Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Series - Open Access, Volume 3
Fulminant Guillain-Barre syndrome associated with SARS-COV-2: A case series
Nasrin Forghani1, Zahra Jalali2, Hormoz Ayramlou3, Pooya Jalali4*
1Tabriz University of Medical Sciences, Tabriz, Iran.
2Beth Israel Deaconess Medical Center, Harvard Medical school, 330 Brookline Avenue, Boston, MA, 02215, USA.
3Professor of Neurology, Tabriz University of Medical Sciences, Tabriz, Iran.
4Islamic Azad University Tabriz Branch, Tabriz, Iran
*Corresponding Author: Pooya Jalali
Univesity Main St, Tabriz, East Azerbaijan Province, Iran.
Tel: +98-9187859587;
Email: pooya.jalali1995@gmail.com
Received : Nov 02, 2021 2021
Accepted : Jan 24, 2022
Published : Jan 31, 2022
Archived : www.jcimcr.org
Copyright : © Thierry YTR (2022).
Abstract
Pneumonia caused by unknown etiology occurred in December 2019 in Wuhan, Hubei Province, China, confirmed to be caused by a novel coronavirus. The Symptoms of SARS-Cov-2 depend on the patient’s age and underlying disease, as well as the state of the immune system. Guillain-Barré Syndrome (GBS) is an immune-mediated polyradiculoneuropathy inflammatory disease characterized by by flaccid paralysis. GBS cases associated with coronavirus (Cov) family infections have been described as Acute Respiratory Syndrome (SARS) Cov and the Middle East Respiratory Syndrome (MERS) COV. Recently, during the pandemic infection of the SARSCOV-2, some cases of immune-mediated neuropathies have been identified. In this Case Series study, we describe four patients who presented with fulminant GBS associated with SARS-COV-2.
COVID-19; Guillain-Barré syndrome; Fulminant Guillain Barre syndrome; SARS-CoV-2.
Citation: Forghani N, Jalali Z, Ayramlou H, Jalali P. Fulminant Guillain-Barre syndrome associated with SARS-CoV-2: A case series. J Clin Images Med Case Rep. 2022; 3(1): 1626.
Introduction
Pneumonia caused by unknown etiology occurred in December 2019 in Wuhan, Hubei Province, China, confirmed to be caused by a novel coronavirus [1-3]. The Symptoms of SARSCov-2 depend on the patient’s age and underlying disease, as well as the state of the immune system [4]. The neurological signs of the SARS-Cov-2 infection due to its impact on the Central Nervous System (CNS) (headache, dizziness, consciousness disorder, acute brain disease, seizures, and ataxia), [5] Peripheral Nervous System (PNS) (anosmia, visual impairment, Nerve pain) and skeletal muscles [6]. Mao et al. [7] Evaluated neurological symptoms in 214 patients infected with SARS-Cov-2. Of the 214 patients, 36.4% had signs of the nervous system including dizziness, headache, hypozoic, muscle injury, ischemic, and hemorrhage stroke.
Guillain-Barré Syndrome (GBS) is an immune-mediated polyradiculoneuropathy inflammatory disease characterized by flaccid paralysis [8]. In this disorder, the concentration of protein in the Cerebrospinal Fluid (CSF) increases, while the number of white blood cell count is normal [9]. Symptoms peak within 4 weeks and patients should be monitored because 20 to 30% of them require mechanical ventilation [10,11]. GBS cases associated with coronavirus (Cov) family infections have been described as Acute Respiratory Syndrome (SARS) Cov [12] and the Middle East Respiratory Syndrome (MERS) COV [13]. Recently, during the pandemic infection of the SARS-COV-2, some cases of immune-mediated neuropathies have been identified [14-18]. Severe inflammatory response and critically ill nature Numerous COVID-19 patients present a challenge in differentiating GBS from other neurological mimicking states, including critical polyneuropathy and myopathy [19]. In this study, we describe GBS symptoms in four infected patient with SARS-COV-2 in Imam Reza Hospital, Tabriz, Iran.
Case presentation
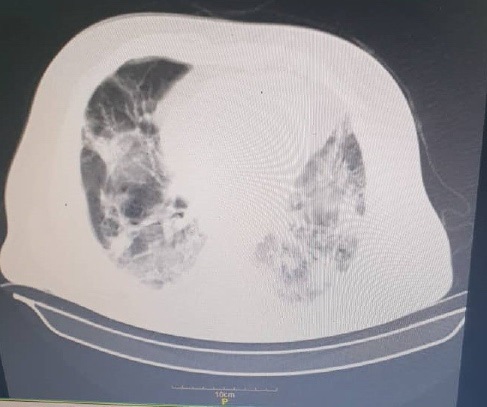
Case 1: A 44-year-old man developed dyspnea, loss of consciousness, and fever of up to 39°C lasting 4 days. He had a history of diabetes. A nasopharyngeal swab testing for SARS-CoV-2 with real-time polymerase chain reaction assay (RT-PCR) was positive. He was admitted to our unit to receive treatment for SARS-COV-2. Spiral Computed Tomography (CT) scan was shown in Figure 1. Hallucination was added to the patient’s symptoms after 3 days. Brain Magnetic Resonance Imaging (MRI) and Magnetic Resonance Venography (MRV) were taken from the patient and they did not have any pathological abnormalities. Lumbar Puncture (LP) was performed and did not have any abnormalities. After 9 days, he was discharged with stable vital signs. The patient developed lower extremity weakness 12 days after he was discharged. A neurological examination showed weakness in the dorsiflexion of the foot [Medical Research Council (MRC) score: 3/5 on the right and left side], weakness in the extension of hand and fingers (MRC score: 3/5 bilaterally), gait ataxia, loss of touch and vibration sensation in the feet, weak tendon reflexes in the upper and the lower limbs. Electromyography (EMG) and Nerve Conduction Velocity (NCV) was performed and Acute Motor Axonal Neuropathy (AMAN) was detected (Table1) and he was diagnosed with GBS and admitted to the Intensive Care Unit (ICU) of Imam Reza Hospital. One day after admission he recieved plasmapheresis. LP was performed and characteristic findings on CSF analysis was an elevation in CSF protein (90 g/L) without an elevation in white blood cells (WBC=0). Two days after admission, the patient underwent Endotracheal intubation because of respiratory distress, hypoxemia, and loss of consciousness. Ten hours after intubation the patient had Cardiac arrest and after an unsuccessful Cardiopulmonary Resuscitation (CPR) he was expired.
Table 1: Nerve conduction study parameters of case 1.
Nerve |
SNAP Onset |
SNAP Peak |
SCV (m/s) |
SNAP Amplitude |
Right Sural |
2.2 |
20 |
45 |
2.8 |
Left Sural |
2.2 |
22 |
NR |
2.7 |
Right Median |
2.1 |
35 |
51 |
2.8 |
Left Median |
2.3 |
32 |
49 |
2.8 |
Right Ulnar |
1.9 |
31 |
54 |
2.5 |
|
|
|
|
|
Nerve |
CMAP Distal |
CMAP Amplitude |
Velocity (m/s) |
F wave latency |
Right Tibial |
5.1 |
0.4 |
41 |
NR |
Left Tibial |
4.7 |
0.4 |
45 |
NR |
Right Peronal |
NR |
NR |
|
|
Left Peronal |
NR |
NR |
|
|
Right Ulnar |
3.4 |
0.1 |
44 |
|
Left Ulnar |
3.8 |
0.4 |
65 |
|
Right Median |
3.7 |
2.6 |
50 |
|
Left Median |
3.7 |
3.8 |
44 |
|
|
|
|
|
|
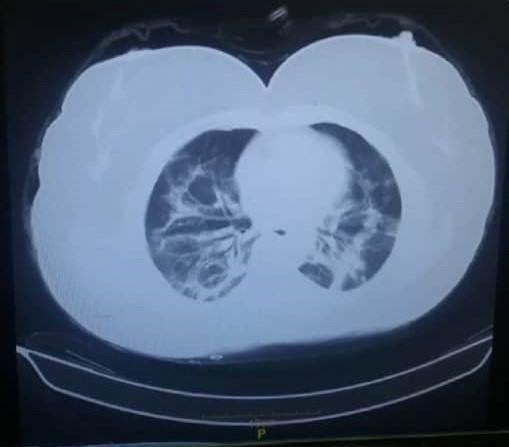
Case 2: A 22-year-old man with no history of an underlying disease six days after fever, headache, cough, and myalgia, four limbs became weakening, starting from the lower limbs and spreading to the upper limbs. On initial examination, There was symmetric limb weakness (Medical Research Council score 3/5 at upper limbs and 1/5 at lowerlimbs) without swallowing disorders and respiratory distress. EMG-NCV (Table 2) and Spiral chest CT scan were performed (Figure 2). According to the EMG-NCV, AMAN type was reported. According to the EMG-NCV, DTR's absence, history of fever and cough, and positive RT-PCR assays of the nasopharyngeal sample for SARS-CoV-2, he underwent IVIG treatment with a diagnosis of GBS and SARS-COV-2 (Figure 3). During hospitalization, after receiving IVIG, the weakness of the patient's limbs was recovered and after receiving IVIG, he was discharged with a Medical Research Council score of 4/5 at upper limbs and 3/5 at lower limps
Table 2: Nerve conduction study parameters of case 2.
Nerve |
SNAP Onset |
SNAP Peak |
SCV (m/s) |
SNAP Amplitude |
Left Sural |
2.5 |
2.5 |
0 |
NR |
Right Median |
9.7 |
0 |
0 |
10.2 |
Right Ulnar |
9.2 |
0 |
0 |
NR |
|
|
|
|
|
Nerve |
CMAP Distal |
CMAP Amplitude |
Velocity (m/s) |
F wave latency |
Right Tibial |
2.2 |
0 |
NR |
NR |
Right Peronal |
NR |
NR |
NR |
|
Left Peronal |
NR |
NR |
NR |
|
Right Ulnar |
7.7 |
0.9 |
56 |
45.1 |
Right Median |
15.7 |
0.7 |
46 |
|
Left Median |
NR |
NR |
NR |
|
|
|
|
|
|
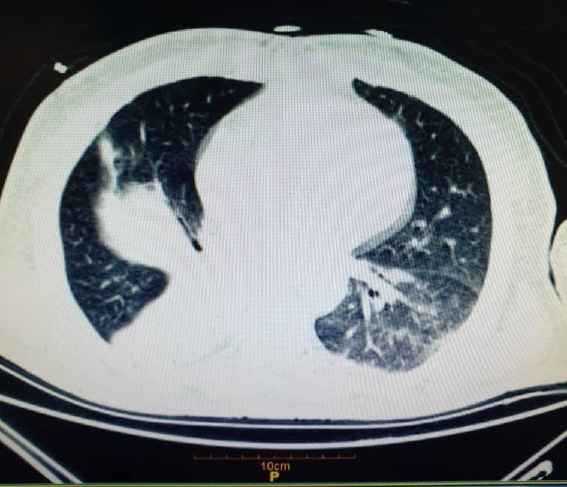
Case 3: A 51-year-old man with a history of Hypertension (HTN) and Coronary Artery Bypass Graft (CABG), after 7 days of fever, myalgia, and cough, first distal limbs dysesthesias, then there was symmetric limb weakness with distal to a proximal pattern. Spiral Chesht CT and RT-PCR assays of the nasopharyngeal sample were performed and there were posotive for SARSCov-2. He was admitted with a diagnosis of SARS-COV-2 (Figure 4). After 2 days, he developed swallowing disorders and respiratory distress. All 4 limbs had DTR absent and MF=1/5. Six hours later, he was admitted to the ICU and intubated due to respiratory distress exacerbation. With suspicion of GBS, EMG-NCV was performed and Acute Inflammatory Demyelinating Polyneuropathy (AIDP) was detected (Table 3). IVIG treatment was started with a total dose of 140 gr, and supportive measures were taken for the patient. Seven days later, the patient was extubated. After extubation, the patient's M/F was 2.5, which was 4.5 in all 4 Limps after physiotherapy and repeat IVIG therapy.
Table 3: Nerve conduction study parameters of case 3.
Nerve |
SNAP Onset |
SNAP Peak |
SCV (m/s) |
SNAP Amplitude |
Right Sural |
3.75 |
6.8 |
53.33 |
4.24 |
Left Sural |
4.4 |
6.22 |
45.45 |
5 |
Right Median |
2.85 |
31.35 |
52.63 |
3.47 |
Left Median |
3.2 |
40.45 |
46.88 |
3.76 |
Left Ulnar |
2.65 |
45 |
49.06 |
3.17 |
Right Ulnar |
2.5 |
15.90 |
52 |
3.31 |
|
|
|
|
|
Nerve |
CMAP Distal |
CMAP Amplitude |
Velocity (m/s) |
F wave latency |
Right Tibial |
10.8 |
1.16 |
37.04 |
42.25 |
Left Tibial |
16 |
0.89 |
32 |
43.05 |
Right Peronal |
17.6 |
0.07 |
37.7 |
|
Left Peronal |
7.5 |
0 |
NR |
29.2 |
Right Ulnar |
3.65 |
2.93 |
NR |
65.4 |
Left Ulnar |
2.81 |
2.11 |
NR |
57.75 |
Right Median |
4 |
1.98 |
54.7 |
57.4 |
Left Median |
4.83 |
2.66 |
54 |
57.85 |
|
|
|
|
|
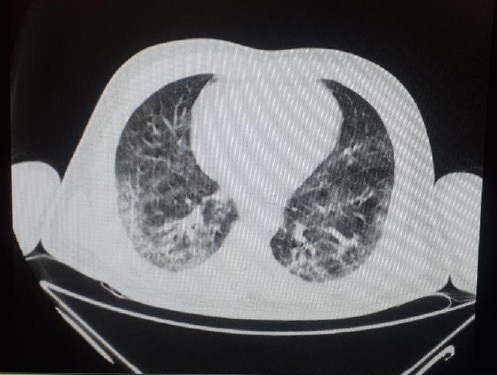
Case 4: A 71-year-old man with a history of hypertension was referred to our center with an initial complaint of limb dysesthesia. He had symptoms of fever and myalgia for 5 days. At the beginning of the visit, due to suspicion of SARS-COV-2, a spiral chest CT scan was performed from the patient (Figure 4). RTPCR assays of the nasopharyngeal sample for SARS-CoV-2 were positive. On neurological examination, DTR was absent and the Medical Research Council score of all 4 limbs was 3/5. EMG-NCV was performed due to suspicion of GBS and AIDP was detected (Table 4). Finally, he was admitted to our center with a diagnosis of GBS and SARS-COV-2. During hospitalization on the third day, he developed severe weakness and swallowing disorders and respiratory distress, which he intubated and he was expired on the 4th day due to an Anterolateral massive Myocardial infarction.
Table 4: Nerve conduction study parameters of case 4.
Nerve |
SNAP Onset |
SNAP Peak |
SCV (m/s) |
SNAP Amplitude |
Right Sural |
3.6 |
4.4 |
38.89 |
6.24 |
Left Sural |
3.5 |
4.5 |
36.01 |
5.21 |
Right Median |
3.83 |
5.5 |
36.6 |
4.48 |
Left Median |
3.28 |
4.8 |
42.75 |
10.19 |
Left Ulnar |
3.5 |
4.53 |
42.1 |
5.34 |
Right Ulnar |
3.4 |
4.65 |
42.28 |
5.21 |
|
|
|
|
|
Nerve |
CMAP Distal |
CMAP Amplitude |
Velocity (m/s) |
F wave latency |
Right Tibial |
NR |
NR |
NR |
NR |
Left Tibial |
12.43 |
0.1 |
31 |
NR |
Right Peronal |
NR |
NR |
NR |
NR |
Left Peronal |
NR |
NR |
NR |
NR |
Right Ulnar |
7.14 |
4.2 |
41 |
35 |
Left Ulnar |
6.18 |
3.1 |
37 |
36 |
Right Median |
8.93 |
2.91 |
41 |
38 |
Left Median |
7.13 |
1.64 |
40 |
36 |
|
|
|
|
|
Discussion
In this Case Series study, we describe four patients who presented with fulminant GBS associated with SARS-COV-2. SARS-COV-2 epidemic indicates all efforts for the early identification and treatment of its manifestations. Similar to other viruses belonging or not to the coronavirus family, neurological complications are emerging in SARS-COV-2 as one of the most significant clinical components of this epidemic [20,21]. The Clinical features and electrophysiological findings, along with the epidemiological background and IgG presence to SARSCOV-2, support the diagnosis of post-SARS-COV-2 GBS [22]. IVIG and PE are two treatment options for moderate to severe GBS [23]. In our cases, we started treatment with IVIG therapy right upon admission, and it was repeated in one patient due to weakness at the lower limbs. Among the CBC parameters confirmed by SARS-COV-2 cases, normal or increased monocytes and decreased lymphocytes have been pointed out [22]. In our cases, lymphopenia and increased LDH was found in laboratory results. One of the most common neurological symptoms in patients with GBS is acute muscle weakness [24]. The pattern of muscle weakness may help to diagnose GBS [25]. Acute flaccid quadriparesis and weakness in the limbs were seen in most GBS reports. Besides, demyelinating polyneuropathy is commonly seen in most of these reports. Some of the SARS-COV-2-related GBS patients had the axonal variants of GBS [25]. In our study, AIDP and AMAN were detected in EMG-NCV. Coronaviruses are believed to cause GBS in certain patients directly through the capacity for neuroinvasion (ACE2 receptors on neuronal tissues) or indirectly through the immune system response (an inflammatory mechanism) [26,27]. The data show that SARSCoV-2 can induce an immune response by increasing levels of interleukin-6 (IL-6), which stimulates the inflammatory cascade and damages tissues. Therefore, inflammatory factors can play a significant role in limb function in patients' organ dysfunctions with SARS-COV-2 infection [28-30]. The mechanism of GBS formation in patients infected with SARS-COV-2 has not yet been investigated. SARS-COV-2 stimulates inflammatory cells and produces various inflammatory cytokines, resulting in immunemediated processes [31]. GBS is an immune-mediated disorder, and molecular mimicry as an autoimmune disorder mechanism plays an essential role in its development [31]. It is unknown whether SARS-COV-2 causes antibodies against certain gangliosides that commonly appear in certain forms of GBS [32].
References
- Zhu N , Zhang D , Wang W , Li X , Yang B , Song J , et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020; 382: 727–733.
- Huang C , Wang Y , Li X , Ren L , Zhao J , Hu Y , et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan. China. Lancet. 2020; 395: 497–506.
- Chen N , Zhou M , Dong X , Qu J , Gong F , Han Y , et al. Epidemiological and clini- cal characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet. 2020; 395: 507–513.
- Wang W, Tang J, Wei F. Updated understanding of the outbreak of 2019 novel coronavirus (2019-nCoV) in Wuhan, China. J Med Virol. 2020; 92: 441–447.
- Wang D, Hu B, Hu C, Zhu F, Liu X, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. Jama. 2020; 323:1061–1069.
- Mao L, Jin H, Wang M, Hu Y, Chen S, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA neurology. 2020.
- Mao L, Wang M, Chen Sh, He Q, Chang J, Hong C, et al. Neurological Manifestations of Hospitalized Patients with SARS-COV-2 in Wuhan, China: A Retrospective Case Series Study. 2020.
- Hu J, Luo X, Wang Y, Prado E, Fu Q, Shao A. Fulminant GuillainBarré Syndrome and Spontaneous Intraventricular Hemorrhage: A Case Report and Literature Review. Frontiers in neuroscience. 14, 633.
- Guillain G, Barré JA, Strohl A. Radiculoneuritis syndrome with hyperalbuminosis of cerebrospinal fluid without cellular reaction. Notes on clinical features and graphs of tendon reflexes. 1916. Annales de medecine interne. 1999; 150: 24–32.
- Sejvar JJ, Baughman AL, Wise M, Morgan OW. Population incidence of Guillain-Barré syndrome: A systematic review and meta-analysis. Neuroepidemiology. 2011; 36:123–133.
- Orlikowski D, Prigent H, Sharshar T, Lofaso F, Raphael JC. Respiratory dysfunction in Guillain-Barré syndrome. Neurocrit Care. 2004; 1: 415–422.
- Tsaim LK. Neurological manifestations in severe acute respiratory syndrome. Acta Neurol. Taiwanica. 2005; 14: 113–119.
- Kim JE. Neurological complications during treatment of Middle East respiratory syndrome. J. Clin. Neurol. (Seoul, Korea) 2017; 13: 227–233.
- Gutiérrez-Ortiz Consuelo. Miller Fisher Syndrome and polyneuritis cranial in COVID 19. Neurology. 2020.
- Toscano G. Guillain Barre Syndrome Associated with SARS cov2. N. Engl. J. Med. 2020; 382: 2574–2576.
- Sedaghat Z. Guillain Barre Syndrome Associated with covid19 infection: A case report. Clin. Neurosci. 2020; 15 (S0967– 5868(20)30882–1.
- Zhao H. Guillain Barre Syndrome Associated with SARS cov2 infection: Causality or coincidence? Lancet Neurol. 2020; 19: 383–384.
- Assini A. New clinical manifestation of SARS-COV-2 related Guillain-Barrè Syndrome highly responsive to intravenous immunoglobulins: two Italian cases. Neurol. Sci. 2020; 41: 1657–1658
- Bueso T, Montalvan V, Lee J, Gomez J, Ball S, et al. Guillain-Barre Syndrome and COVID-19: A case report. Clin Neurol Neurosurg. 2021; 200: 106413.
- Kim JE, Heo JH, Kim HO, et al. Neurological complications during treatment of Middle East Respiratory Syndrome. J Clin Neurol. 2010; 13: 227–233.
- Desforges M, Le Coupanec A, Brison E, et al. Neuroinvasive and neurotropic human respiratory coronaviruses: Potential neurovirulent agents in humans. Adv Exp Med Biol. 2014; 807: 75–96.
- Jin Y-H, Cai L, Cheng Z-S, Cheng H, Deng T, Fan Y-P, et al. A rapid advice guideline for the diagnosis and treatment of 2019 novel coronavirus (2019-nCoV) infected pneumonia (standard version) Military Med Res. 2020; 7:4.
- Pfefferkorn T, Dabitz R, von Wernitz-Keibel T, Aufenanger J, Nowak-Machen M, et al. Acute polyradiculoneuritis with lockedin syndrome in a patient with Covid-19. Journal of neurology. 2020; 267: 1883–1884.
- Zito A, Alfonsi E, Franciotta D, Todisco M, Gastaldi M, et al. SARSCOV-2 and Guillain-Barré Syndrome: A Case Report and Review of Literature. Frontiers in neurology. 2020; 11: 909.
- Rahimi K. Guillain-Barre syndrome during SARS-COV-2 pandemic: An overview of the reports. Neurological sciences: Official journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology. 2020; 41: 3149–3156.
- Zhou Z, Kang H, Li S, Zhao X. Understanding the neurotropic characteristics of SARS-CoV-2: from neurological manifestations of SARS-COV-2 to potential neurotropic mechanisms. J Neurol. 2020; 1–6.
- Zhao H, Shen D, Zhou H, Liu J, Chen S. Guillain-Barré syndrome associated with SARS-CoV-2 infection: Causality or coincidence? The Lancet Neurology. 2020; 19: 383–384.
- Carod-Artal FJ. Neurological complications of coronavirus and SARS-COV-2. Rev Neurol. 2020; 70: 311–322.
- Helms J, Kremer S, Merdji H, Clere-Jehl R, Schenck M, et al. Neurologic features in severe SARS-CoV-2 infection. N Engl J Med. 2020; 382: 2268–2270.
- Sedaghat Z, Karimi N. Guillain Barre syndrome associated with SARS-COV-2 infection: A case report. Journal of clinical neuroscience : Official journal of the Neurosurgical Society of Australasia. 2020; 76: 233–235.
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020; 395: 497–506.
- Jacobs BC, Rothbarth PH, van der Meché FG, Herbrink P, Schmitz PI, et al. The spectrum of antecedent infections in Guillain-Barré syndrome: A case-control study. Neurology. 1998; 51: 1110– 1115.