Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
A rare case of gastric outlet obstruction in a patient with multiple myeloma
Haeuptle R1; Driessen C2; Borovicka J1; Neuweiler J3; Brand S1; Sawatzki M1
1 Department of Gastroenterology, Kantonsspital St.Gallen, Switzerland.
2 Department of Medical Oncology and Hematology, Kantonsspital St.Gallen, Switzerland
3 Institute of Pathology, Kantonsspital St.Gallen, Switzerland
*Corresponding Author: Sawatzki M
Division of Gastroenterology, Kantonsspital St.Gallen,
Switzerland.
Email: mikael.sawatzki@kssg.ch
Received : Dec 21, 2021
Accepted : Jan 25, 2022
Published : Feb 01, 2022
Archived : www.jcimcr.org
Copyright : © Sawatzki M (2022).
Abstract
Gastric amyloidosis is a very rare cause of gastric outlet obstruction. Therefore, general treatment recommendations are missing. A 61-year-old man with multiple myeloma and cast-nephropathy with suspected manifestation of amyloidosis was admitted for gastroscopy because of nausea and emesis. Video fluoroscopy demonstrated a gastric outlet obstruction with subtotal stenosis of the pylorus. Gastric amyloidosis (AL-amyloid) was confirmed histologically by deroofingbiopsies which was consistent with the clonotype of the patient’s myeloma. Botox injection was ineffective for control of symptoms. Therefore, endoscopic balloon-dilatation (15 mm to 20 mm) was performed with a long-lasting clinical response of 3 to 6 months.
Citation: Haeuptle R, Driessen C, Borovicka J, Neuweiler J, Brand S, Sawatzki M. A rare case of gastric outlet obstruction in a patient with multiple myeloma. J Clin Images Med Case Rep. 2022; 3(2): 1629.
Introduction
Gastric amyloidosis is a very rare cause of gastric outlet obstruction. In a retrospective analysis, including 769 patients suffering from systemic amyloidosis, only 8 patients (1%) had a histologically proven gastric amyloidosis. Among these 8 patients, only 1 (0.13%) had gastric outlet obstruction [1]. Therefore, there is limited evidence for potential treatment options in these patients. Performing a PubMed search for gastric outlet obstruction and amyloidosis, we found 4 case reports on the therapy of gastric outlet obstruction in patients with gastric amyloidosis. The first case was a 64-year-old man with multiple myeloma, who showed a good clinical outcome after treating the underlying disease with chemotherapy [2]. The second case report described a 52-year-old man with monoclonal gammopathy of unknown significance (MGUS)/multiple myeloma with a partial clinical response to chemotherapy and prokinetic agents [3]. In a third case, a 80-year-old woman with systemic amyloidosis most likely due to multiple myeloma refused further therapy and died due to disease progression 34 days later [4]. The last case was on a 56-year-old man, who was suspected to suffer from diffuse gastric cancer (linitis plastica) and therefore underwent total gastrectomy. Only after histological analysis of the surgical specimen, the diagnosis of gastric amyloidosis was made [5].
Case report
A 61-year-old man suffering from nausea and emesis for 2 months, particularly after copious meals, was admitted for gastroscopy to the outpatient center of our hospital. A previously prescribed proton pump inhibitor (pantoprazole 40 mg once daily) was ineffective for symptom relief. As underlying disease, the patient was suffering from multiple myeloma ISS Stadium II with expression of light chain lambda which had been diagnosed 3 years ago. As complication of the multiple myeloma, the patient received haemodialysis three times a week due to a cast-nephropathy and was suspected of having systemic manifestation of amyloidosis, which so far could not have been proven. Cardiac MRI did not show any signs of amyloid deposition, although this was suspected in cardiac ultrasound. However, biopsies taken during a diagnostic laparoscopy were negative for amyloid. For therapy of the multiple myeloma, the patient had received 4 cycles of chemotherapy with VCD (bortezomib, cyclophosphamide, dexamethasone) with partial remission (PR), following high dose chemotherapy with melphalan and autologous stem cell transplantation with PR. Because of a disease relapse with rising light chain lambda after half a year, a third chemotherapy with velcade/dexamethasone (for 12 months) and endoxan (for 6 months) was given. This resulted in PR. At the time of the gastroscopy (after 3 different therapies including high dose chemotherapy with stem cell transplant), the patient had no ongoing therapy for his myeloma.
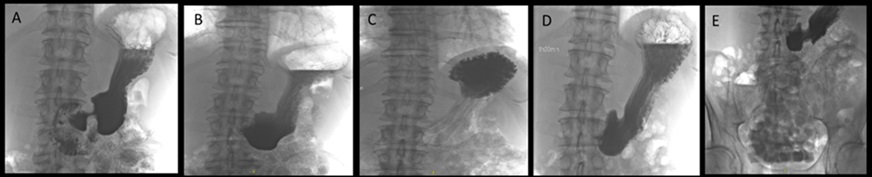
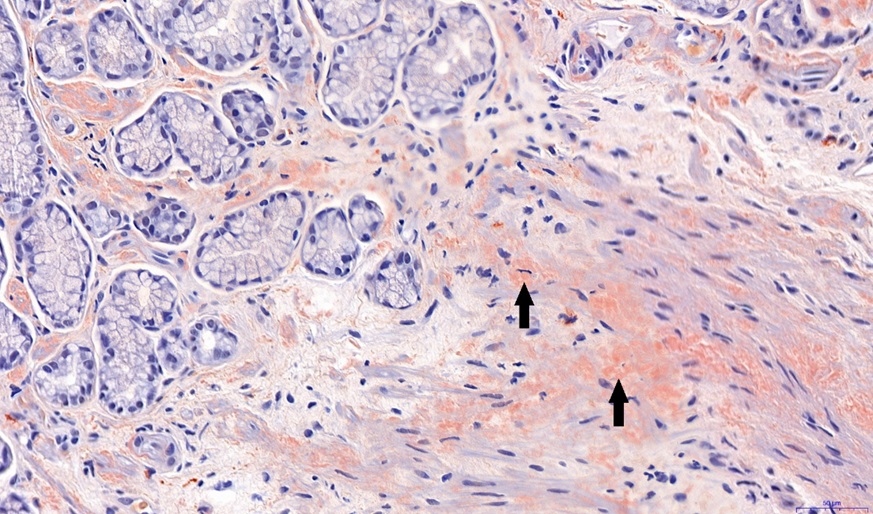
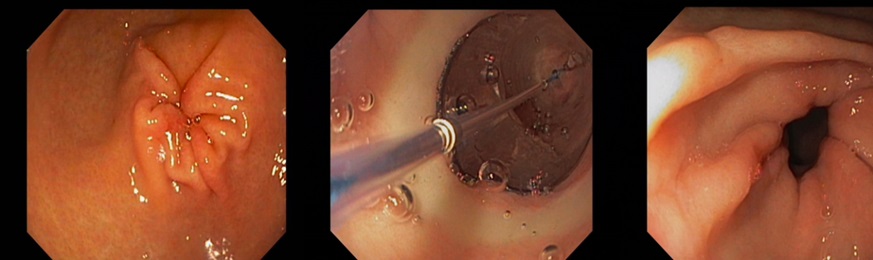
An initial gastroscopy was performed; this showed macroscopically no correlation to the nausea and emesis. Furthermore, there was no evidence of amyloidosis in the duodenal and gastric biopsies (no Congo red-positive areas found in the histological analysis). On an onsite test for lactase deficiency (Lactose intolerance quick test by BIOHIT Healthcare, Helsinki, Finland) was negative. Despite strict lactose-free diet and a trial with a prokinetic agent (domperidone), the symptoms persisted. Therefore, further diagnostic work-up with a video fluoroscopy was conducted. Here, a subtotal stenosis of the pylorus was highly suspected due to probably transient spasms of the pylorus (Figure 1). Thus, the symptoms of the patient were now explained by a gastric outlet obstruction probably caused by gastric amyloidosis. A second gastroscopy was performed. Considering the new diagnostic information of the video fluoroscopy, the pylorus appeared narrowed and showed muscular spams but endoscopic passage was possible with a standard gastroscope (diameter 10.8 mm) without difficulties. After deroofing of the gastric mucosa, submucosal biopsies were taken from the prepyloric antrum and botulinum toxin (Botox®) injections were performed in the 4 quadrants around the pylorus using a total amount of 10 ml Botox® to treat the transient pyloric spasms. Gastric amyloidosis could be confirmed histologically revealing Congo red depositions (Figure 2). The amyloid was classified as AL-amyloid of the kappa/lambda light chain, consistent with the clonotype of the patient’s myeloma. However, the patient did not benefit from the botulinum toxin injections. Suggesting a structurally fixed rather transient spastic stenosis, a third gastroscopy with balloon-dilatation of the pylorus (with dilatation from 15 mm to 20 mm) was performed. Although there were no visible mucosal tears even after dilatation to 20 mm, the pylorus was now visibly open (Figure 3). This was consistent with a good and persistent clinical response of the patient. The patient’s clinical response lasted from 3 to 6 months. In this patient, balloon-dilatation was repeated 4 times without adverse events (notably, there were no bleeding events despite a potentially higher bleeding risk due to amyloidosis).
However, due to progression of the myeloma (with rising serum levels of light chain lambda), chemotherapy with velcade/dexamethasone had to be started again. Under treatment with velcade/dexamethasone, the patient developed polyneuropathy and therefore chemotherapy had to be switched to an immunotherapy with daratumumab. Unfortunately, 15 months after the first ballon-dilatation, the patient died of pneumonia.
Discussion
In this patient, the histological diagnosis of amyloidosis was established by submucosal biopsies (“deroofing biopsies”) only because the amyloid deposits are usually found in the subepithelial layers. After failure of symptomatic relief by prokinetic agents and botulinum toxin injections and given the lack of alternative therapies in the current literature [1-4], we decided to perform endoscopic dilatation therapy in this patient, considering the structural changes in the pyloric region due to chronic amyloid deposition. With only one session of balloon dilatation of the pylorus, a long-lasting clinical response with total regression of the emesis and nausea could be achieved. The potential value of endoscopic balloon dilatation has not yet been described in the literature. Our case illustrates that endoscopic balloon dilatation appears to be a valid treatment option in patients with gastric outlet obstruction due to amyloidosis.
References
- Menke DM, Kyle RA, Fleming CR et al. Symptomatic gastric amyloidosis in patients with primary systemic amyloidosis. Mayo Clinic Proc. 1993; 68: 763-767.
- Cohen JA, An J. Brown AW et al. Gastric outlet obstruction due to gastrointestinal amyloidosis. J Gastrointest Surg. 2017; 21: 600- 601.
- Lee ASY, Lee DZQ, Vasanwala FF. Amyloid light-chain amyloidosis presenting as abdominal bloating: a case report. J Med Case Rep. 2016; 30: 10:68
- Park SW, Lee HW, Cho EJ. Systemic amyloidosis manifested by gastric outlet obstruction. Clin Endosc. 2013; 46: 579-581.
- Bedioui H, Chebbi F, Ayadi S et al. Gastric amyloidosis mimicking malignancy. A case report. Ann Chir. 2006; 131: 455-458.