Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Brain metastases in anal squamous cell carcinoma: Case report and review of the literature
Midhun Malla1; Hassan Hatoum2; Sagila George2
1 Department of Internal Medicine, Division of Hematology/Oncology, West Virginia University, USA.
2 Department of Internal Medicine, Division of Hematology/Oncology, University of Oklahoma Health Sciences, USA.
*Corresponding Author: Midhun Malla
Assistant professor, Department of Internal Medicine,
Section of Department of Hematology/Oncology, West
Virginia University, USA.
Email: Midhun-malla@hsc.wvu.edu
Received : Dec 24, 2021
Accepted : Jan 14, 2022
Published : Jan 21, 2022
Archived : www.jcimcr.org
Copyright : © Malla M (2022).
Citation: Malla M, Hatoum H, George S. Brain metastases in anal squamous cell carcinoma: Case report and review of the literature. J Clin Images Med Case Rep. 2022; 3(1): 1602.
Introduction
Anal cancer is a rare malignancy and constitutes only 2.5% of all digestive system cancers in the USA. There is an uptrend in the incidence of anal cancers in the general population, which could be attributed to human papilloma virus and human immunodeficiency virus infections, increase in lifetime number of sexual partners, receptive anal intercourse, number of patients on immunosuppressive treatments for organ transplants, cigarette smoking and genital warts [1]. Rate of metastasis from squamous cell cancer (SCC) of the anus is minimal and liver and lungs are the most commonly involved organs [2]. Metastasis to the brain is extremely rare [3]. So, far only 5 cases have been reported in the literature. In this manuscript we report an unusual presentation of brain only metastasis from anal cancer and a review of the literature on this topic. To our knowledge, this case represents the sixth reported case of anal cancer with brain metastasis in the literature. Moreover, this is the first reported case of anal cancer that presented with brain only metastasis within 3 months while in remission after completing treatment with chemoradiotherapy (CRT) for the primary tumor.
Case presentation
Our patient is a 54 year old woman who is a chronic smoker initially presented in September 2016 with a 6 month history of urinary retention, worsening constipation and perianal pain. Digital rectal exam showed rectal tenderness and a palpable mass in anal canal. Colonoscopy findings include a 7 cm fungating non obstructing mass in distal rectum protruding from anal canal. Biopsy was positive for poorly differentiated squamous cell cancer (SCC) with Ki-67 at 80%. CRT with 5-Fluorouracil and Mitomycin was initiated for T3N0M0 (Stage IIB) SCC of the anal canal. Mitomycin dose was omitted on day 29 due to pancytopenia from myelosuppression. Six weeks after completion of CRT (December 2016), patient was found to be in remission when anoscopy demonstrated complete resolution of malignancy
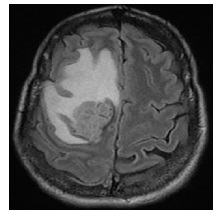
Three months later (March 2017), she presented with left arm weakness, numbness and reported dropping things from her left hand. Further work up with computed tomography (CT) and magnetic resonance imaging (MRI) brain showed a T1 hyper intense heterogeneously enhancing 2.9 X 2.9 X 3.2 cm right frontal mass associated with edema along with a 9 mm right to left midline shift (Figure 1). CT chest abdomen pelvis (CT-CAP) was negative for metastasis. She underwent right craniotomy for resection of mass and pathology was suggestive of poorly differentiated SCC consistent with her primary diagnosis. Immunohistochemistry (IHC) for p63 and pancytokeratin was positive in both primary and metastatic sites. P16 staining was also strongly positive. She underwent gamma knife radiosurgery in April 2017 and her post-treatment course was complicated with scalp wound infection with methicillin sensitive staphylococcus aureus infection. She was started on antibiotics and underwent wound washout, bone excision and had a mesh placement. She underwent WBRT in June 2017 with 15 fractions and 3000 Gy followed by intensity modulated radiation therapy (IMRT). She was lost to follow up after and re-presented in February 2018 with worsening neurological symptoms. MRI brain showed progression of disease in brain and she received a second gamma knife treatment. In September 2018, she had another local progression in brain and was admitted to the hospital for further treatments. Foundation testing revealed no known targetable mutations, tumor was microsatellite stable (MSS) along with a low tumor mutation burden (TMB). Option of starting immunotherapy was discussed with patient. Patient declined further therapy and opted for hospice in December 2018.
Discussion
The number of newly diagnosed anal cancer cases have been increasing with an average of 2.2% rise each year over the last 10 years. Mortality has also been rising on an average of 2.9% each year from 2006-2015. Females are commonly affected when compared to males (1.9:1) [1]. Based on data from SEER 2008-2014, five year survival of patients with anal cancer is 67.4%. Metastatic anal cancers are associated with a poor prognosis with documented median survival rates varying from 8 to 34 months. 5 year survival drops significantly down to 15% for metastatic squamous cell cancers and 7% for non-squamous cancers of anal canal [4].
It is important to accurately identify originating location of the cancer. Since anal canal is only around 5 cm long, tumors arising from anus can be seen extending to the rectum or vice versa. Our patient was originally thought to have rectal cancer, but on further review was found to have tumor originating from anal canal. Hernando Cubero [5] reported a case of brain metastasis secondary to basaloid undifferentiated anal carcinoma. On review of the manuscript, the cancer was identified within 6 cm of anal verge and it was unclear whether the tumor was originating from anal canal or from distal rectum. Their patient also presented with multiple pulmonary and liver metastases, both of which are the commonly involved organs affected with metastases from cancers arising above dentate line in the intestine. Since pathology was suggestive of basaloid variant, it was deemed to be originating from anal canal and managed accordingly.
To date, only 5 cases of metastatic anal SCC to the brain have been reported in the literature. To our knowledge, our case is the first reported case of localized anal SCC that was treated with Nigro protocol with 5-FU and Mitomycin and RT, and presented with brain metastasis shortly after achieving complete response. Interestingly, our patient did not have evidence of disease elsewhere in the body except in the brain at progression. The case reported by Davidson et al [6] in 1991 had a similar presentation with brain only progression eight years after frontline therapy; however, patient was managed with surgery and not CRT, unlike in our case. With the exception of case reported by Davidson et al, rest of the reported cases of anal cancer developed brain metastasis within 15 months of completion of initial therapy
In our patient, solitary brain lesion was initially presumed to be a primary CNS malignancy as the appearance of tumor on MRI brain was concerning for GBM. Metastasis from anal SCC was initially not considered in the differential diagnosis due to early onset of brain metastasis within 3 months of completion of curative therapy and that the patient had no evidence of disease on CT CAP. To our surprise, pathology was positive for anal SCC. We found an interesting observation that metastatic recurrences to the brain were solitary in 3 of the 5 reported cases in the literature, similar to our patient. The other two reported cases were stage 4 at diagnosis with metastases to liver at presentation and developed brain metastases either during or shortly after curative treatment. To our knowledge, none of these cases had features on MRI brain that mimicked GBM. Our case stresses the importance of obtaining tissue in the setting of management of recurrent tumors.
Prognostic factor analysis for anal cancer from prospective data is limited. A phase III study on 110 patients by The European Organization for Research and Treatment of Cancer (EORTC) reported that lymph node status, skin ulceration and male gender were associated with poor overall survival (OS) and local disease control. Another larger prospective database study, RTOG 98-11, confirmed male sex, and positive lymph node status as poor prognostic factors and also showed that tumor diameter >5 cm was associated with reduced DFS and OS [9]. This study had shown that patients with >5cm tumor and N1 status has only 30% chance of being diseasefree at 3 years [10]. The other reported prognostic factors in the literature include age over 65 years, advanced T stage and poorly differentiated histology. 11 Our patient’s tumor size is >5 cm (7 cm), has advanced T stage (T3) with poorly differentiated histology, both of which explains the shorter disease free survival of 3 months. Ki-67% was also elevated in our patient at 80%. Based on extrapolation of data from other solid tumors like breast cancer and neuroendocrine tumors, we think that elevated Ki-67% could also have played a role in poor prognosis of our patient.
In addition to stage and grade, the tumor human papilloma virus (HPV) status has been identified as a significant prognostic and predictive factor in anal SCC treated with CRT. In a retrospective report, Yhim et al, had described the outcomes of 47 anal SCC patients treated with CRT based on HPV status. In their report, 77% of the patients had HPV positive tumors; of those 66% were genotype 16 (HPV-16 positive). Patients with HPV-16 positive tumors, determined by HPV DNA chip method, had a significantly better 4-year progression-free survival (PFS, 63.1% vs. 15.6%, p < 0.001) and overall survival (OS, 84.6% vs. 39.8%, p < 0.008) compared to patients with HPV-16 negative tumors. Patients with p16-positive tumor, determined by IHC, also, had a better 4-year PFS (52.5% vs. 25.0%, p=0.014) than those with p16-negative tumor. The median follow-up was 51.7 months (range: 5.1–136.0 mos). The authors, also, concluded, that IHC may be used as a surrogate test for determining HPV status instead of HPV DNA chip method. Interestingly, our patient had strong positivity for P16 stain [12].
Currently, PDL-1 expression has been identified as a predictive factor for immunotherapy treatment with PD-1 inhibitors, Pembrolizumab and Nivolumab. Additional markers include tumor suppressor genes p53 and p21 that were found to have a potential prognostic value in patients with anal SCC treated with CRT [13]. However, the results were not consistent in other studies, which could be due to either small sample size or methodological differences like the choice of antibodies used in immunohistochemical analysis [14].
Hence, we recommend medical oncologists to assess the HPV-16 status for both prognostic and predictive purposes, and PDL-1 expression as a predictive marker. Nowadays, in the era of molecular advancements and tumor agnostic therapies, comprehensive genomic profiling is an essential tool for identifying potential targetable mutations and develop novel therapeutic options for the patients diagnosed with anal SCC.
Table 1:List of published cases of anal cancers in literature. (APR: abdominoperineal resection, BM- brain metastasis, C- Caucasian, GK- gamma knife, NA- not available, RT- radiation therapy, SCC- Squamous cell carcinoma, OS- overall survival).
First Author |
Year of publication |
Age/Sex |
Race |
Size (cm) |
Histology |
Stage at diagnosis |
Location of metastasis at diagnosis |
Initial therapy |
Months to BM |
Location of BM |
Treatment of BM |
Median OS |
Klotz 3 |
1966 |
NA |
NA |
NA |
Cloacogenic |
NA |
NA |
NA |
NA |
NA |
NA |
NA |
Davidson 6 |
1991 |
61/F |
NA |
|
SCC |
II |
NA |
Surgery |
96 |
NA |
Surgery +RT |
NA |
Rughani 7 |
2011 |
63/F |
C |
2.7 x 4.7 |
SCC |
IV |
Liver |
Capecitabine+RT |
0.5 |
Parietal region |
Surgery +RT followed by WBRT |
3.5 mos. |
Gassman 8 |
2012 |
67/M |
NA |
4 |
SCC |
IV |
Liver |
Cisplatin+ 5-FU +RT followed by APR and liver resection |
9 |
Sphenoid sinus and temporal bones |
None |
NA |
Hernando-cubero 5 |
2014 |
69/M |
NA |
NA |
Basaloid undifferentiated carcinoma |
IV |
Liver, lung, bone,mesorectum |
Cisplatin +5FU |
14 |
Cerebrum and cerebellum |
|
3 mos. |
Present case |
|
56/F |
C |
7 |
Poorly differentiated SCC |
II (T3N0M0) |
Negative |
5FU+Mitomycin +RT |
3 |
Frontal lobe |
Resection + GK |
18 mos. |
Conclusion
Anal SCC can present with brain only metastasis shortly after achieving remission from initial CRT for limited stage disease. The clinical presentation can mimic stroke. The radiologic presentation of solitary brain metastasis can mimic other brain tumors such as GBM especially for those patients who do not have evidence of disease recurrence elsewhere on imaging. Treating oncologists need to be aware of this entity and should include it in the differential diagnosis when patients with a history of anal cancer present with new neurologic symptoms even if the cancer seems to be in remission on rest of the body imaging. We also realize the importance of IHC testing on all newly diagnosed anal SCC tumors for HPV (16 &18 genotypes) due to its potential role as a predictive and prognostic marker and PDL-1 testing as a predictive marker for response to immunotherapy in locally advanced or metastatic anal SCC.
Declarations
About the author: In the last 12 months, Dr. Midhun Malla has received grant funding from National Institute of General Medical Sciences-NIH (Grant # 5U54GM104942-05). The content is solely the responsibility of the author and does not necessarily represent the official views of the National Institutes of Health. He is on speaker’s bureau for Natera, received honoraria from Omni Health, and Curio Science.
Conflicts of Interest: The rest of the authors have no conflicts of interest to disclose.
Funding: There is no grant support or funding received for completion of this case report.
References
- American Cancer Society. Cancer Facts & Figures. 2018.
- Palefsky JM. Anal human papillomavirus infection and anal cancer in HIV-positive individuals: an emerging problem. Aids. 1994; 8(3): 283-296.
- Klotz Jr RG, Pamukcoglu T, Souilliard DH. Transitional cloacogenic carcinoma of the anal canal. Clinicopathologic study of three hundred seventy-three cases. Cancer. 1967; 20(10): 1727-1745.
- American Cancer Society. Cancer Facts & Figures. 2017.
- Hernando-Cubero J, Alonso-Orduña V, Hernandez-Garcia A, et al. Brain metastasis in basaloid undifferentiated anal carcinoma: A case report. Oncology letters. 2014; 7(4): 1276-1278.
- Davidson NG, Yong PP. Brain metastasis from basaloid carcinoma of the anal canal 8 years after abdominoperineal resection. Eur J Surg Oncol. 1991; 17: 227-230.
- Rughani AI, Lin C, Tranmer BI, et al. Anal cancer with cerebral metastasis: a case report. Journal of neuro-oncology. 2011; 101(1): 141-143.
- Austin Gassman A, Fernando E, Holmes CJ, et al. Development of cerebral metastasis after medical and surgical treatment of anal squamous cell carcinoma. Case reports in oncological medicine, 2012.
- Bartelink H, Roelofsen F, Eschwege F, et al. Concomitant radiotherapy and chemotherapy is superior to radiotherapy alone in the treatment of locally advanced anal cancer: results of a phase III randomized trial of the European Organization for Research and Treatment of Cancer Radiotherapy and Gastrointestinal Cooperative Groups. Journal of clinical oncology. 1997; 15(5): 2040-2049.
- Ajani JA, Winter KA, Gunderson LL, et al. Prognostic factors derived from a prospective database dictate clinical biology of anal cancer: the intergroup trial (RTOG 98‐11). Cancer. 2010; 116(17): 4007-4013.
- Dewdney A, Rao S. Metastatic squamous cell carcinoma of the anus: time for a shift in the treatment paradigm? ISRN oncology. 2012.
- Yhim HY, Lee NR, et al. The prognostic significance of tumor human papillomavirus status for patients with anal squamous cell carcinoma treated with combined chemoradiotherapy. International journal of cancer. 2011; 129(7): 1752-1760.
- Morris VK, Ciombor KK, Salem ME, et al. NCI9673: A multiinstitutional eETCTN phase II study of nivolumab in refractory metastatic squamous cell carcinoma of the anal canal (SCCA). 2016.
- Lampejo T, Kavanagh D, Clark J, et al. Prognostic biomarkers in squamous cell carcinoma of the anus: a systematic review. British journal of cancer, 2010; 103(12): 1858.