Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Focal nodular hyperplasia in infancy: A case report
Sanaa Bouramdane1*; Sarra Benmiloud1; Mohamed Hbibi1; Mariam Boubbou2; Youssef Bouabdellah3; Nawal Hammas4; Mustapha Hida1
1 Department of Pediatrics, Hassan II University Hospital Center, Fez, Morocco.
2 Department of Radiology, Hassan II University Hospital Center, Fez, Morocco.
3 Department of Pediatric Surgery, Hassan II University Hospital Center, Fez, Morocco.
4 Department of Anatomopathology, Hassan II University Hospital Center, Fez, Morocco.
*Corresponding Author: Sanaa Bouramdane
Department of Pediatrics, Hassan II University Hospital
Center, Fez, Morocco.
Email: sanaabouramdane@gmail.com
Received : Jan 15, 2022
Accepted : Feb 09, 2022
Published : Feb 16, 2022
Archived : www.jcimcr.org
Copyright : © Bouramdane S (2022).
Abstract
Focal nodular hyperplasia is a benign disorder of the liver, and it rarely occurs among children. It is characterized by hepatocyte hyperplasia with a central stellate scar. The diagnostic is usually based on radiological investigations, yet a needle biopsy could be necessary if there is still a diagnostic doubt. The difficulty lies in managing children without comorbidities and underlying liver disease. We report a case of a 10 year old girl who has a focal nodular hyperplasia affecting the left hepatic lobe; due to the enlargement of the tumor, she underwent a successful surgical treatment. The aim of our case report is to put the light on the focal nodular hyperplasia in children.
Keywords: focal nodular hyperplasia; benign liver disorder; children; imaging; watchful waiting; surgery.
Citation: Bouramdane S, Benmiloud S, Hbibi M, Boubbou M, Bouabdellah Y, et al. Focal nodular hyperplasia in infancy: A case report. J Clin Images Med Case Rep. 2022; 3(2): 1665.
Introduction
Primary liver tumors represents 1 to 2% of all pediatric solid tumors [1]. They are malignant in 57% of cases and benign in 43% of them [1]. Focal nodular hyperplasia (FNH) represents less than 2% [2]. The pathogenesis is not totally understood yet. It’s a well-circumscribed mass with a central stellate scar [3]. It’s usually asymptomatic, so the diagnosis is made incidentally [1,4]. In most cases, the diagnostic can be made by imaging only; however, sometimes it is necessary to make a needle biopsy to exclude a malignant liver tumor. The observation with serial checks and surgery are the two first-line management options [4]. In this observation, we report the case of a young girl who underwent a surgical treatment after the increase in her tumor’s size.
Case report
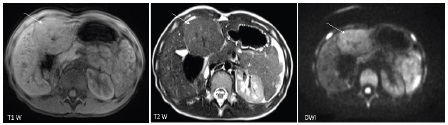
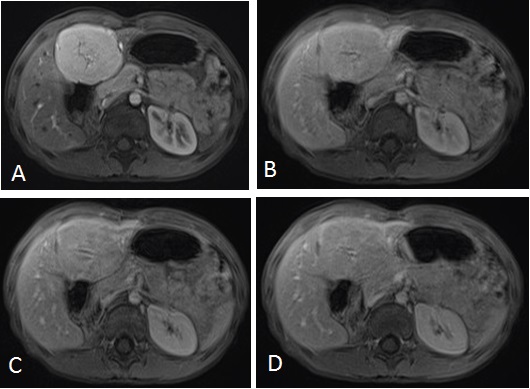
A 10 year old girl, with no past medical history, was admitted in our institution for an abdominal mass that was detected coincidentally by her mother. During the physical examination, we found an isolated mass in the left hepatic lobe. The patient had no associated symptoms. The laboratory tests showed a normal liver function, a normal alpha-fetoprotein (AFP) and beta HCG levels. The abdominal ultrasound (US) imaging revealed no abnormalities. The liver MRI showed a 55 mm X 50 mm well circumscribed mass in the left liver; isointense to the liver parenchyma in T1 weighted sequences (Figure 1). In T2 weighted sequences, the mass was slightly hyperintense to the liver parenchyma (Figure 1); the central stellate scar was hyperintense. After intra venous injection of gadolinium, the dynamic contrast enhanced sequences demonstrated an early enhancement (wash in) in the arterial phase, without wash out in the portal phase, and the enhancement of the central scar in the late phase (Figure 2). The lesion of FNH was feeding by a branch of the hepatic artery.
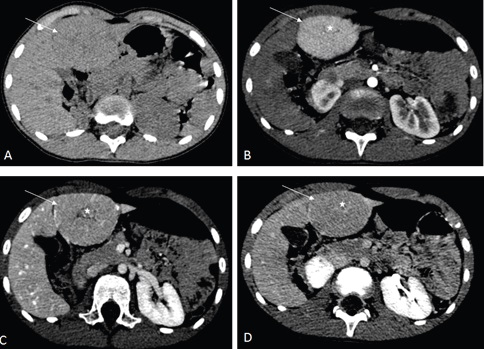
The management of this patient was based on watchful waiting. The re evaluation 6 months later by the computed tomography (CT) scan showed a 62 mm X 50 mm X 60 mm liver lesion, with the some imaging features as seen in the MRI (Figures 3 and 4).
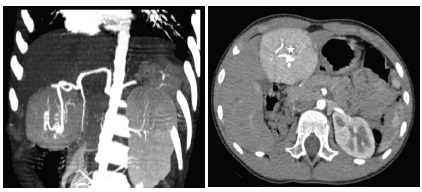
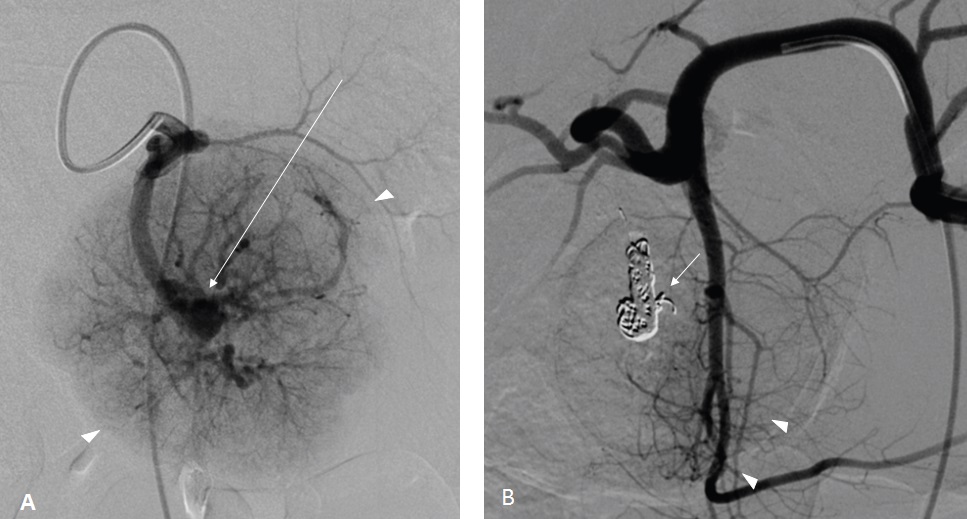
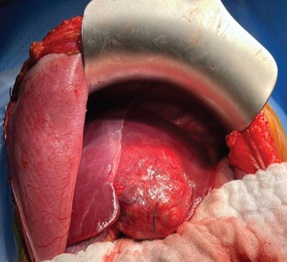
The surgical resection of the mass was indicated according to the increasing size of the tumor. An embolization of the feeding artery was done first; the flow to the FNH was reduced by 70% (Figure 5). The surgeon found a well-defined encapsulated mass in the 4th liver segment (Figure 6); he performed a total tumorectomy (Figure 7); the feeding vessels were ligated and dissected.
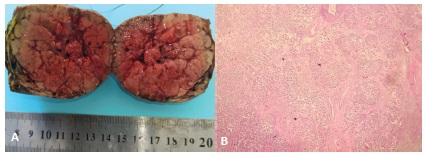
The pathological examination of the specimen showed a 60 mm X 58 mm X 45 mm lobular well-defined tumor, brown in color, with a nodular structure and central stellate scar; the lesion was composed of blund hepatocyts, surrounded by fibrous septas, that contain artery branches (Figure 8). The morphologic features were consistent with FNH. After surgery, the girl recovered well without any complications.
Discussion
The FNH is a benign hepatic tumor that usually affects middle aged women. It rarely occurs among the pediatric population [3,5,6]. In the literature, there have been reported about 200 cases of children aged between 7 and 14 years [7]. The etiology is unclear, many theories discussed aberrant vascularization by anomalous artery, reactive hyperplasia after hepatic injury or aberrant increased blood flow [7]. The risk factors include history of malignancy diseases (Wilms’ tumors, neuroblastoma...), liver diseases (biliary atresia, Alagille syndrome….), chemotherapy, radiotherapy, congenital or surgical portosystemic shunts and vascular malformations [2,4,7-9]. The FNH is occasionally multifocal; it’s often occurs in the left hepatic lobe [1]. The histological composition of FNH include a normal hepatocytes proliferation, bile ducts and kupffer cells separated by septas, these ones are originating from a central fibrous scar rich on blood vessels [3,9].
Most of the patients are asymptomatic at the diagnosis. The mass is incidentally detected on imaging for other clinical condition check-ups [1,4]. During the physical examination, the patient usually has a right upper abdominal mass. Sometimes, an abdominal pain may be present as well [1,10]. The liver function tests and the levels of AFP are normal in the majority of cases [6,10]; however, few patients with increased AFP are reported in the literature [2]. In our case, it was a fortuitous discovery; the patient has only a palpable mass.
The abdominal ultrasound plays a prominent role in the diagnosis of FNH. It typically appears as a well-circumscribed homogeneous mass, mostly isoechoic to the adjacent liver and the typical central scar is slightly hyperechoic but not always visualized (20% of cases) [1,2,9]. On the CT scan, the lesion characteristics are more specific; before the intravenous contrast injection, the FNH is usually looks focal homogenous and iso or slightly hypodense compared to the normal liver tissue, a central hypodense scar can be observed as well. After contrast administration, rapid uniform enhancement is seen in the arterial phase more than the liver tissue with the hypodense stellate scar in some cases. The mass become iso attenuating to the liver in the portal venous and delayed phases. The central scar is only seen in 50 to 70% of the cases, it typically shows late enhancement [1,9]. The MRI demonstrates a slightly hypointense mass to normal parenchyma in T1 weighted images. A mildly hyperintense lesion with a scar is seen in T2 weighted sequences. Dynamic contrast enhanced sequences shows an intense centrifugal enhancement on the arterial phase with retention contrast on delayed images, and a late scar enhancement (46,8% of cases) [2,3,9,11]. A biopsy is recommended when the patient have an atypical imaging features [1,3,10,11].
Recently, the management guidelines for FNH in pediatric population has evolved. Zarfati et al [4] proposed watchful waiting with serial checks in the absence of major symptoms or compression of the adjacent structures. The regular checkups are based on physical examination, AFP and liver function tests, abdominal ultrasound and/or MRI every 6 months for 2-4 years then reduced to once per 12 months [7]. During this period, if the patient presents major symptoms or complications, a radiological and surgical evaluation must be performed. The surgical treatment is indicated for symptomatic or complicated cases and for diagnostic doubt. Isolated growth in the size of the lesion should not be considered as an indication for surgery [1,3,4,9,10,12]. Moreover, a successful trans-arterial embolization of FNH in pediatric population have been reported, as aminimally invasive approach especially for hypervascular lesions [3]. In our situation, we indicated surgical excision because of the tumor’s increasing size and the parent’s insistence to remove the mass. Thus far, no histologic malignant transformations in FNH have been reported [1,3,10]; the progressive growth of the tumor can cause venous compression and mass effect on the adjacent structures [4,8].
Conclusion
The rarity of FNH in the pediatric population makes the management a challenge. It’s based at first on confident diagnosis. This one is difficult due to clinical and imaging similarities with differential diagnoses, such as fibrolamellar hepatocellular carcinoma and other malignancies.
References
- Zhuang, et al. Huge focal nodular hyperplasia presenting in a 6-year-old child: A case presentation. Int. J. Surg. Case Rep. 2016; 29: 76-79.
- Aboughalia KM, Chisholm RS, Iyer. Focal nodular hyperplasia masquerading as malignancy in an infant with elevated alphafetoprotein: A case report with literature review. Clin. Imaging 2021; 69: 228‑232.
- Shanmugasundaram, et al.Embolization of a rare case of focal nodular hyperplasia in an adolescent boy, J. Pediatr. Surg. Case Rep. 2021; 64: 101713.
- Zarfati, et al. Management of focal nodular hyperplasia of the liver: Experience of 50 pediatric patients in a tertiary center. J. Pediatr. Surg. 2020; 55(9): 1885‑1891.
- Ribera Cano M, Debette-Gratien B, Descottes, J Languepin. Hyperplasie nodulaire multifocale compliquant un cavernome porte. Arch. Pédiatrie. 2007; 14(11): 1315‑1317.
- RH O’Kelly, DJ Mole. Benign liver lesions. Surg. 2020; 38(8): 472-479.
- Fuchs SW, Warmann C, Urla JF, Schäfer, et al. Management of benign liver tumors. Semin. Pediatr. Surg.2020; 29(4): 150941.
- Akhaladze, et al. Liver resection for atypical giant focal nodular hyperplasia. J. Pediatr. Surg. Case Rep. 2019; 47: 101202.
- T Ma, et al. Focal nodular hyperplasia in children: An institutional experience with review of the literature. J Pediatr. Surg. 2019; 50(3): 382-387.
- De Luca M, Zamparelli C, Fadda, Martone, et al. Focal nodular hyperplasia of the liver in infancy: a case report. J. Pediatr. Surg. 2006; 41(2): 456‑457.
- Lautz N, Tantemsapya A, Dzakovic, Superina, et al. Focal nodular hyperplasia in children: clinical features and current management practice. J. Pediatr. Surg. 2010; 45(9): 1797‑1803.
- Fuchs SW, Warman C, Urla JF, Schäfer, et al. Management of benign liver tumors. Semin. Pediatr. Surg. 2020; 29(4): 150941.