Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Successful treatment of congenital chylothorax using povidone iodine in trisomy 21 newborn
FZ Elhanafi1,2*; F Bennaoui1,2; Z Lazrak1,2; N Elidrissi Slitine1,2; FMR Maoulainine1,2
1 Neonatal Intensive Care Unit, Mother and child Pole, Mohammed VI University Hospital, Marrakesh, Morocco.
2 Child Health and Development Research Laboratory, Marrakech School of Medicine, Cadi Ayyad University, Marrakech, Morocco.
*Corresponding Author: Fatima Ezzohra Elhanaf
Neonatal Intensive Care Unit, Mother and child
Pole, Mohammed VI University Hospital, Marrakesh, Morocco.
Email: fz.elhanafi@gmail.com
Received : Jan 05, 2022
Accepted : Feb 11, 2022
Published : Feb 18, 2022
Archived : www.jcimcr.org
Copyright : © Elhanafi FZ (2022).
Abstract
Congenital chylothorax is known as abnormal leakage and accumulation of chyle in the pleural space. As one of the minimally invasive treatments, chemical pleurodesis has been more frequently used in treating pediatric chylothorax in recent years. We report through this observation the clinical case of a newborn male with Down’s syndrome born at 38 weeks of amenorrhea admitted to our unit for a respiratory distress after his birth. On exploration we concluded right unilateral congenital chylothorax. Treatment consisted in an evacuation of pleural fluid with a chemical pleurodesis based on povidone iodine with good clinical and radiological outcome.
Keywords: congenital chylothorax; chemical pleurodesis; down’s syndrome; povidone iodine.
Citation: Elhanafi FZ, Bennaoui F, Lazrak Z, Slitine NE, Maoulainine FMR. Successful treatment of congenital chylothorax using povidone iodine in trisomy 21 newborn. J Clin Images Med Case Rep. 2022; 3(2): 1673.
Introduction
Congenital chylothorax is often caused by congenital lymphatic system malformation or obstruction of its main tributaries [1]. Although congenital chylous effusions are relatively rare in infancy, they have serious clinical consequences and can be a potentially life-threatening discorder [1]. It was reported that approximately 4.9% of foetuses diagnosed with Down syndrome had pleural effusions [2]. At present, it is mainly treated through conservative and minimally invasive approaches however, few patients undergo surgical treatment such as ligation of thoracic duct [3]. In recent years, Povidone-Iodine Chemical Pleurodesis (PICP) is expected to be a new minimally invasive treatment for pediatric spontaneous chylothorax following the reports of some successful cases [4]. We report a case of congenital chylothorax treated effectively using povidone iodine.
Observation
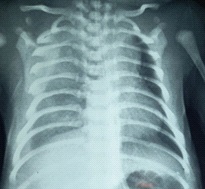
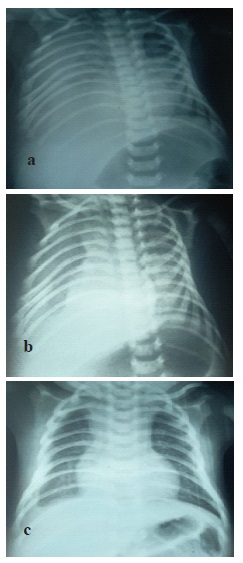
He was a newborn male, from a followed monofetal pregnancy estimated at 38.5 weeks of gestation. The mother was 38 years old, with no notable pathological history, 4 gestity, 4 parity. The duration of labor was not specified, she had a non instrumental vaginal delivery, the birth weight was 3700 g, the amniotic fluid was clear. Cephalic presentation was noted and the apgar score was 10/10th at the first and at the 5th min. The newborn was admitted to the neonatal intensive care unit for respiratory distress at birth rated at 3/10th according to the Silverman score associated with cyanosis with 76% oxygen saturation in the air room, and a facial dysmorphism compatible with trisomy 21. The imaging confirmed pleural effusion with minimal abundance on the right side (Figure 1).
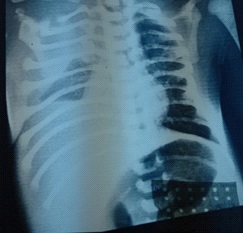
The infectious assessment was normal. The exploratory pleural puncture bringing back 20 ml of citrus yellow liquid. The analysis of the pleural fluid had confirmed the chylous nature with a rate of white blood cells which was 1920/mm3 and lymphocytes at 80%, a protein level at 32 g/l, and a dosage of triglycerides at 270 mg/dl, the liquid’sculture was sterile. The newborn was put on oxygen therapy. Nutritionally, the child was not receiving parenteral nutrition due to the unavailability in our context, so we kept the child under breastfeeding and artificial milk enriched with medium chain triglycerides. Considering trisomy 21, the karyotype showed homogeneous trisomy 21. A thyroid checkup was performed showing congenital hypothyroidism for which the child was put on levothyroxine sodium. The echocardiogram was normal. On the 8th day of life, the newborn presented worsening signs of respiratory status and desaturation to 84% and an increase in the quantity of pleural fluid (Figure 2). A second puncture was made bringing 40 ml of orange yellow liquid having allowed the stabilization of the patient.
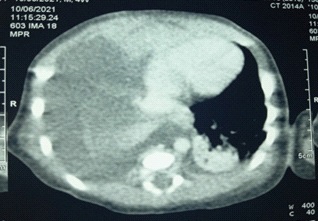
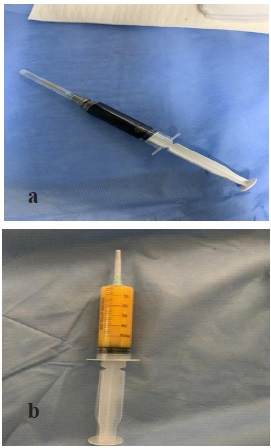
A complement by CT scan was requested to research a Congenital Pulmonary Lymphangiectasia (CPL) or lung malformation (Figure 3). In the 18th day of life, the decision of a chemical pleurodesis with povidone iodine was taken due to the persistence and worsening of the radiological pleural effusion. Intra pleural instillation of 5 ml of povidone iodine diluted with 0.9% saline was performed after evacuation of 60 ml of chyle under ultrasound guidance (Figure 4). Monitoring of renal function before and after the procedure did not reveal any abnormality.
Vital Parameters monitoring did not show any complications after instillation. The patient could be weaned from oxygen on the eighth day after instillation. He was discharged on day 29 of life with good clinical and radiological outcome.
Discussion
Congenital chylothorax is the most common cause of pleural effusion in the neonatal period in the absence of trauma or surgery [5], with an incidence of 1 in 6,000 to 24,000 live births. The condition carries a high mortality rate up to 60% and affects boys more than girls, at a ratio of about 2:1 [6]. This results in acute respiratory distress and can lead to immunodeficiency and malnutrition if persistent [7]. IN 1977, YOSS et al, first reported two patients with trisomy 21 complicated by congenital chylothrax [8]. It can be idiopathic due to congenital lymphatic malformation, atresia or hypoplasia of the thoracic duct or may be associated with various genetic diseases such as Noonan’s, Turner’s and Down’s syndromes [1]. Our patient was diagnosed as trisomy 21, and his spontaneous unilateral chylothorax might be related with genetic disease. The neonatal chylothorax appear in 75% of cases during the first week [9], this was the case of our patient in whom the symptoms were present at birth. Regarding localization, in the study by Randrianambinina et al, bilateral chylothorax dominated in 52.94% of cases, the same results found in the study by Bellini et al with predominance of bilateral involvement in 66.6% of cases [10,11]. The diagnosis of chylothorax is established by measuring triglycerides level in the pleural fluid. A concentration of greater than 110 mg/dL strongly supports the diagnosis of a chylothorax [1]. Chest X-ray should be performed to identify pleural effusion. Ultrasonography will determine whether there is free fluid in the pleural space or organised as seen in empyema. Chest CT scans and lymphangiography can be helpful to identify the site of chyle leakage and to determine the cause of chylothorax [1]. The ideal management of the patients with chylothorax is unknown. Primary hydrothorax with small volumes of pleural fluid and no hydrops is more likely to resolve or remain stable, so more conservative treatment can be provided [12]. Failure of conventional treatment should lead to intravenous or subcutaneous administration of somatostatin or its synthetic analogue (Octreotide), chemical pleurodesis and as a last resort surgical treatment. Chemical pleurodesis is a well accepted treatment for persistent or recurrent pleural effusion [13]. Its mainly used as a minimally invasive treatment for patients with pneumothorax and pleural effusion [14], but its use is still rarely reported in the treatment of chylothorax. Brissaud first reported using PICP to treat congenital chylothorax in 2003 [15]. However, the efficiency of PI as a sclerosant has not been fully demonstrated up to now, as the incidence of chylothorax is low and there have been few related studies. Borcyk et al. [16] and Hmami et al. [15] respectively reported that a child with repeated chylothorax who responded poorly to conservative therapies was cured by using PI. Murki et al. [1] reported that using 4 mL of 4% PI diluted with 6 mL of normal saline into 10 mL and keeping in the thoracic cavity for 5 h by bilateral CP 3 times cured a child with congenital chylothorax. We used this technique in our patient after failure of conventional treatment with good clinical and radiological results after a single intrathoracic instillation of povidone iodine. Regarding the side effects of this technique; It was reported that either topical iodopovidone applications or the use of iodine-based contrast agents may cause transient thyroid dysfunction. High concentrations of exogenous iodine are capable of blocking the organic-binding and coupling reactions and decrease thyroid hormone synthesis. Some cases of renal failure were reported [17]. Our patient was diagnosed with congenital hypothyroidism but no worsening of thyroid or renal function was noted after intrathoracic instillation of povidone iodine.
Conclusion
Congenital chylothorax is a known complication of trisomy 21 that should be detected in any neonatal respiratory distress in this population. Certainly rare but can be fatal if treatment is delayed. Treatment by chemical pleurodesis with povidone iodine remains an inexpensive and effective therapeutic alternative to be offered in the event of failure of conservative treatment, as proven in our patient.
References
- William TM. Congenital chylothorax and trisomy 21 syndrome. J Pediatr Neonat Individual Med. 2018; 7: e070133.
- Hagay Z, Reece A, Roberts A, Hobbins JC. Isolated fetal pleural effusion: A prenatal management dilemma. Obstet Gynecol. 1993; 81: 147-152.
- Tutor JD. Chylothorax in infants and children. Pediatrics. 2014; 133: 722-733.
- Wen-Jie Wang. Povidone-iodine chemical pleurodesis in treating spontaneous chylothorax in pediatric patients. Ann Palliat Med. 2020. http://dx.doi.org/10.21037/apm-20-926.
- Nathan S. Rubalcava et al. Efficacy of Early Pleurectomy for Severe Congenital Chylothorax. Journal of surgical research. 2020; 433-438.
- White MK, Bhat R, Greenough A. Neonatal Chylothorax: A 10- Year Experience in a Tertiary Neonatal Referral Centre. Case Rep Pediatr. 2019; 2019: 3903598.3
- Philippa Lauren Ball, et al. Rare case of congenital chylothorax and challenges in its management. BMJ Case Rep. 2019; 12: e228023.
- Ozden turan, et al. Chylothorax and respiratoy distress in a new born with trisomy 21. Eur J Pediatr. 2001; 160: 744-745.
- Randrianambinina F, et al. Management of chylothorax in children in Madagascar Prise en charge des chylothorax de l’enfant à Madagascar. Journal of Functional Ventilation And Pulmonology © 2020 Jfvp. www.jfvpulm.com.
- Bellini C, Ergaz Z, Radicioni M, et al. Congenital fetal and neonatal visceral chylous effusions: Neonatal chylothorax and chylous ascites revisited. A multicenter retrospective study. Lymphology 2012; 45: 91–102.
- Meira O, et al. Neonataler chylothorax bei trisomy 21. Z Geburtsh neonatal. 2004; 208: 29-31.
- Li Cao, et al. Fetal pleural effusion and Down syndrome. Intractable & Rare Diseases Research. 2017; 6: 158-162.
- Hmami F, et al. Chylothorax congénital: Une réponse rapide et totale à la polyvidone iodée. Archives de Pe´diatrie. 2014; xxx: 1-4.
- Mierzejewski M, Korczynski P, Krenke R, et al. Chemical pleurodesis - A review of mechanisms in volved in pleural space obliteration. Respir Res. 2019; 20: 247-253.
- Brissaud O, Desfrere L, Mohsen R, et al. Congenital idiopathic chylothorax in neonates: Chemical pleurodesis with povidoneiodine (Betadine). Arch Dis Child Fetal Neonatal Ed. 2003; 88: F531-F533.
- Borcyk K, Kamil A, Hagerty K, et al. Successful management of extremely high- output refractory congenital chylothorax with chemical pleurodesis using 4% povidone-iodine and propranolol: A case report. Clin Case Rep. 2018; 6: 702-708.
- Maoulainin FMR, et al. Congenital Chylothorax: Rapid and Effective Response to Polyvidone Iodine. Open Journal of Pediatrics. 2016; 6: 10-13.