Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Research Article - Open Access, Volume 3
Cryptococcus pneumonia: Helpful findings for diagnosing cryptococcal pneumonia on CT in residents
Kai Ke Li, MD1; Gong Yong Jin MD, PhD1*; Keun Sang Kweon, MD2
1 Department of Radiology, Research Institute of Clinical Medicine of Jeonbuk National University-Biomedical Research Institute of Jeonbuk National University Hospital, Jeonbuk National University and Medical School, Jeonju, South Korea.
2 Province, Research Institute of Clinical Medicine of Jeonbuk National University-Biomedical Research Institute of Jeonbuk National University Hospital, Jeonbuk National University and Medical School, Jeonju, South Korea.
*Corresponding Author: Gong Yong Jin
Department of Radiology, Research Institute of Clinical
Medicine of Jeonbuk National University-Biomedical
Research Institute of Jeonbuk National University
Hospital, Jeonbuk National University and Medical
School, Jeonju, Korea.
Email: gyjin@jbnu.ac.kr
Received : Jan 07, 2022
Accepted : Jan 25, 2022
Published : Feb 01, 2022
Archived : www.jcimcr.org
Copyright : © Jin GY (2022).
Abstract
Background: Pulmonary cryptococcosis has various CT findings which can be often misdiagnosed as other diseases to delay patient’s treatment. It is actually very difficult to diagnose pulmonary cryptococcal pneumonia on chest CT, especially for inexperienced residents.
Purpose: To investigate whether there are new CT findings that may be helpful to residents when diagnosing cryptococcal pneumonia with CT.
Materials and methods: We retrospectively reviewed chest CT findings of 23 patients with pathologically proven PC from 2007 to 2018 in our hospital. Eight residents independently read chest CT twice at regular intervals, and at the second reading, the patient’s clinical information and one additional CT finding were provided. Correlation between CT findings and diagnosis accuracy for cryptococcal pneumonia were compared by simple liner regression.
Results: Most common pulmonary lesion identified on CT was multiple nodules (n = 15, 65.2%), followed by masses (n=8, 34.8%). The distribution of located within 2 cm below pleural and distributed along the bronchi, which 19 (82.6%) patients were observed in their CT images, and enhancement of nodule or mass were significant correlated for CT findings and diagnostic accuracy on cryptococcal pneumonia, which p-value were 0.018 and 0.007 respectively.
Conclusion: In order to accurately diagnose pulmonary cryptococcal pneumonia, clinical information including the patient’s immune status and nodules clustered within 2 cm of the bronchi on the chest CT verified in this study are helpful to trainers
Keywords: pulmonary; infection; cryptococcosis; radiographic features; diagnosis.
Abbreviations: PC: Pulmonary cryptococcosis; CT: Computed Tomography; TB: tuberculosis; PTNB: percutaneous transthoracic needle biopsy; HU: Hounsfield units.
Citation: Li KK, Jin GY, Kweon KS. Cryptococcus pneumonia: Helpful findings for diagnosing cryptococcal pneumonia on CT in residents. J Clin Images Med Case Rep. 2022; 3(2): 1630.
Introduction
Cryptococcus neoformans is a ubiquitous encapsulated yeast-like fungus that is found worldwide; particularly in soils that are contaminated with pigeon excreta and decayed wood [1-3]. Inhalation of cryptococcal particles into the lungs is a route of pulmonary infection, and subsequent hematogenous dissemination may cause central nervous system infection. Pulmonary cryptococcosis (PC) in the immunocompetent host is rare and may be asymptomatic [2,4]. The most common radiographic manifestations of pulmonary cryptococcosis consist of single or multiple pulmonary nodules, segmental or lobar consolidation, or a reticulonodular pattern of opacities [5-8]. However, these findings often appear to very depending on when patient is infected and are often non-specific. It previously reported that patient once experienced unnecessary antibiotics, even anti-TB treatment. Pretty high rate for initial misdiagnosis on immunocompetent patients with PC was commonly reported in domestic and oversea studies. In absence of explicit etiology evidences, various symptoms and confused radiographic manifestations by PC always lead to mazy options in differential diagnosis and make reaching definite diagnosis like a tricky gamble in real clinical settings [9]
PC has various CT findings according to the patient’s immune status, the time of CT examination, and the degree of cryptococcus inhalation at diagnosis. Because of this, although there are typical CT findings of PC, typical CT findings itself are often difficult to differentiate from other diseases such as lung cancer or pulmonary tuberculosis. [10] In particular, for trainers without knowledge about this disease, it is actually very difficult to diagnose pulmonary cryptococcal pneumonia on chest CT. Recently, Wang et al [11] reported that the major CT manifestations were a cluster of nodules or masses located within 2 cm below the pleura and distributed along the bronchi. Comparison of CT finding with the pathological manifestation of pulmonary cryptococcosis with multiple nodules or masses indicated that the pathological results may explain imaging findings. This finding may provide a deeper understanding of this disease and improve the accuracy of its diagnosis. Then, we investigated whether this CT finding help diagnose cryptococcal pneumonia by reviving CT of patients diagnosed with cryptococcal pneumonia. We tried to find out what clues are helpful for inexperienced trainers to diagnose cryptococcal pneumonia.
Material and methods
This is a retrospective study for patients diagnosed with PC and treated with medication therapy from April 2007 to February 2018 in our hospital. Our Institutional Review Board approved our retrospective study with a waiver of the informed consent from each patient. 23 patients were identified who had pathologically proven PC. The specimens were obtained via a percutaneous transthoracic needle biopsy (PTNB) in 12 patients and surgery in 11 patients. Medical records of 23 patients were reviewed for demographic data, consisting of underlying conditions, clinical features, radiological studies, pathology results, treatment and follow-up information. The population of patient consisted of 10 men and 13 women, with an age range from 49 years to 82 years (median, 63 years; mean age ± standard deviation [SD]:63 ± 10.67 years).
CT examination and image analysis
All subjects underwent volumetric assessment using a 16 or 128 multidetector CT scanner (Somatom Sensation 16, Siemens Medical Solutions, Erlangen, Germany; Somatom Definition AS, Siemens Medical Solutions, Forchheim, Germany, respectively) with contrast in the craniocaudal direction while they were placed in the supine position; scans were taken at full inspiration. Volumetric CT scans were obtained both at full inspiration (200 MAs) using 120 kvp tube current, 0.5 or 0.33 second rotation time, submillimeter reconstruction (thickness: 0.75 mm, interval: 0.5 mm), slice thickness (2 mm), and 16 X 0.75 or 128 X 0.6 mm acquisition. Images were displayed in the lung window to assess the lung parenchyma (window level, -600 to -700 HU; window width, 1200–1500 HU).
The CT image analysis of patients with cryptococcal pneumonia was performed by a total of eight residents (2 residents each from 1st to 4th year), including residents without CT reading experience for cryptococcal pneumonia. They independently evaluated the CT images and were blinded to the clinical information for nodules, masses, ground-glass opacity (GGO), consolidation, number (single or multiple) and distribution (central, peripheral or random) of lesions, cavitation and air bronchograms, halo sign, reticular opacity, bronchial wall thickening, lymphadenopathy and pleural effusion. Nodules and masses were defined as rounded opacities that were at least moderately well marginated. Nodules were ≤3 cm in maximum diameter and masses were >3 cm. GGO was identified as a lesion with the hazy increased attenuation of the lung, but with preservation of bronchial and vascular margins. If a rim of GGO or hazy increased attenuation surrounded a nodule, it was said to have a halo sign. If the short-axis diameter of a lymph node was >10 mm, it was considered to be an enlargement. The treein-bud pattern was defined as centrilobular branching structures that resemble a budding tree. The trans-axial distribution of the abnormalities was also identified. Peripheral distribution was considered to be the abnormalities that were mostly located in the outer third of the lung, and central distribution was considered to be the abnormalities located in the inner third of the lung.
The first chest CT was to analyze chest CT findings without clinical information (age, sex, immune status, underlying disease, etc.) on cryptococcal infection. The second CT reading was performed 3 months after the first CT reading, and the CT findings were analyzed after providing clinical information (age, sex, immune status, underlying disease, etc.) on cryptococcal infection. In addition, when the second chest CT was analyzed, a cluster of nodules or masses located within 2 cm below the pleura and distributed along the bronchi, which could be an CT finding of cryptococcal infection, was added. To review chest CT findings of cryptococcal pneumonia, a checking sheet was made with known CT findings and presented to trainers (Supplementary Table 1 and Table 2).
Statistical analysis
The data are collected and calculated each average value, Mann-Whitney U Test are employed to compare each factor between first and second CT reading, to find which factors has significant difference, means these factors will affect clinical diagnosis of PC. By checking the statistics data, all p value of CT finding factors >0.05, which means no factor has significant difference between first and second CT reading. Simple linear regression is employed to calculate correlation between CT factors and diagnosis accuracy for cryptococcal pneumonia on chest CT.
Results
Demographic data for patients of cryptococcal pneumonia was in Table 1. Of the 23 patients, 18 (78%) had no symptoms or signs. In 5(22%) patients with symptoms, symptoms including cough (n=1), chest pain (n=3), and fever(n=1) were observed. 3(13%) patients were smoker and 18(87%) was no-smoker. 6(26%) patients were in immunocompetent status and 17(74%) patients were in immunocompromised status; diabetes mellitus (n=10), cervical cancer (n=2), colon cancer (n=1), gastric cancer (n=1), hormone therapy (n=1) and steroid therapy (n=2). 18 (87%) patients were incidentally detected on chest radiographs.
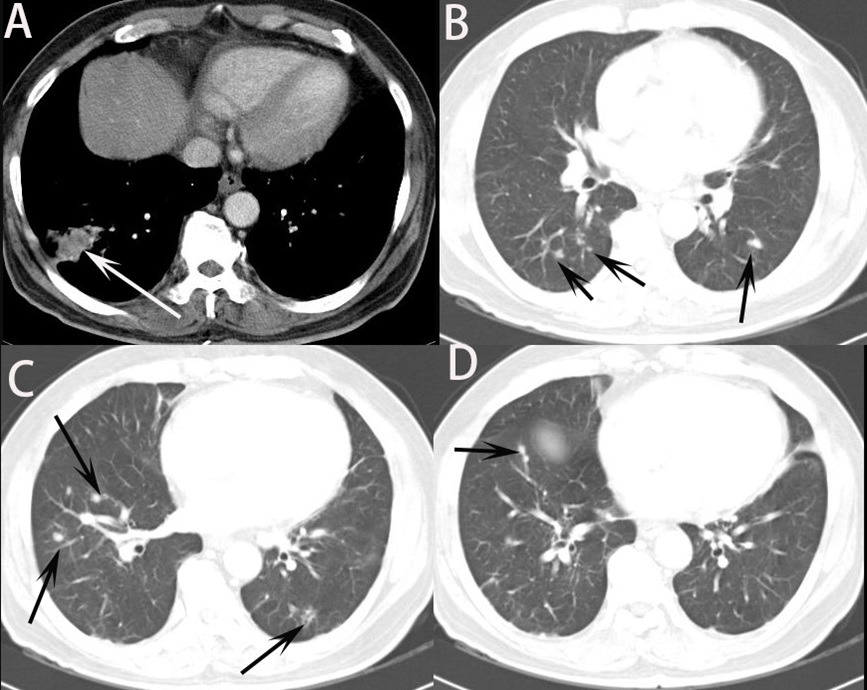
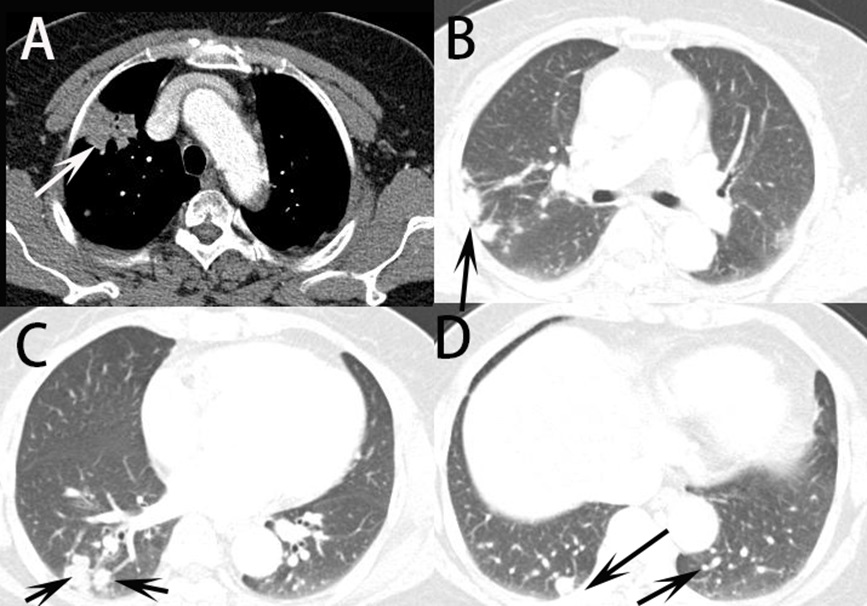
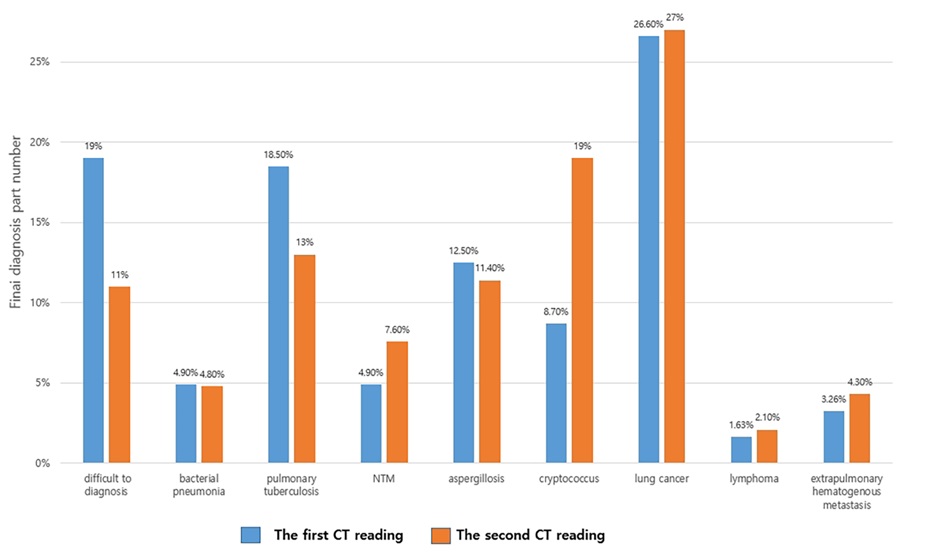
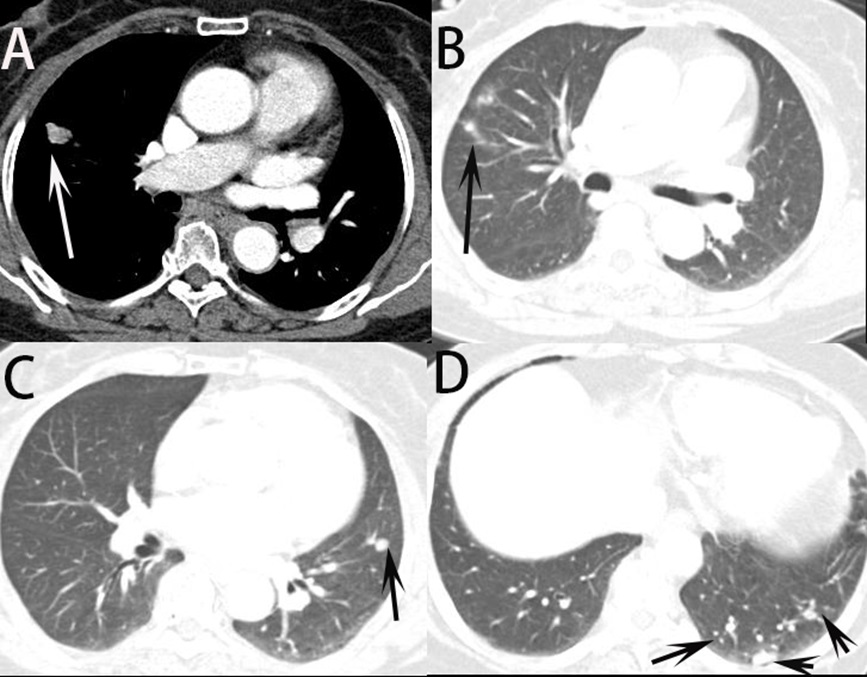
The distribution pattern of nodules or masses on chest CT were collected in Table 2. About the number distribution, the single nodule or mass on chest CT was observed in 4 (17.4%) patients and multiple nodules or mass in 19 (82.6%) patients (Figure 1). About the distribution of located within 2 cm below the pleural and distributed along the bronchi, 19 (82.6%) patients were observed in their CT images (Figure 2). Therefore, it can be predicted that these findings can be very important findings for diagnosing PC when multiple pulmonary nodules are seen on chest CT. Comparison of CT scan finding between first and second CT reading in all trainers were collected in Table 3. By using statistical analysis between first reading and second reading, all p value of CT finding factors >0.05, which means no factor has significant difference between first and second reading in trainer. In other words, it was found that the CT findings of cryptococcal pneumonia could be accurately obtained by trainers by having trainers are asked to check the CT findings in the checking sheet presented in our study. It was found, also, that the CT findings of cryptococcal pneumonia can be described relatively consistently regardless of the presence or absence of experience or knowledge about cryptococcal pneumonia although the experience of cryptococcal pneumonia differs depending on the trainers. Simple regression results for CT finding and second diagnostic accuracy for cryptococcal pneumonia was in Table 4. The distribution of located within 2 cm below pleural and distributed along the bronchi (p=0.018) and enhancement of nodule or mass (p=0.007) were significant in statics (Figure 3). When the trainer was given additional clinical information and additional CT finding information, the diagnosis rate of cryptococcal pneumonia was significantly increased compared to other diseases (Figure 4).
Table 1: Demographic and clinical information of 23 patients who diagnosed Cryptococcal Pneumonia.
Patients |
Age/Sex |
Presenting |
Immune Status/Underlying disease |
Smoking |
Incidental |
1 |
50/F |
No |
Compromised/DM |
No |
Yes |
2 |
61/M |
Chest pain |
Competent/No |
No |
No |
3 |
50/F |
No |
Compromised/cervical cancer |
No |
Yes |
4 |
50/M |
No |
Compromised/DM |
No |
Yes |
5 |
55/F |
No |
Compromised/DM |
No |
Yes |
6 |
55/F |
Chest pain |
Competent/No |
No |
No |
7 |
50/F |
No |
Compromised/cervical cancer |
No |
Yes |
8 |
63/M |
No |
Compromised/DM |
No |
Yes |
9 |
57/F |
No |
Compromised/DM |
No |
Yes |
10 |
66/F |
Cough |
Compromised/hormone Therapy |
No |
No |
11 |
62/M |
No |
Compromised/DM |
No |
Yes |
12 |
78/M |
No |
Competent/pneumoconiosis |
Yes |
Yes |
13 |
73/M |
No |
Compromised/gastric cancer with rectal cancer |
Yes |
Yes |
14 |
76/F |
Chest pain |
Compromised/DM |
No |
No |
15 |
82/M |
No |
Compromised/steroid therapy |
No |
Yes |
16 |
49/M |
No |
Compromised/DM |
No |
Yes |
17 |
76/M |
No |
Competent/No |
Yes |
Yes |
18 |
75/F |
No |
Compromised/ steroid therapy |
No |
Yes |
19 |
50/M |
Fever Cough |
Compromised/colon cancer |
No |
No |
20 |
65/F |
No |
Competent/No |
No |
Yes |
21 |
71/F |
No |
Compromised/DM |
No |
Yes |
22 |
69/F |
No |
Compromised/DM |
No |
Yes |
23 |
67/F |
No |
Competent/No |
No |
Yes |
Table 2: Summary of distribution pattern with pulmonary cryptococcosis on chest CT (N=23).
Location |
Size (mm) |
number |
Small nodules located within 2 cm below the pleura and distributed along the bronchi |
|
1 |
LLL RLL |
11 x 12 |
multiple |
yes |
2 |
RLL |
44 x 27 |
multiple |
yes |
3 |
LUL RLL |
5 |
multiple |
yes |
4 |
RLL |
37 x 25 |
single |
no |
5 |
RLL |
15 x10 |
multiple |
yes |
6 |
RML |
48 x 36 |
multiple |
yes |
7 |
RLL |
16 x 8 |
multiple |
yes |
8 |
RUL LLL |
5 x 5 |
multiple |
yes |
9 |
LUL |
12 x10 |
multiple |
yes |
10 |
RML RUL |
24 x14 |
multiple |
yes |
11 |
RLL LLL |
40 x 29 |
multiple |
yes |
12 |
Both lungs |
40 x 26 |
multiple |
yes |
13 |
RUL |
5 x 5 |
single |
no |
14 |
Right lung |
30 x 20 |
multiple |
yes |
15 |
LLL |
13x10 |
single |
no |
16 |
Right lung |
17 x 15 |
multiple |
yes |
17 |
RLL |
5 x 5 |
multiple |
no |
18 |
RUL |
14 x11 |
multiple |
yes |
19 |
Both lungs |
40 x 20 |
multiple |
yes |
20 |
Both lungs |
43 x 20 |
multiple |
yes |
21 |
LLL |
21 x 9 |
multiple |
yes |
22 |
RLL LLL |
22 x16 |
multiple |
yes |
23 |
RLL |
17x9 |
multiple |
yes |
Table 3: Comparison of CT scan finding between first and second CT reading in all trainers except new added information during second CT reading*.
CT findings |
First |
Second |
p |
Location (central, peripheral, subpleural) |
2.12 |
2.21 |
0.144 |
Number of involved segments |
2.41 |
1.88 |
0.246 |
Number of nodules (no, single, multiple) |
1.34 |
1.49 |
0.315 |
Tree-in-bud centrilobular nodule (no, yes) |
0.11 |
0.09 |
0.612 |
Distribution and Shape of nodule or mass (tree-in-bud, round, ellipsoid, others) |
2.59 |
2.92 |
0.225 |
Number of mass (no, single, multiple) |
0.65 |
0.69 |
0.958 |
Pneumonic consolidation (no, yes) |
0.32 |
0.51 |
0.455 |
Cavity of nodule or mass (no, yes) |
0.36 |
0.38 |
0.787 |
Lymph node enlargement associated pulmonary lesions (no, yes) |
0.14 |
0.13 |
0.957 |
Pleural effusion (no, yes) |
0.01 |
0.00 |
0.317 |
Enhancement of nodule or mass (no, yes) |
0.48 |
0.58 |
0.129 |
If enhanced, check heterogenous or homogenous status |
1.11 |
1.10 |
0.666 |
*The second CT reading provided clinical information and new CT finding such as a cluster of nodules or masses located within 2 cm below the pleura and distributed along the bronchi. Mann-Whitney U Test are employed to compare each factor between first and second CT reading. *These numbers are calculated to the average value on different CT items, which provided by all residents score.
Table 4: Simple linear regression results for CT findings and second diagnostic accuracy for Cryptococcal Pneumonia.
P value |
Adjusted R Square |
|
location |
0.941 |
-0.047 |
number of involved segments |
0.991 |
-0.048 |
number of nodules |
0.437 |
-0.017 |
tree-in-bud centrilobular nodule |
0.798 |
-0.044 |
distribution along the bronchi and shape of nodule or mass |
0.018 |
0.203 |
number of masses |
0.068 |
0.109 |
pneumonic consolidation |
0.738 |
-0.042 |
cavity of nodule or mass |
0.807 |
-0.045 |
lymph node enlargement associated pulmonary lesions |
0.686 |
-0.039 |
Enhancement of nodule or mass |
0.007 |
0.268 |
Simple liner regression is employed to calculate correlation between CT factors and diagnosis accuracy for cryptococcal pneumonia on chest CT.
Discussion
Through this study, it was found that pulmonary cryptococcosis with multiple nodules or masses distributed mostly located within 2 cm below the pleural and distributed along the bronchi, which could play an important role in diagnosing cryptococcal pneumonia on chest CT. It is important for the radiologist to be familiar with the CT appearance of this disease to expedite the diagnosis. Hui et al [12] reported that the largest lesions of 47.6% of the multiple nodules/masses had a diameter of 1.1 to 2.0 cm. Most of the lesions were circular and distributed along the bronchial bundle, without the treein-bud pattern. Nodules/masses with an irregular margin were often located in the peripheral region of the lung without pleural effusion. These findings are similar to those in our study. Also, Xie et al [13] describe that the laterality of parenchymal changes was right-sided (n=28, 38.9%), left-sided (n=18, 25%), or bilateral (n=26, 36.1%). The transaxially distribution of the abnormalities was predominately peripheral (n=47, 65.2%), central (n=2, 2.8%), peribranchial (n=14, 19.4%), and random (n=9, 12.5%). Wang et al [11] demonstrated correlation CT findings for cryptococcal pneumonia with pathologic findings for distribution of pulmonary nodules. They found that pulmonary cryptococcosis has certain unique characteristics that multiple nodules or masses mostly located within 2 cm below the pleura, the circular nodules distributed along the bronchi are mostly lobulated, cavities and calcifications may be present. In this study, 19 (82.6%) patients were observed the distribution of located within 2 cm below the pleural and distributed along the bronchi on chest CT. These findings on CT are thought to play a very important role in differentiating PC from other diseases, such like bacterial pneumonia, lung cancer and tuberculosis.
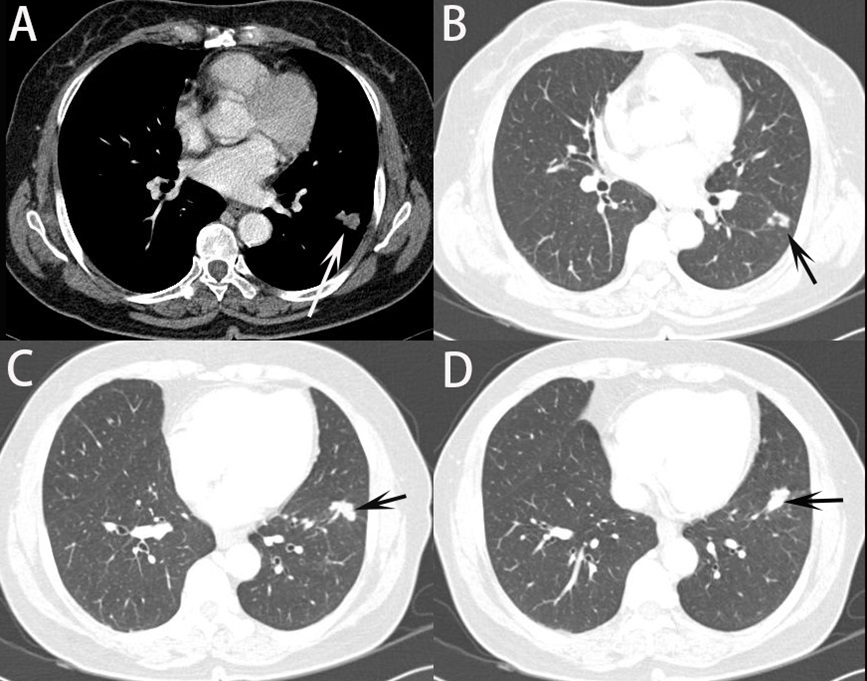
CT features of PC could mimic those of malignant lesions, leading to misdiagnosis [14-16]. PC in immunocompetent patients is indolent and usually presents as a localized nodule/ mass. Some nodules and masses were ill-defined or eccentric with irregularly thick-walled cavities, resulting in misdiagnosis of tuberculosis, lung cancer or hematogenous metastasis, et al (Figure 5). Xin et al. [17] reported that thirteen (31.0%) patients were initially misdiagnosed with lung cancer because CT findings of PC is sometimes nonspecific. In this case, clinically, it is very common for the patient to have to undergo unnecessary examination. In our study, 26.7% patients were initially misdiagnosed with lung cancer, which was similar with Xin et al’s article. In practice, it is still difficult to make differential diagnosis between lung cancer and pulmonary cryptococcosis on CT images. In our study, however, after providing the clinical information, the diagnosis accuracy of PC improved in the second CT reading (from 8.70% to 19%, p=0.007). Therefore, when it is difficult to differentiate PC from other diseases such as lung cancer on CT, it is necessary to make an effort to make an accurate diagnosis by referring to clinical information and personal history. Clinical information played an extremely important role in the analysis and interpretation of CT scans and follows Bayesian logic (conditional probability). Clinical information must be accurate to improve the radiological report. In a Leslie et al [18], 19% of CT reports were changed after clinical information was known, over half the changes being major. The more complex the investigation and the more images acquired, the more likely were changes to be made. When clinical information was correct, 83% of reports became more accurate and 17% became less accurate after the clinical information was known. In our study, after clinical information was known, 82.6% of reports became more accurate to improve correct diagnosis of PC. It can be predicted that clinical information is very important factor for diagnosis PC
On pathology, due to the small diameter of the spores of cryptococcus neoformans (1-2 µm), they are prone to be inhaled to reach the bronchioles and terminal bronchioles [19], where they are engulfed by a large number of macrophages around the tracts below the bronchioles and within alveolar septa, [20] to then form inflammatory granulomas. Inflammatory granulomas will cause edema, neovascularization, and early stage fibrosis [21], which is easily to increase attenuation in the lung on CT, and the heterogeneous pattern is easily to find. If such pathological findings of PC are well recognized and these findings are applied when reading chest CT, it is judged that it will be very useful for diagnosing PC on CT and differentiating it from other diseases.
Limitations
There were several limitations to this study. It was a retrospective study that mostly depended on medical records and patients at different stages of disease. This may have influenced the frequency of findings such as nodule and mass. Besides, the number of patients in our study is limited. The limitations of this study also include the lack of blood laboratory test results. Antigen detection of cryptococcus is also important for the diagnosis of cryptococcal disease, but such tests were not performed. We did not culture and type the cryptococcal strains; thus, the distributions of C. neoformans and C. Gattai in 23 patients and their relationship with the patients’ immune status remain unknown. The unavailability of histological specimens for all patients limited the pathological–radiological correlation.
Conclusion
In conclusion, we found that in order to accurately diagnose pulmonary cryptococcal pneumonia, clinical information including the patient’s immune status and nodules clustered within 2 cm of the bronchi on the chest CT verified in this study are helpful. For improve accuracy on diagnosis PC, in addition, enhancement of nodule or mass on chest CT should be more considered.
Acknowledgments: I would like to thank all trainers (Jung Hwan Kim, Dong Han Shin, Min Jee Kim, Young Ju Song, Hyun Soo Bae, Seo Yun Choi, Da Eul Lee, Su Bin Lee) to read chest CT for cryptococcal pneumonia twice.
Conflict of interest statement and funding: This paper has no conflicts of interest and a statement indicating any source of funding or financial interest.
References
- Sarosi GA. Cryptococcal pneumonia. Semin Respir Infect. 1997; 12: 50-53.
- Woodring JH, Ciporkin G, Lee C, et al. Pulmonary cryptococcosis. Semin Roentgenol. 1996; 31: 67-75. DOI: https://doi. org/10.1055/s-0031-1295720.
- Levitz SM. The ecology of Cryptococcus neoformans and the epidemiology of cryptococcosis. Rev Infect Dis. 1991; 13: 1163- 1169. DOI: https://doi.org/10.1093/clinids.
- Rozenbaum R, Gonçalves AJ. Clinical epidemiological study of 171 cases of cryptococcosis. Clin Infect Dis. 1994; 18: 369-380. DOI: https://doi.org/10.1093/clinids/18.3.369.
- Kerkering TM, Duma RJ, Shadomy S. The evolution of pulmonary cryptococcosis: clinical implications from a study of 41 patients with and without compromising host factors. Ann Intern Med. 1981; 94: 611-616. DOI: https://doi.org/10.7326/0003-4819-94- 5-611.
- Khoury MB, Godwin JD, Ravin CE, et al. Thoracic cryptococcosis: immunologic competence and radiologic appearance. AJR Am J Roentgenol. 1984; 142: 893-896. DOI: https://doi.org/10.2214/ ajr.142.5.893.
- Gordonson J, Birnbaum W, Jacobson G, et al. Pulmonary cryptococcosis. Radiology. 1974; 112: 557-561. DOI: https://doi. org/10.1148/112.3.557.
- Feigin DS. Pulmonary cryptococcosis: radiologic-pathologic correlates of its three forms. AJR Am J Roentgenol. 1983; 141: 1262-1272. DOI: https://doi.org/10.2214/ajr.141.6.1262.
- Liang Y, Shen Y, Zhang J, et al. Pulmonary Cryptococcosis Among 3 Immunocompetent Patients Misdiagnosed as Bacterial Pneumonia and Pulmonary Tuberculosis. Chest. 2016; 149: A114. DOI: https://doi.org/10.1016/j.chest.2016.02.119.
- Sung JH, Kim DH, Oh MJ, et al. A Case of Pulmonary Cryptococcosis in an Immunocompetent Male Patient Diagnosed by a Percutaneous Supraclavicular Lymph Node Biopsy. Tuberc Respir Dis (Seoul). 2015; 78: 276-280. DOI: https://doi.org/10.4046/ trd.2015.78.3.276.
- Wang D, Wu C, Gao J, et al. Comparative study of primary pulmonary cryptococcosis with multiple nodules or masses by CT and pathology. Exp Ther Med. 2018; 16: 4437-4444. DOI: https:// doi.org/10.3892/etm.2018.6745.
- Deng H, Zhang J, Li J, et al. Clinical features and radiological characteristics of pulmonary cryptococcosis. J Int Med Res. 2018; 46: 2687-2695. DOI: https://doi.org/10.1177/0300060518769541.
- Xie LX, Chen YS, Liu SY, et al. Pulmonary cryptococcosis: comparison of CT findings in immunocompetent and immunocompromised patients. Acta Radiol. 2015; 56: 447-453. DOI: https://doi. org/10.1177/0284185114529105.
- Sudhakaran S, Bashoura L, Stewart J, et al. Pulmonary Cryptococcus Presenting as a Solitary Pulmonary Nodule. Am J Respir Crit Care Med. 2017; 196: 1217-1218. DOI: https://doi. org/10.1164/rccm.201703-0601IM.
- Wang SY, Chen G, Luo DL, et al. (18)F-FDG PET/CT and contrastenhanced CT findings of pulmonary cryptococcosis. Eur J Radiol. 2017; 89: 140-148. DOI: https://doi.org/10.1016/j. ejrad.2017.02.008.
- Guimaraes MD, Marchiori E, Meirelles GS, et al. Fungal infection mimicking pulmonary malignancy: clinical and radiological characteristics. Lung. 2013; 191: 655-662. DOI: https://doi. org/10.1007/s00408-013-9506-0.
- Sui X, Huang Y, Song W, et al. Clinical features of pulmonary cryptococcosis in thin-section CT in immunocompetent and nonAIDS immunocompromised patients. Radiol Med. 2020; 125: 31- 38.
- Leslie A, Jones AJ, Goddard PR. The influence of clinical information on the reporting of CT by radiologists. Br J Radiol. 2000; 73: 1052- 1055. DOI: https://doi.org/10.1259/bjr.73.874.11271897.
- Zaragoza O, Rodrigues ML, De Jesus M, et al. The capsule of the fungal pathogen Cryptococcus neoformans. Adv Appl Microbiol. 2009; 68: 133-216. DOI: https://doi.org/10.1016/S0065- 2164(09)01204-0.
- Ma H, May RC. Virulence in Cryptococcus species. Adv Appl Microbiol. 2009; 67: 131-190. DOI: https://doi.org/10.1016/ s0065-2164(08)01005-8
- Shah KK, Pritt BS, Alexander MP. Histopathologic review of granulomatous inflammation. J Clin Tuberc Other Mycobact Dis. 2017; 7: 1-12. DOI: https://doi.org/10.1016/j.jctube.2017.02.001.