Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Cushing’s disease recurrence in two patients in the postpartum period: A case report
Zahra Davoudi1*; Kave Ebrahimzade2; Mohammadjavad Honarvar3; Elena Jamali4; Guive Sharifi2
1 Department of Endocrinology, Skull Base Research Center of Shahid Beheshti University of Medical Sciences, Tehran, Iran.
2 Department of Neurosurgery, Skull Base Research Center of Shahid Beheshti University of Medical Sciences, Tehran, Iran.
3 Endocrine Research Center, Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Iran
4 Department of Pathology, Loghman Hakim Medical Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
*Corresponding Author: Zahra Davoudi
Department of Endocrinology, Skull Base Research
Center of Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Email: dr.daavoudi@sbmu.ac.ir, davoudi5842@yahoo.com
Received : Dec 06, 2021
Accepted : Jan 06, 2022
Published : Feb 02, 2022
Archived : www.jcimcr.org
Copyright : © Davoudi Z (2022).
Abstract
Introduction: Few reports are available on Cushing’s Disease (CD) occurrence in pregnancy. We report two cases of pituitary macroadenoma with initial remission, who experienced uneventful pregnancies in the setting of CD recurrence in the postpartum period.
Case presentation: The cases were two women in their 30s with irregular menses, weight gain, and other CD symptoms. The laboratory and imaging workups indicated the diagnosis of CD, and subsequently, the patients were treated with Endoscopic Transsphenoidal Surgery (ETSS). After one and four years of remission, the patients became pregnant, and no signs or symptoms of CD were present in the meantime; 5-6 months after the delivery of healthy babies, the patients attended our clinic with complaints of weight gain and abnormal menstrual patterns. Findings from the workups confirmed CD recurrence. The patients underwent surgery and are now in the phase of remission.
Conclusion: Overall, patients in remission seem to have a higher risk of CD recurrence in gestation and the postpartum period. This may raise the hypothesis that hypothalamic-pituitaryadrenal axis hyperactivity and hypercortisolism result in ACTH pituitary adenoma during pregnancy. Regarding negative results for estrogen receptors in the patients’ pathology, the association of hyperestrogenemia with hypercortisolism tends is questioned. Thus, the precise mechanism of CD recurrence in pregnancy is unknown, and further studies need to be performed.
Keywords: cushing disease; pituitary corticotroph adenoma; gestation; peripartum; relapse.
Abbreviations: CD: Cushing’s Disease; CRH: Corticotropin Releasing Hormone; LH: Luteinizing hormone; FSH: Follicle-Stimulating Hormone; IGF-1: Insulin-Like Growth Factor; ACTH: Adrenocorticotrophic Hormone; UFC: Urinary Free Cortisol; ODST: Cortisol After Overnight Dexamethasone Suppression Test; HDST: Cortisol after High Dose Dexamethasone Suppression Test; TSH: Thyroid-Stimulating Hormone; IHC: Immunohistochemistry; ETSS: Endoscopic Transsphenoidal Surgery; ER: Estrogen Receptor.
Citation: Davoudi Z, Ebrahimzade K, Honarvar M, Jamali E, Sharifi G. Cushing’s disease recurrence in two patients in the postpartum period: A case report. J Clin Images Med Case Rep. 2022; 3(2): 1632.
Introduction
Cushing’s disease (CD) is a rare endocrinopathy mainly due to pituitary adenoma with an incidence rate of 1.2-6.2 cases per million that mostly affects women of reproductive ages [1]. CD incidence is reported to be less common during gestation, probably due to hypercortisolism and hyperandrogenism in which several etiologies such as hypothalamic-pituitary-gonadal axis impairment may be responsible [2]. Hypercortisolism during pregnancy may lead to different complications, including hypertension, hyperglycemia, cardiovascular events, preeclampsia, and even death; hence, our knowledge in early diagnosis of CD in pregnancy should improve [3,4].
There are a handful of case reports regarding CD occurrence and recurrence [5,6]. Recent studies have focused on pregnancy-associated Cushing’s disease (CD onset in pregnancy or postpartum period) and assessing its relationship with pregnancy [3,7]. In case reports in the literature, almost all pregnancyassociated CD patients had symptoms during gestation. We report two patients with successful uncomplicated pregnancies diagnosed with CD recurrence in the year following childbirth.
Case presentations
First case
In 2016, a 30-year-old married woman was referred to Loghman Hakim hospital endocrinal outpatient clinic with complaints of abnormal menstrual patterns and weight gain by 45 kg in recent years. Clinical presentations of this patient were weakness, fatigue, hair loss, and blurred vision. The patient also had a history of infertility. Observed signs in physical exam included moon face appearance, plethoric face, acne, central weight gain, alopecia, hirsutism, striae, easily bruised skin, and proximal muscle weakness. The patient weighed 123 kg for 167 cm resulting in a BMI of 44.1.
According to the differential diagnoses, requested laboratory tests were as follows (Table 1): Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH) serum level of 3 mIU/ ml (normal range: 5-20 IU/L), Thyroid-Stimulating Hormone (TSH) of 7 mU/L (reference range: 0.5-4.5 mU/L), T4 of 4.7 mcg/ dl (reference range: 4-12.5 mU/L), prolactin of 30 ng/mL (reference range: 2-29 ng/mL), Insulin-Like Growth Factor (IGF-1) of 131 ng/ml (reference range:105-300 mcg/ml), and serum cortisol base of 28 mcg/dl (reference: 6-23 mcg/dl). Also, an elevated urine-free cortisol (850 mcg/24hr; reference range: 23-195 mcg/24 hr) was present in lab results. Adrenocorticotrophic Hormone (ACTH), cortisol after Overnight Dexamethasone Suppression Test (ODST) concentration and urine-free cortisol after High Dose Dexamethasone Suppression Test (HDST) were 65 pg/ml (reference range: 5-37 pg/ml), 12 mcg/dl, and 120 mcg/24 hr, respectively.
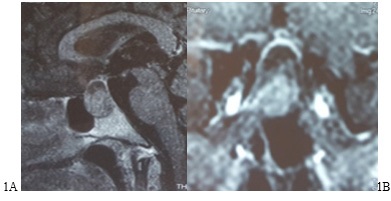
In the next step, high-resolution brain Magnetic Resonance Imaging (MRI) was performed. A 2 cm X 2.5 cm pituitary macroadenoma with extension to the suprasellar area was reported (Figure 1). The findings in laboratory values and imaging suggested the diagnosis of CD. Subsequently, the patient was treated with Endoscopic Transsphenoidal Surgery (ETSS). Histopathologic findings revealed a 0.2 cm X 0.6 cm X 1 cm pituitary adenoma with increased mitosis, a positive Immunohistochemistry (IHC) staining of ACTH, and a negative IHC staining of GH, FSH, LH, and prolactin. There was also a positive PS3 and a ki67 more than 7-8%. After the surgery, the patient entered the remission phase with a 50 kg weight loss and started to have regular menses. Post-operation laboratory values are illustrated in Table 1.
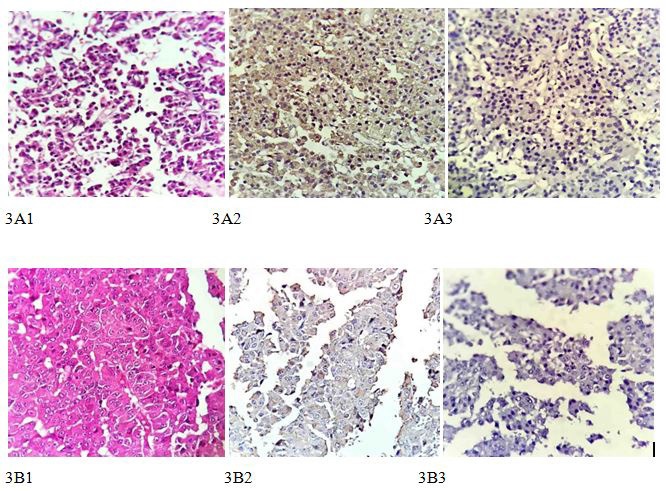
Four years after surgery, the patient became pregnant. During pregnancy, neither clinical presentations related to CD nor any abnormalities in blood pressure and blood glucose test were detected in routine antenatal clinical visits by the Obstetrics and Gynecology (OB-GYN) specialist. Six months after the baby’s delivery, the patient attended our outpatient clinic due to menstrual irregularity and weight gain. Laboratory values at the time of attendance are shown in Table 1. In the new MRI images from the patient, a remnant mass (microadenoma) in the right side of Sella turcica suggestive of CD recurrence was reported. After completion of tests and imaging, the patient once again underwent surgery and is now in remission. Post-up laboratory studies of the patient are shown in Table 1. Histopathological findings revealed a positive immunohistochemistry staining for ACTH, negative for P53 and Estrogen Receptor (ER), and Ki67 about 6%. (Figure 3).
Second case
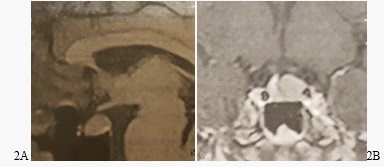
A 69-yIn 2017, a 31-year-old multiparous woman presented to Loghman Hakim outpatient clinic of endocrinology due to abnormal menstrual cycles, weight gain, hirsutism, and a history of hypertension. In clinical examination, moon face appearance, plethora, and central weight gain were detected. Measured anthropometric values in the clinic were weight, height, and BMI of 75 kg, 165 cm, and 27.54, respectively. Laboratory tests performed showed a serum level LH of 0.7 mIU/ml, FSH of 3 mIU/ml, TSH of 0.2 mU/L, T4 of 4.7 mcg/dl, prolactin of 12 ng/mL, and IGF-1 of 200 ng/ml, cortisol base of 16 mcg/dl and a Urinary Free Cortisol (UFC) base of 476 mcg/24 h. Cortisol after ODST and UFC after HDST were 7 mcg/dl and 140 mcg/24 hr, and ACTH was 66 pg/ml. MRI imaging reported a macroadenoma (1.1 X 1 cm mass) with extension to the left suprasellar area (Figure 2), confirming Cushing’s disease. Hence, the patient was treated with ETSS. Gross pathology showed a 0.6 X 0.5 X 0.2 mass with ACTH immunoreactive cells in immunohistochemistry. Later after surgery, the patient’s symptoms, menstrual cycles, and high blood pressure resolved, and her weight returned to normal range, decreasing by 16 kg. Post-up laboratory followup is shown in Table 1.
After being in remission for a year, the patient got pregnant. During gestation, she did not come for follow-up visits to the endocrine clinic and only attended OB-GYN follow-ups. In the meantime, no symptoms or clinical findings indicating CD recurrence were reported in the routine OB-GYN clinical visits by the specialist or the patient. Five months after delivery, the gynecologist referred the patient to the endocrinology clinic due to dysfunction in menstrual cycles and weight gain. Laboratory studies are illustrated in Table 1.
Imaging of MRI showed a 3 X 4 mm mass in the left side of Sella turcica in favor of CD recurrence. The patient underwent a second surgery and is now in remission. We performed IHC staining for ACTH and Estrogen Receptor (ER) on pathology samples of the patient, which showed to be positive for ACTH and negative for estrogen (Figure 3).
Discussion
Imaging features of HCC on MRI
Both of our cases primarily had CD secondary to ACTH-secreting macroadenoma, accounting for only 6% of CD cases [8]. The patients achieved remission and fertility restoration after transsphenoidal surgery and subsequently successful uneventful pregnancies. Five to six months after delivery, CD recurrence was detected. It is noteworthy that in addition to suggested long-term surveillance for all patients with a history of previously treated CD [9], screening or surveillance prior to pregnancy is particularly of high importance in female cases.
CD mostly affects women of reproductive age, and no definite reason has been described for this age and gender distribution. Although some studies have investigated CD occurrence, most have excluded CD in pregnant women due to its scarcity, thus missing pregnancy as a potential factor [10]. In our case report, as well as previously reported cases, the random association between pregnancy and CD occurrence is under question. There may be several reasons why pregnancy-associated CD was not reported in the past. Firstly, in most studies, pregnant patients were excluded as confounders. Secondly, due to physiology in pregnancy (HPA axis activation) and its after effect, recognition and diagnosis of CD by the patient and the caregiver are problematic, and misdiagnosis or late diagnosis are probable.
There are three scenarios and roles for pregnancy in CD recurrence. First, these patients may have had dormant or small slow-growing pre-existing corticotroph microadenomas that grew as a result of hypercortisolism during pregnancy and following childbirth [3], indicating pregnancy as a stimulant for CD. Alternatively, pregnancy may have induced pituitary corticotroph cells’ tumorigenesis through changes in pituitary cells’ receptors, Corticotropin-Releasing Hormone (CRH), or estrogen levels and subsequently resulted in CD. In a recent retrospective study in the United States, of 64 women diagnosed with CD, 11 patients (17% of women and about 27% of women of reproductive age) were detected within a year of pregnancy. Thirty-six percent of pregnancy-associated CD patients had a prior CD surgery, while this was 43% for patients without pregnancy-associated CD. Also, pregnancy-associated CD patients had a median of 2 pregnancies prior to CD, whereas this was zero for the nonpregnancy-associated group [7]. This may indicate pregnancy as a potential independent risk factor for CD occurrence and recurrence.
In this case report, our patients may have had hypercortisolism without metabolic complications (hypertension and hyperglycemia) during pregnancy that was clinically asymptomatic or not chemically significant; patients with mild hypercortisolism as our patients, are diagnosed in late trimesters of pregnancy or deferred until after delivery. The reason may be the cumulative effect of hypercortisolism in pregnancy and subsequent tumorigenesis. To this date, it is not known whether pregnancy is a risk factor for CD recurrence or acts more of a stimulant for a pre-existing pituitary adenoma. According to the evidence, all pregnancy-associated CD cases in the literature had at least a prior pregnancy or a history of CD [3,7].
Secondly, there are other various possible explanations apart from just hypercortisolism for the increase in corticotroph stimulation, such as CRH production by the placenta, hypersensitivity of corticotroph cells to CRH, or hyposensitivity to negative feedback of cortisol [7,11,12]. Thirdly, the reason for the stimulation of pituitary corticotroph cells may lie within changes in hormone levels, namely an over 100-fold elevation in estrogen level within gestation [13]. The subsequent increase in corticotroph activity may trigger proliferation and tumorigenesis in these cells resulting in pregnancy-associated CD [11,14]. The start and development of tumorigenesis in pituitary cells can occur through nuclear receptors of ERα and ERβ, as ERα and ERβ were expressed in pituitary adenomas of different subtypes (e.g. growth hormone, prolactin, and ACTH- secreting adenomas) [15].
To investigate the relationship between the effect of estrogen and pituitary corticotroph hyperactivity, we performed IHC staining for Estrogen Receptor (ER) in addition to ACTH on pathology samples of the patients. ACTH staining in IHC was positive in both patients, which was in line with the pathology of previous studies. Regarding the negative result for estrogen receptors in these tumors, the association of hyperestrogenemia with hypercortisolism is questioned as a stimulus; further research is required to define the exact role of estrogen in pituitary ACTH-secreting adenomas.
Conclusion
Our findings in this case report study suggest clinicians raise their clinical awareness in pregnancy-associated CD settings and take caution with symptoms of weight gain or inability of weight loss in postpartum, which were the most frequent complaints and presentations, especially in the presence of CD history. From the findings of this study and with respect to the pathology of the tumors, estrogen may not play an important role in pregnancy-associated CD.
Declarations
Acknowledgment: None.
Funding: The Authors of this paper have no funding to declare.
Disclosure of interest: The authors report no conflict of interest.
References
- Broder MS, Neary MP, Chang E, et al. Incidence of Cushing’s syndrome and Cushing’s disease in commercially-insured patients <65 years old in the United States. Pituitary. 2015; 18: 283-289.
- Unuane D, Tournaye H, Velkeniers B, et al. Endocrine disorders & female infertility. Best Pract Res Clin Endocrinol Metab. 2011; 25: 861-873.
- Tang K, Lu L, Feng M, et al. The Incidence of Pregnancy-Associated Cushing’s Disease and Its Relation to Pregnancy: A Retrospective Study. Front Endocrinol (Lausanne). 2020; 11: 305.
- Pivonello R, De Martino MC, Auriemma RS, et al. Pituitary tumors and pregnancy: the interplay between a pathologic condition and a physiologic status. J Endocrinol Invest. 2014; 37: 99- 112.
- Semple CG, McEwan H, Teasdale GM, et al. Recurrence of Cushing’s disease in pregnancy. Case report. Br J Obstet Gynaecol. 1985; 92: 295-298.
- Costenaro F, Rodrigues TC, de Lima PB, et al. A successful case of Cushing’s disease pregnancy treated with ketoconazole. Gynecol Endocrinol. 2015; 31: 176-178.
- Palejwala SK, Conger AR, Eisenberg AA, et al. Pregnancy-associated Cushing’s disease? An exploratory retrospective study. Pituitary. 2018; 21: 584-592.
- Woo YS, Isidori AM, Wat WZ, et al. Clinical and biochemical characteristics of adrenocorticotropin-secreting macroadenomas. J Clin Endocrinol Metab. 2005; 90: 4963-4969.
- Atkinson AB, Kennedy A, Wiggam MI, et al. Long-term remission rates after pituitary surgery for Cushing’s disease: The need for long-term surveillance. Clin Endocrinol (Oxf). 2005; 63: 549-559.
- Braun LT, Rubinstein G, Zopp S, et al. Recurrence after pituitary surgery in adult Cushing’s disease: A systematic review on diagnosis and treatment. Endocrine. 2020; 70: 218-231.
- Lindsay JR, Nieman LK. The hypothalamic-pituitary-adrenal axis in pregnancy: Challenges in disease detection and treatment. Endocr Rev. 2005; 26: 775-799.
- Polli N, Pecori Giraldi F, Cavagnini F. Cushing’s disease and pregnancy. Pituitary. 2004; 7: 237-241.
- Brue T, Amodru V, Castinetti F. MANAGEMENT OF ENDOCRINE DISEASE: Management of Cushing’s syndrome during pregnancy: solved and unsolved questions. Eur J Endocrinol. 2018; 178: R259-R266.
- Mastorakos G, Ilias I. Maternal and fetal hypothalamic-pituitaryadrenal axes during pregnancy and postpartum. Ann N Y Acad Sci. 2003; 997: 136-149.
- Shimon I, Melmed S. Pituitary Tumor Pathogenesis1. The Journal of Clinical Endocrinology & Metabolism. 1997; 82: 1675-1681.