Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Encephaloma, neurocysticercosis, angiocavernoma or others: A case report
Guo Zhi-bao; Yang Yi*
Jiangsu Provincial Key Laboratory of Critical Care Medicine, Department of Critical Care Medicine, Zhongda Hospital, School of Medicine, Southeast University, Nanjing, 210009, Jiangsu, China.
*Corresponding Author: Yang Yi
Jiangsu Provincial Key Laboratory of Critical Care
Medicine, Department of Critical Care Medicine,
Zhongda Hospital, School of Medicine, Southeast
University, Nanjing, 210009, Jiangsu, China.
Email: yiyiyang2004@163.com
Received : Dec 16, 2021
Accepted : Jan 18, 2022
Published : Jan 25, 2022
Archived : www.jcimcr.org
Copyright : © Yi Y (2022).
Abstract
It is difficult to make a diagnosis for this case. A 38-year-old man suffered from mild cognitive dysfunction for 3 months and intermittent headache for 20 days, and his focal neurological signs were unconspicuous. Cerebrospinal fluid (CSF) showed normal biochemical and negative bacteriologic examinations. Parasitological tests of blood and CSF were negative. Imaging examination showed multiple intracranial lesions, which possessed the characteristics of multidate, fluid levels, bleeding, and without edema zone. The possible diagnosis included metastatic encephaloma, neurocysticercosis and angiocavernoma or other diseases. The patient was treated with dehydration of intracraninal pressure and the neurological symptoms were improved slightly. For this case, with slight abnormal clinical symptoms and complicated imaging findings, it is difficult to diagnose, and brain biopsy may be the only method for the definitive diagnosis.
Citation: Zhi-bao G, Yi Y. Encephaloma, neurocysticercosis, angiocavernoma or others: A case report. J Clin Images Med Case Rep. 2022; 3(1): 1611.
Case report
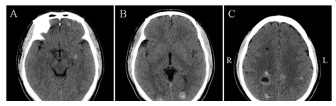
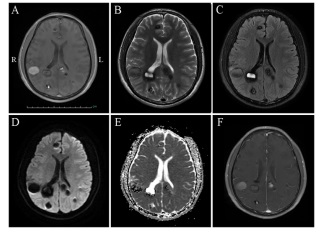
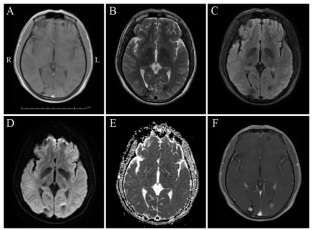
A 38-year-old man, with memory deterioration for 3 months and intermittent headache for 20 days, came to our hospital for treatment. He had no fever, vomiting, limb weakness, seizure or impairment of consciousness. His medical history included hypertension for 12 years, chronic gastritis for 5 years, drinking and cigarette smoking for 20 years, and breeding livestock for 20 years. Physical examination revealed clear consciousness, decreased memory and computational power. Meningeal irritation sign was negative. The skin of the left knee had melanin deposition. Cerebrospinal fluid revealed elevated intracranial pressure, normal biochemical and negative bacteriologic examinations. Parasitological tests of blood and CSF were negative. Brain computed tomography scan (Figure 1) and magnetic resonance imaging (MRI) scan (Figure 2, 3) showed multiple lesions in frontal and occipital lobe. The patient was treated with dehydration of intracraninal pressure and the neurological symptoms were improved slightly.
After multidisciplinary consultation, these brain lesion characterization for this patient was considered as neurocysticercosis, metastatic encephaloma, angiocavernoma or others. Brain biopsy was suggested to get definitive diagnosis. Due to poor economic situation, the patient refused to adopt brain biopsy, and discharged from hospital voluntarily. 2 months after hospital discharge, the patient’s condition was fine from a return visit by telephone. 2.5 years later, the patient died for an unknown reason.
Discussion
In present study, the patient’s clinical manifestations did not match his imaging examinations to a great extent. The patient had slight abnormal clinical symptoms, but his imaging findings was complicated. With multidisciplinary consultation, metastatic encephaloma, neurocysticercosis, or angiocavernoma were considered.
Metastatic encephaloma are the most common type of neoplasm in adults, accounting for approximately 10% of adult cancer patients [1], and are associated with high mortality as well as poor prognosis [2]. The most common primary malignancies that metastasize to the brain are lung, breast, melanoma, colon cancers, whereas other malignant tumors such as prostate, liver, bladder, pancreatic, and uterine have a lower propensity to metastasize the brain [3,4].Hematogenous spread and geographic invasion are the two common forms of central nervous system metastasis. Any encephalic region of the brain can be affected by metastatic tumor, however, the most common site of disease involvement is the gray-white matter junction [5,6]. The most common clinical symptoms in patients with brain metastases are headache and epilepsy. Asymptomatic patients usually detect lesions on imaging scans. Neuroimaging plays an important role in patients with metastatic encephaloma. MR imaging is currently considered as the optimal imaging modality for the diagnosis of metastatic disease, monitoring of therapeutic response, and assessment for disease progression [7]. In general, a majority of lesions are isointense or slightly hypointense relative to normal brain tissue on T1-weighted imaging. Hyperintensity on precontrast T1-weighted MR imaging can occurin in late subacute hemorrhage, protenaceous fluid, focal macrocellular fat, or melanin deposition. Most of cerebral metastases demonstrate prolonged T2 relation times and are, therefore, hyperintense on T2-weighted sequences. The exception, however, is that densely cellular tumors or mucin-secreting neoplasms can appear hypointense on T2-weighted imaging. After the administration of contrast media, most metastases dramatically enhance on contrast-enhanced T1-weighted MR imaging, and typical imaging findings are well-circumscribed nodules with varying internal contents, including hemorrhagic fluid, mucinous material, or necrotic debris [7,8], In present study, the patient had intermittent headache for 20 days, and his MR imaging showed multiple, multi-phase, scattered lesions with liquid flat in frontal and occipital lobe. Physical examination revealed that skin of the left knee had melanin deposition. Based on the above evidence, malignant metastasis, especially melanoma, can not be ruled out.
Neurocysticercosis, which is typically caused by the tapeworm Taenia solium after ingesting contaminated food or water, is one of the most common parasitic diseases of the nervous system in humans [9]. The clinical manifestations of neurocysticercosis can vary from completely asymptomatic infection to death. The nervous system manifestations usually include seizures, headache, intracranial hypertension, focal neurological signs and mental disorder [10]. Diagnosis of neurocysticercosis mainly includes clinical manifestations, serum immunology and neuroimaging [9]. In general, canonical neurocysticercosis cases can be easily diagnosed by neuroimaging of the brain. On the other hand, if the clinical presentations are atypical, neuroimaging findings are short of specificity, and immunodiagnostic tests show suboptimum predictive values, diagnosis of these neurocysticercosis cases will be difficult. In present case, the patient’s clinical manifestations were nonspecific, and parasitological tests of blood and CSF were negative. However, the neuroimaging finds were similar to that of neurocysticercosis. Therefore, the possible diagnosis of this patient cannot exclude neurocysticercosis.
Angiocavernoma presents in 0.4-0.8% of the population, and can occur in a sporadic form characterized by isolated lesions and a familial form characterized by multiple lesions with an autosomal dominant mode of inheritance [11]. Most common symptoms include seizures, hemorrhage, and focal neurological dysfunction. High-resolution MR imaging is a useful diagnostic tool with a high degree of sensitivity and specificity. In general, the lesions are hyper- or hypointense or has a core of reticulated signal intensity surrounded by a hypointense rim on precontrast T2-weighted images [11,12]. In our study, the patient had intermittent headache and unilateral nasolabial sulcus was shallow. More over, the MR imaging finds were roughly consistent with those of angiocavernoma, therefore, angiocavernoma can be considered as one of the possible diagnoses of the disease.
Conclusion
In a word, the diagnosis of this patient is difficult in the absence of disease biopsy, nevertheless the above three diagnoses should be considered according to the patient’s clinical manifestations, physical examination, laboratory examination and neuroimaging finds.
Declarations
Funding: This work was supported by the Fundamental Research Funds for the Central Universities of Southeast University (2242020K40166).
Conflict of interest: No conflict exists: all authors declares that they have no conflict of interest.
Ethical approval: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
References
- Singh M, Manoranjan B, Mahendram S, McFarlane N, Venugopal C, Singh SK. Brain metastasis-initiating cells: survival of the fittest. Int J Mol Sci. 2014; 15(5): 9117-9133.
- Sjobakk TE, Vettukattil R, Gulati M, Gulati S, Lundgren S, Gribbestad IS, et al. Metabolic profiles of brain metastases. Int J Mol Sci. 2013; 14(1): 2104-2118.
- Rahmathulla G, Toms SA, Weil RJ. The molecular biology of brain metastasis. J Oncol. 2012; 2012: 723541.
- Soffietti R, Ruda R, Mutani R. Management of brain metastases. J Neurol. 2002; 249(10): 1357-1369.
- Nussbaum ES, Djalilian HR, Cho KH, Hall WA. Brain metastases. Histology, multiplicity, surgery, and survival. Cancer. 1996; 78(8): 1781-1788.
- Das A, Hochberg FH. Clinical presentation of intracranial metastases. Neurosurg Clin N Am. 1996; 7(3): 377-391.
- Barajas RF, Cha S. Metastasis in Adult Brain Tumors. Neuroimaging Clin N Am. 2016; 26(4): 601-620.
- Walker MT, Kapoor V. Neuroimaging of parenchymal brain metastases. Cancer Treat Res. 2007; 136: 31-51.
- Garcia HH, Nash TE, Del Brutto OH. Clinical symptoms, diagnosis, and treatment of neurocysticercosis. Lancet Neurol. 2014; 13(12): 1202-1215.
- Singh G, Burneo JG, Sander JW. From seizures to epilepsy and its substrates: neurocysticercosis. Epilepsia. 2013; 54(5): 783-792.
- Tokunaga K, Date I. Clinical features and management of cavernous and venous angiomas in the head. Brain Nerve. 2011; 63(1): 17-25.
- Toyoda K, Oba H. Diagnostic imaging of hemangiomas in the brain. Brain Nerve. 2011; 63(1): 5-15.