficiency, broviac catheter |
FLZ, AMB |
Fungemia |
Survived |
USA |
2007 |
38/F |
AML, hemochromatosis, chemotherapy, intravenous, catheter |
AMB |
Fungemia +catheter |
Survived |
Lebanon |
2008 |
3/F |
Ascaris peritonitis, CVC, multiple antibiotics |
liposomal AMB |
Fungemia |
Survived |
Brazil |
2009 |
71/F |
DM, tinea pedis, cellulitis |
FLZ then AMB |
Fungemia |
Survived |
Taiwan |
2009 |
Neonate/M |
prematurity, arterial and venous catheter |
AMB |
Fungemia |
Died |
India |
2009 |
55/M |
Alcoholic hepatitis, duodenal ulcer, CVC |
FLZ then caspofungin |
Fungemia |
Survived |
Taiwan |
2010 |
34/M |
Asthma, alcohol abuse, thrombophlebitis, venous catheter, transesophageal fistula |
Micafungin |
Fungemia |
Survived |
USA |
2010 |
58/F |
SCC of esophagus, nosocomial pneumonia, PICC |
FLZ |
Fungemia +catheter |
Died |
Taiwan |
2010 |
43/M |
Rheumatic heart disease, endocarditid HBV carrier |
ITZ |
Endocarditis |
Survived |
China |
2010 |
Neonate/M |
Enterocolitis, endocarditis, intravenous catheter |
AMB |
Endocarditis |
Died |
India |
2011 |
Neonate/F |
Prematurity |
AMB |
Fungemia |
Survived |
Kuwait |
2011 |
46/M |
Pneumonia, HBV carrier, MODS, CVC |
FLZ then caspofungin, |
fungemia |
Survived |
China |
2013 |
62/M |
Gastric perforation, pneumonia, acute renal insufficiency, intravenous catheter |
VRZ then AMB |
Fungemia |
Died |
||
75/M |
Post total colectomy |
AMB |
Fungemia |
Survived |
India |
2015 |
80/M |
AMI, retrocolic gastric bypass surgery, and splenectomy due to colon cancer, CVC, IABP |
FLZ then L-AMB |
Fungemia |
Died |
Italy |
2015 |
58/M |
RA, acute pancreatitis, CVC |
micafungin |
Fungemia +catheter |
Survived |
Japan |
2017 |
81/M |
Mild cognitive disorders |
Caspofungin then voriconazol |
Fungemia |
Survived |
France |
2019 |
25/M |
hypoxic-ischemic encephalopathy, pulmonary infection, and multiple organ dysfunction |
Caspofungin |
Catheter |
Survived |
China |
2020 |
47/M |
Diabetes |
FLZ then L-AMB |
Fungemia |
Survived |
Italia |
2021 |
MIC: Minimum Inhibitory Concentration; AMB: Amphotericin B; FLCZ: Fluconazole; DM: Diabetes Mellitus; CVC: Central Venous Catheter; ND: No Data Reported; PM: Pacemaker; HCV: Hepatitis C Virus; SCC: Squamous Cell Carcinoma; VP Shunt: Ventriculoperitoneal Shunt; ALL: Acute Lymphoblastic Leukemia; CML: Chronic Myelogenous Leukemia; TB: Tuberculosis; CRF: Chronic Renal Failure; L-AMB: Liposomal Amphotericin B; PICC: Peripherally Inserted Central Venous Catheter; HBV: Hepatitis B Virus; ITCZ: Itraconazole; SDD: Susceptible Dose Dependent; MVR: Mitral Valve Replacement; TVP: Tricuspid Valve Plasty; MODS: Multiple Organ Dysfunction Syndrome; VRCZ: Voriconazole; AMI: Acute Myocardial Infarction; IABP: Intraaortic Balloon Pumping; RA: Rheumatoid Arthritis.
For the isolation of K. ohmeri, blood cultures were used in 81.8% [9], cultures from nail specimens in 9.1%, and urine culture, peritoneal dialysis dialysate, oral swab, and deep wound infection in 2.7% each. On solid media, K.ohmeri forms Candida-like colonies and changes their color from pink to blue within 48 h on CHROM agar medium. Of the 5 species recognized in the Kodamaea genus, only K.ohmeri can grow at 37°C and cause human disease. Molecular diagnosis is available through the amplification and sequencing of the ITS2 region localized on the rRNA gene of the 5.8S and 28S subunit.
Our isolate in the present study was sensitive to AMB and caspofungin and had intermediate sensitivity to Fluconazole. The Literature reveals that K. ohmeri isolates have a low MIC for all antifungal agents except for a high range of MICs against fluconazole and caspofungin in some isolates [3,8]. Yuling et al 2013, found poor results in patients treated with fluconazole as the sole antifungal despite in vitro sensitivity. Treatment with amphotericin B, alone or in combination with flucytosine/fluconazole, produced more favorable results, with a survival rate of 66.7%. Echinocandin treatment was successful in 3 patients.
The study by Yang et al 2008 showed that the MICs of all isolates were sensitive to amphotericin B. Of the 13 antifungal susceptibility results available, seven isolates were sensitive to fluconazole (MIC 2.0 to 8, 0 mg/l) and six were intermediate including the remaining isolate (MIC 16.0 to 32.0 mg/l) [3,6,10].
Two cases in the pediatric intensive care unit were the first outbreak of K. ohmeri infection [1,8]. Both patients were infected with K. ohmeri sensitive to fluconazole (MIC 8.0 mg/l). One survived after treatment with amphotericin B, but the other died despite treatment with fluconazole. So far, all isolates of K. ohmeri are susceptible to amphotericin B [4,6,11]. Since amphotericin B is nephrotoxic in a dose-dependent manner, the use of a lower dose is preferred. However, in severely immunocompromised patients with systemic mycosis, a less favorable clinical outcome may result from the use of low doses of amphotericin.
The most common antifungals for the treatment of K ohmeri infections were amphotericin B in 59.5%, fluconazole in 35.1%, micafungin in 13.5%, voriconazole in 5,4%, and caspofungin and 5-flucytosine in 2.7% each, while 8.1% received no treatment [3].
According to the Clinical Practice Guidelines for the Management of Candidiasis from the Infectious Diseases Society of America, an echinocandin is recommended as initial therapy [12], (yuling et al 2013), fluconazole, the loading dose of 800 mg (12 mg/kg), then 400 mg (6 mg/kg) per day, is an alternative for patients who are not seriously ill and who have never been exposed to azoles.
Conclusion
K. ohmeri can cause life-threatening infections. Favorable results for this potentially fatal fungal infection are likely to be associated with early diagnosis and optimal antifungal regimens. Fluconazole is the least effective against K. ohmeri infections. Although more study is needed to establish the optimal antifungal regimens.
Declaration
Ethics approval and consent to participate: Not applicable.
Availability of data and materials: Not applicable.
Conflict of interest: This case report is not supported by any financial or personal interests.
References
- Al Sweih N, Khan ZU, Ahmad S, Devarajan L, Khan S, Joseph L, et al. Kodamaea ohmeri as an emerging pathogen: A case report and review of the literature. Medical mycology. 2011; 49: 766- 770.
- Diallo K, Lefevre B, Cadelis G, Gallois JC, Gandon F, Nicolas M, et al. A case report of fungemia due to Kodamaea ohmeri. BMC infectious diseases. 2019; 19: 1-3.
- Ioannou P, Papakitsou I. Kodamaea ohmeri infections in humans: A systematic review. Mycoses. 2020; 63: 636-643.
- Kanno Y, Wakabayashi Y, Ikeda M, Tatsuno K, Misawa Y, Sato T, et al. Catheter-related bloodstream infection caused by Kodamaeaohmeri: A case report and literature review. Journal of Infection and Chemotherapy. 2017; 23: 410-414.
- Koebel C, Letscher Bru V, Varela D I, Waller J, Marescaux J, Dupeyron P, et al. À propos d’un cas de fongémie à Kodamaea ohmeri: Difficultés d’identification et revue de la littérature. Journal de mycologie médicale. 2008; 18: 106-110.
- Mtibaa L, Souid H, Jemli B, Hajjej Z, Halweni C, Rebai A, et al. Kodamaea ohmeri, An Emerging Yeast in Tunisia: First Identification in Three Case Reports and Literature Review. Med Microb Diagn. 2019; 8: 2.
- Menon T, Herrera M, Periasamy S, Palanivelu V, Sikhamani R., Wickes B. Oral candidiasis caused by Kodamaea ohmeri in a HIV patient in Chennai, India. Mycoses. 2010; 53: 458-459.
- Sundaram PS, Bijulal S, Tharakan JA, Antony M. Kodamaea ohmeri tricuspid valve endocarditis with right ventricular inflow obstruction in a neonate with structurally normal heart. Annals of pediatric cardiology. 2011; 4: 77.
- Tashiro A, Nei T, Sugimoto R, Watanabe A, Hagiwara J, Takiguchi T, et al. Kodamaea ohmeri fungemia in severe burn: Case study and literature review. Medical mycology case reports. 2018; 22: 21-23.
- Vivas R, Beltran C, Munera MI, Trujillo M, Restrepo A, Garcés C, et al. Fungemia due to Kodamaea ohmeri in a young infant and review of the literature. Medical mycology case reports. 2016; 13: 5-8.
- Yanghua Q, Weiwei W, Yang L, Jian X, Qian S. Isolation, identification, and antifungal susceptibility test for Kodamaea ohmeri: A case report on endocarditis. Journal of Medical Colleges of PLA. 2010; 25: 252-256.
- Xiao Y, Kang M, Tang Y. Kodamaea ohmeri as an emerging pathogen in mainland China: 3 case reports and literature review. Laboratory Medicine. 2013; 44: 1-9.
- HOU, Chenrui. Catheter-related bloodstream infection caused by Kodamaea ohmeri in China. Infection Prevention in Practice, 2020; 1: 100006.
- Prabhu K, Chinniah TR. A case report of fungemia due to Kodamea ohmeri. Sri Lankan Journal of Infectious Diseases, 2021; 11(1): 42-46