Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
The impact of immunotherapy in the treatment of solid tumors. What we found under 2021 Christmas tree
Antonio M Grimaldi*; Mario Vittorio Nicola Ferrante; Vincenza Tinessa; Domenico Germano; Maria Anna Bareschino; Vincenzo Ricci; Immacolata Paciolla; Emiddio Barletta
Medical Oncology Unit, AORN “San Pio”, Benevento, Italy.
*Corresponding Author: Antonio M Grimaldi
Medical Oncology Unit, AORN “San Pio”, Benevento,
Italy
Email: dott.a.m.grimaldi@gmail.com
Received : Jan 18, 2022
Accepted : Feb 14, 2022
Published : Feb 21, 2022
Archived : www.jcimcr.org
Copyright : © Grimaldi AM (2022).
Abstract
The treatment of solid tumors sees an increasing use of immune checkpoint inhibitors (anti PD-1/PDL-1, anti CTLA-4). The new drug combinations have revolutionized the treatment of different types of cancer, from melanoma to chest neoplasms, renal and esophageal cancer. All of these different diseases have one common denominator: the immune system. Only the immune system, properly stimulated, can attack and destroy different types of cancer, as if they were an infection. This review describes the most important milestones achieved in 2021 by combination treatment with immune checkpoint inhibitors.
Keywords: immunotherapy; Anti CTLA-4; Anti PD-1/PDL-1; combination immunotherapy; melanoma; lung cancer; kidney cancer; esophageal cancer.
Citation: Grimaldi AM, Ferrante MVN, Tinessa V, Germano D, Bareschino MA, et al. The impact of immunotherapy in the treatment of solid tumors. What we found under 2021 Christmas tree. J Clin Images Med Case Rep. 2022; 3(2): 1680.
Introduction
The data presented in 2021 confirmed the increasingly important role of immunotherapy in the treatment of solid tumors. About 10 years after the approval of the first immune checkpoint inhibitor in the treatment of metastatic melanoma, ipilimumab, the therapeutic scenario has significantly changed not only for melanoma, but also for other solid tumors. In fact, the anti PD-1 drugs (nivolumab, pembrolizumab, cemiplimab), anti PD-L1 (atezolizumab, durvalumab,) and anti CTLA-4 (ipilimumab, tremelimumab), both in monotherapy and in combination, are revolutionizing the prognosis of patients with different neoplasms. Clinical trials with longer follow-up have clearly demonstrated what the real benefit of modern immunotherapy is the long-term benefit, the patient survival.
Metastatic melanoma
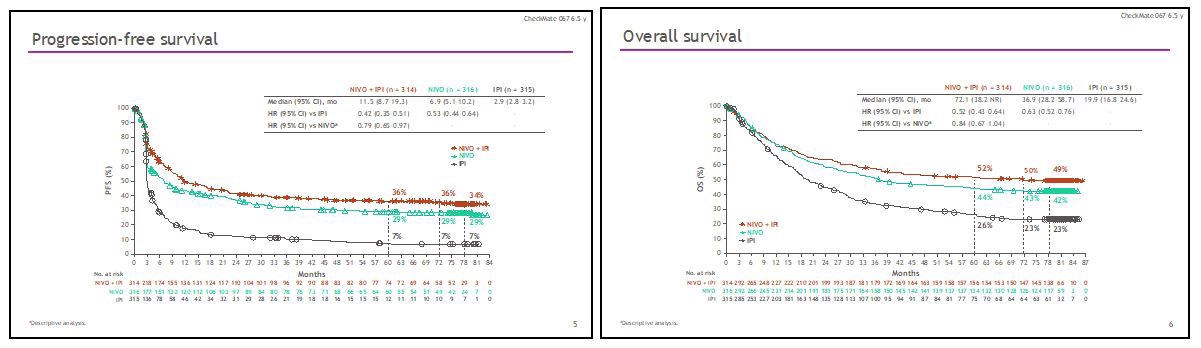
Metastatic melanoma has been the test bench of modern immunotherapy. Precisely in this cancer, with a particularly poor prognosis and orphan of effective therapies, in which chemotherapy had a merely palliative effect [1], immunotherapy has shown that it can revolutionize the survival of patients even in particularly difficult subsets. The clinical trial Checkmate 067 randomized patients with both BRAF mutant and wild type metastatic melanoma to the treatment with nivolumab, ipilimumab, or the combination of the two immune checkpoint inhibitors. This is the clinical trial with longest follow-up in metastatic melanoma. What emerges clearly from the latest update of this study is the progressive increase in the response rate obtained with immunotherapy, both in monotherapy and in combination. This is due to the progressive effect of the immune response which, once triggered, remains lasting and therefore a partial response can become complete over time. In fact, at the various time-points of the study, the complete response rate went from 17.2% for ipilimumab plus nivolumab and 14.9% for nivolumab alone, to 19.4% and 16.5%, to 21.3% and 17.7%, up to 23 % and 19% at current 6.5-year follow-up. This phenomenon also occurs in patients who have discontinued treatment for reasons other than disease progression. With the latest update of the study, the reported objective response is 58% for the combination and 45% for nivolumab alone, while the median duration of responses has not yet been reached for both treatments.
In addition, with a minimum follow-up of 77 months, 34% of the patients treated with the combination and 29% of the patients treated with nivolumab did not develop disease progression. At the same time point, the median survival for this study is 72.1 months (Figures 1 and 2) for the combination of ipilimumab plus nivolumab, the longest ever achieved in a clinical trial in the treatment of metastatic melanoma, and is 36.9 months for the monotherapy, while 49% of patients treated with the combination and 42% of patients treated with nivolumab are still alive. Although the number of BRAF-mutated and wild-type patients is not adequately balanced in this study, there is evidence that median survival has not yet been reached in mutated patients treated with the combination and that it is 45.5 months for patients treated with nivolumab, whereas, in wild type patients, the mOS is 39.1 months for the combination and 34.4 months for monotherapy. Furthermore, patients with normal LDH, and fewer than 3 metastatic disease sites, now recognized as favorable prognostic factors in metastatic melanoma, have a significant advantage in terms of both PFS and OS when treated with the combination or monotherapy. In patients with elevated LDH and more than 3 sites of disease, the advantage of PFS and OS of the combination ipilimumab plus nivolumab over monotherapy is particularly significant, a difference not as marked in patients with normal LDH and fewer than three affected sites. Among patients who discontinue therapy due to toxicity, 77% of patients treated with the combination and 69% of patients treated with nivolumab alone are free from subsequent therapy and therefore from progression, further confirmation of the long-term benefit of immunotherapy. So much effectiveness, however, has a high cost in terms of toxicity. In fact, the combination ipilimumab plus nivolumab is burdened by 59% of G.3 / 4 toxicity with 31% discontinuation, compared to 24% of serious adverse events and 8% discontinuation of mono immunotherapy [2].
The combination of the two immune checkpoint inhibitors has achieved incredible results in a particularly difficult subset of patients, those with brain metastases. The encephalic localizations of the disease are often the principal cause of death of patients with metastatic melanoma due to the refractoriness to the various treatments and the consequent complications, such as haemorrhage and epileptic seizures, so much so that the median survival of these patients is about 16 weeks. If anti BRAF target therapy achieve a significant response rate in BRAF-mutated patients, unfortunately this did not guarantee an important survival advantage due to the short duration of the responses obtained.
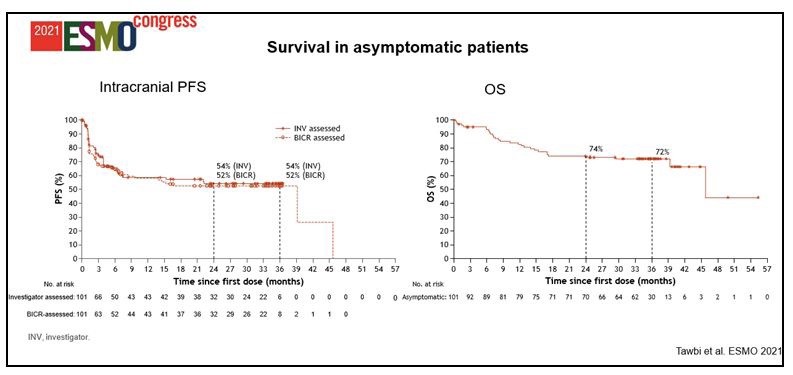
Therefore, the treatment of brain metastases remains an unmet medical need. The Checkmate 204 clinical trial enrolled only patients with brain metastases, dividing them into asymptomatic ECOG PS 0-1 (Cohort A) patients, who were therefore not taking steroids, and symptomatic ECOG PS 0-2 patients on high-dose steroid therapy, and treating them with ipilimumab plus nivolumab as first-line therapy. In cohort A, according to the investigators, the intracranial response rate of the combination was 54%, with 33% complete remissions and 58% of general disease control (clinical benefit rate). The congruity of both intra and extracranial disease responses should also be considered, indicating that the blood-brain barrier does not represent an obstacle for combo-immunotherapy. Another very interesting data is the particularly short median time to response, 1.4 months…! Furthermore, at a median follow up of 34.3 months, 72% of patients are alive with a median survival not yet reached (Figure 3) [3]. This response rate of about 60% with 30% complete remissions was also confirmed by the Australian ABC study which used the combination ipilimumab plus nivolumab in the same subset of patients [4]. Considering these data, the combination of ipilimumab plus nivolumab should be considered the first-line treatment for patients with asymptomatic brain metastases, regardless of BRAF mutational status.
The Cohort B, that of patients with symptomatic brain metastases and, therefore, on high-dose steroid therapy, did not achieve the same results with the combination ipilimumab plus nivolumab both in terms of objective responses, PFS and OS, probably due to cause of the immuno-refractory induced by steroids in these patients. One of the future strategies that could be implemented for this particular subset could be the triple combination of BRAF inhibitors + MEK inhibitors + anti-PD-1, taking advantage of the high response rate of target therapy and the long duration of immunotherapy.
As combinations of highly effective therapies with both molecular targets and immune checkpoint inhibitors are available in the treatment of metastatic BRAF-mutated melanoma, emerges the problem of sequence, the need to answer to the question “which therapy to do first”. This answer was given by two international clinical trials, one randomized phase II, the SECOMBIT trial, and one randomized phase III trial, the DREAMseq. Both studies demonstrated that in patients with BRAFmutated metastatic melanoma, a significant survival benefit is obtained using upfront ipilimumab plus nivolumab therapy and at disease progression, the combination of BRAF plus MEK inhibitors.
A particularly significant result is the 2-year survival rate, 72%, identical in both studies in the cohort of patients treated in first line with ipilimumab plus nivolumab and then with target therapy, a sign of the solidity of the data and its reproducibility in the “Real life” population [5,6]. In view of this, the new treatment algorithms will inevitably indicate that first a combination immunotherapy treatment and then target therapy should be given to the patients with BRAF mutated metastatic melanoma.
Thoracic-pulmonary neoplasms
Immunotherapy has also conquered thoracic oncology both in the treatment of NSCLC, SCLC, and mesothelioma. Just 6 years after the first publication of nivolumab as a therapy for NSCLC, we are observing a real revolution in the long-term prognosis of patients with advanced disease. The latest update of the phase I study CA209-003 of nivolumab in the treatment of patients with advanced and pretreated NSCLC, shows that 5 years after the start of therapy, 16% of patients are still alive. Considering that at 3 years 18% of patients were alive, the longterm benefit given by the anti PD-1 is confirmed, which, with its action, allows to stabilize the survival curve. The comparison with chemotherapy is disarming, because only 4% of these patients were alive at 5 years [7]. A similar data was obtained from the study with pembrolizumab, in which 15.9% of patients were alive at 5 years and which also showed that in patients with PDL-1 expression> 50% 25% were alive at the same follow-up.
Immunotherapy was therefore introduced in second-line clinical practice after the results of 4 phase III clinical trials (Checkmate 017 and 057 with nivolumab, KEYNOTE 010 with pembrolizumab and OAK III with atezolizumab) which demonstrated its efficacy, comparing the treatment with anti PD-1/ PDL-1 with the second/third line standard treatment, the docetaxel. All 4 studies showed a clear and distinct advantage in long-term survival in favor of nivolumab, pembrolizumab and atezolizumab compared to docetaxel, so that chemotherapy was replaced as second-line therapy
Given the efficacy of immunotherapy in pretreated patients, first-line treatment studies have been conducted, and these studies have totally changed the clinical practice. The study KEYNOTE 024 randomized NSCLC patients without EGFR and ALK mutations and PDL-1> 50% to pembrolizumab therapy or 4-6 courses of platinum-based chemotherapy. In this trial, pembrolizumab, compared to chemotherapy, demonstrated a clear advantage in both median (26.3 vs 13.4 months) and 5-year OS (31.9 vs 16.3% - HR 0.62). A similar result has never been achieved in the treatment of NSCLC, with the doubling of survival between the two treatment arms [8]. In light of these successful results, pembrolizumab has been included in international treatment guidelines as first-line therapy for NSCLC patients without EGFR and ALK mutations and with PDL-1> 50%.
In IMpower110 study, atezolizumab, compared with chemotherapy in the treatment of patients with high PDL-1 expression, achieved a significant advantage in median OS (20.2 vs 13.1 months) [9], as well as in EMPOWER Lung 1 trial, another anti PD-1, cemiplimab, in comparison with chemotherapy, in the treatment of NSCLC patients with PDL-1 expression> 50% and in the absence of activating mutations of EGFR and ALK, has shown a significant advantage in survival at 1 (72.4 vs 53.9%) and 2 years (50.4 vs 27.1%) [10].
All the data reported from clinical studies, therefore, reflect what is the action of immunotherapy on the disease and, above all, confirm the long-term benefit given by immunotherapy that reflects in the survival advantage.
In order to increase the proportion of patients who can benefit from immunotherapy, different combinations of treatment with immune checkpoint inhibitors have been tested, and so anti PD-1/PDL-1 drugs have been combined with chemotherapy, anti-angiogenics or other immunotherapeutic agents.
The KEYNOTE189 study compared the combination pembrolizumab plus cis-platinum and pemetrexed with chemotherapy in first-line treatment of NSCLC patients with non-squamous histology, who did not express EGFR and ALK mutations. In this study, chemoimmunotherapy doubled patient survival in all subsets of PDL-1 expression, with particular benefit in patients with PDL-1 expression> 50%, compared to standard chemotherapy [11].
Another interesting combination is the association of immunotherapy with chemotherapy and antiangiogenic therapy. The IMpower150 study combined carboplatin plus paclitaxel standard chemotherapy with bevacizumab and atezolizumab in the treatment of NSCLC patients with non-squamous histology and compared this 4-drug combination with the carboplatin-paclitaxel-bevacizumab and carboplatin-paclitaxel combinations. The 4-drug combination, in this study, achieved the best performance in terms of PFS and OS [12] and is already approved by the FDA.
Regarding NSCLC with squamous histology, the KEYNOTE-407 trial compared the combination of pembrolizumab plus carboplatin and paclitaxel with chemotherapy, demonstrating the superiority of chemo-immunotherapy regardless of PDL-1 expression [13].
Another particularly interesting combination in the treatment of NSCLC is that of two immune checkpoint inhibitors. Nivolumab, anti PD-1 antibody, in combination with ipilimumab, anti CTLA-4, compared with chemotherapy in the firstline treatment of NSCLC without EGFR and ALK mutations, demonstrated a survival advantage over chemotherapy, both in patients with PDL-1 expression > 1% and in PDL-1 negative patients [14].
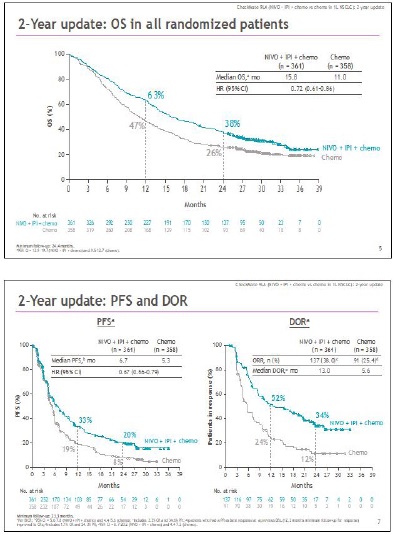
Considering these data, the natural evolution could only be the association of chemotherapy with the combination of anti PD-1 plus anti CTLA-4. The Checkmate 9LA study compared the combination of nivolumab plus ipilimumab and chemotherapy with the same schedule of chemotherapy. In this study, however, chemotherapy in association with immunotherapy was done for only two cycles, while in the control arm for 4-6 cycles.
This innovative design is based on the need to induce a rapid response through chemotherapy, but that the same treatment for 2 cycles can have an additional immune stimulating effect through the release of antigens induced by tumor destruction. The combination of chemo-immunotherapy compared to standard first-line chemotherapy demonstrated a significant advantage in OS both median (15.8 vs 11.0 months) and at 12 (63% vs 47%) and 24 months (38% vs 26%). The efficacy of the 4-drug combination and in particular of immunotherapy is seen above all in PFS at 12 and 24 months, respectively 33% vs 19% and 20% vs 8%, with the prospect that the majority of patients free from progression at 24 months, will remain in this condition at the subsequent follow-up, due to the long-term benefit of immunotherapy (Figures 4 and 5) [15]. One of the issues with treatment with two immune checkpoint inhibitors in combination with chemotherapy could have been the toxicity of the 4-drug combination, but, using ipilimumab at 1 mg/kg every 6 weeks, grade 3 and 4 adverse reactions were 48% in the experimental arm vs 38% in the control arm. This was possible through the containment of the dose of ipilimumab, much lower than the schedule used in metastatic melanoma (3mg/kg in combination with nivolumab 1mg/kg every 3 weeks X 4 cycles, followed by nivolumab alone).
Another study that combined two immunotherapies with two chemotherapeutics was the POSEIDON study [16]. This trial confirmed the data from study 9LA, combining tremelimumab (anti CTLA-4) with durvalumab (anti PDL-1) with dual platinumbased chemotherapy. The chemotherapy treatment, however, in this study was carried out for all 4 cycles, obtaining results comparable to the combination of ipilimumab, nivolumab and only two courses of chemotherapy. The reduction in chemotherapy cycles of study 9LA, burdened by lower treatment toxicity, can therefore increase the number of patients benefiting from the 4-drug combination. Immunotherapy has also revolutionized inoperable third-stage NSCLC. The PACIFIC study demonstrated that after definitive chemo-radiotherapy treatment for inoperable stage III NSCLC, in non-progressed and PDL-1 positive patients, durvalumab maintenance resulted in an impressive survival advantage with a median OS of 47.5 vs 29.1 months as compared with placebo [17]. No maintenance therapy had ever achieved a similar result before this clinical study
The Checkmate 816 clinical trial used nivolumab plus chemotherapy for 3 cycles as neoadjuvant treatment of potentially resectable NSCLC. This combination resulted in a higher pCR and mPR rate (24% and 36%) compared to 2% for chemotherapy. Experimental therapy has also made it possible to reduce major surgery and perioperative complications, which were instead greater in patients treated with chemotherapy [18].
Immunotherapy has also achieved a significant impact in the treatment of SCLC. In extensive disease, the combination atezolizumab plus carboplatin and etoposide has now routinely entered in the clinical practice. This association demonstrated statistically significant superiority compared to chemotherapy alone in the treatment of extended SCLC according with the results of the IMpower133 clinical trial. The advantage was obtained both in mOS (12.3 vs 10.3 months) and at 18 months OS (34.0% vs 21.0%). Although we do not see a striking advantage as in NSCLC, immunotherapy in the treatment of SCLC allowed to give a significant increase in survival [19].
For years the reference treatment of pleural mesothelioma has been the chemotherapy combination cis-platinum plus pemetrexed. The Checkmate 743 clinical trial compared the combination of nivolumab plus ipilimumab with cis-platinum plus pemetrexed as first line treatment of advanced pleural mesothelioma. The median survival in the general study population was higher with immunotherapy (18.1 vs 14.1 months), as was the survival at 12 and 24 months (68% vs 58% and 41% vs 27%). The advantage of combination immunotherapy over chemotherapy becomes striking in the treatment of patients with mesothelioma with non-epithelioid histology. In fact, the median survival was 18.1 months compared to 8.8 months of chemotherapy, and the survival at 12 and 24 months were respectively 63% and 38% vs 32% and 8% of chemotherapy. Also in epithelioid histology, chemo-immunotherapy obtains a significant advantage, but not as striking as in non-epithelioid histology [20]. The possibility of carrying out a first-line immunotherapy treatment also allows to use chemotherapy in second-line, a particularly significant advantage as currently there is no valid therapeutic alternative in this therapy setting. The current research perspectives in the therapy of thoracicpulmonary neoplasms are based on the development of new combination strategies with chemotherapy, anti PD-1 / PDL-1 and anti CTLA-4, in association with new immune checkpoint inhibitors such as anti LAG- 3 and anti TIGIT.
Renal cell carcinoma
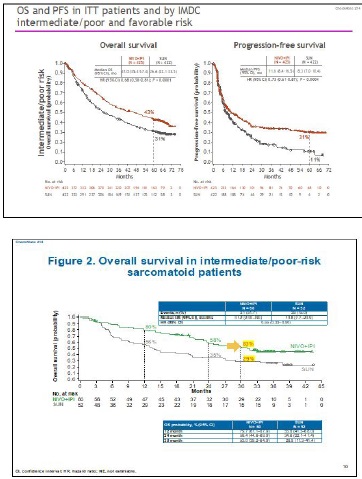
Anti VEGFR tyrosine kinase inhibitors (TKIs) have dramatically changed the natural history of metastatic renal cell carcinoma, but we are experiencing a new revolution with the advent of immune checkpoint inhibitors. In the Checkmate 214 clinical trial, the combination ipilimumab plus nivolumab was compared with sunitinib in the first line treatment of metastatic renal cell carcinoma. At a median follow-up of 48 months, the median survival of patients treated with immunotherapy was not yet reached in the Intent to Treat population, while it was 38.4 months in the arm of patients treated with sunitinib, resulting in 31% reduction in risk of death (HR 0.69 - CI 0.59-0.81). Considering the intermediate and poor prognosis population, the combination of immune checkpoints achieves a mOS of 48.1 months compared to 26.6 months of sunitinib, for a consequent reduction in the risk of death by 35% (HR 0.65). In this subset of patients, the advantage of ipilimumab plus nivolumab is also obtained in PFS with 11.2 months compared to 8.3 months of sunitinib and in terms of objective responses with 42% vs 27%. The study also achieved rapid (2.8 months) and profound responses, with a complete remission rate of 10%, the highest ever achieved in a clinical trial in the treatment of metastatic renal cell carcinoma. The objective responses recorded by the investigators were also confirmed by independent radiologists both in the short term and at a follow-up of 48 months. Another interesting fact is the duration of the response with 86% of responders free from progression 4 years from the start of therapy. As in melanoma, discontinuation of therapy due to toxicity does not represent an unfavorable prognostic factor, indeed 47% of patients who discontinued treatment due to adverse events maintain their response after more than 36 months.
The sarcomatoid variant of renal cell carcinoma has represented for years an unmet medical need as it is resistant to TKIs. This histological type is characterized by a strong PD-L1 expression. In the Checkmate 214 study, there were 139 patients with sarcomatoid variant, 74 treated with ipilimumab plus nivolumab and 65 with sunitinib. Patients treated with immunotherapy achieved a response rate of 61% with 19% complete remissions of the disease. The advantage becomes incredible in PFS, because median PFS was 26.5 months in patients with the sarcomatoid variant treated with ipilimumab plus nivolumab compared to 5.1 months for those treated with sunitinib. At 48 months of median follow-up these combo-treated patients have not yet reached the median OS which is 14.2 months in sunitinib-treated patients (Figures 6 and 7). Also in terms of toxicity, immunotherapy proves to have a better profile as it is burdened by fewer G.3/4 adverse events compared to sunitinib (48% vs 64%). Furthermore, patients who discontinued therapy for toxicity have a longer survival than the population who continued treatment [21].
Esophageal carcinoma
Esophageal carcinoma has always been a malignancy with a very complex management. With 100 patients affected by this tumor, half of them do not practice antineoplastic therapy for reasons related to the patient, 25% practice palliative therapy and 1/4 of the remaining 25% heal, that is about 6%. These results also depend on the patient’s clinical conditions, that often guide therapeutic choices, more than scientific evidence. All of these factors make the management of these patients really complicated.
As a second-line treatment for esophageal cancer, nivolumab was compared with chemotherapy in a phase II study. The benefit was relatively modest with 12- and 18-month survival rates of 47% and 31% compared with 34% and 21% for chemotherapy. Median survival was also superior for nivolumab with 10.9 vs 8.4 months for chemotherapy.
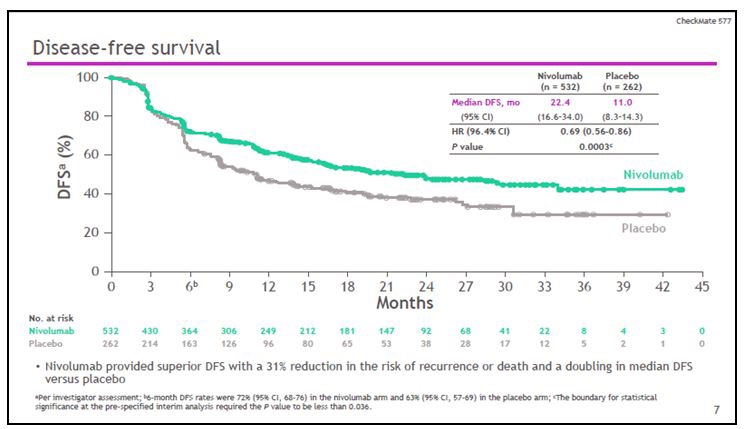
The clinical trial checkmate 577 enrolled patients treated with chemo-radiotherapy and subsequent R0 surgery for esophageal carcinoma with both squamous and adenocarcinoma histology and randomized them (ratio 2: 1) to adjuvant treatment with nivolumab or placebo. The median DFS of the nivolumab arm was doubled compared to placebo with 22.4 vs 11 months, and a reduction in the risk of progression of 31% (HR 0.69). At 36 months, 25% of patients treated with placebo are disease free, as compared with more than 45% in the nivolumab arm (Figure 8). The advantage of immunotherapy is also seen in terms of distant metastasis free survival (DMFS), with a median of 28.3 months for nivolumab compared to 17.6 months for placebo. Furthermore, nivolumab does not negatively impact the quality of life of patients and the benefit of the treatment is evident both in squamous and in adenocarcinoma histology [22].
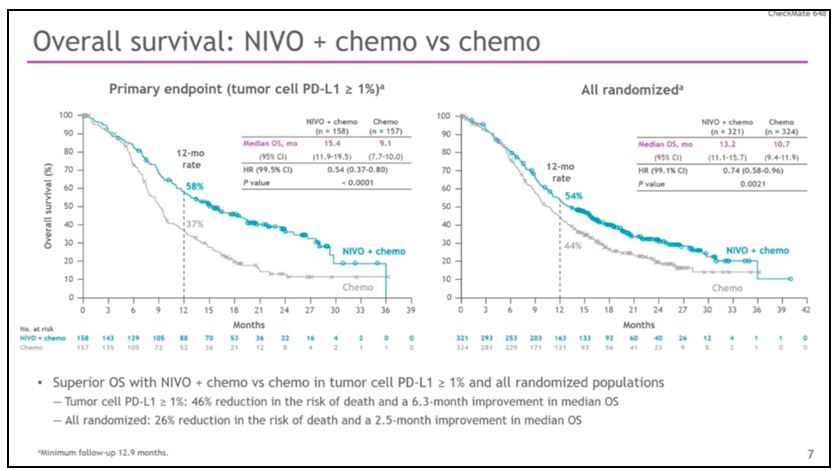
The Phase III clinical trial Checkmate 648 randomized patients with metastatic esophageal carcinoma with squamous histology, to first-line treatment with nivolumab plus chemotherapy, to combo immunotherapy with nivolumab plus ipilimumab, or to chemotherapy with cis-platinum plus 5-fluorouracil. In patients with PDL-1> 1%, the median OS was 15.4 for nivolumab plus chemotherapy versus 9.1 months for chemotherapy alone, with 58% of patients alive at 12 months when treated with the chemo-immune combination versus 37% of patients treated with chemotherapy alone. This leads to a 46% reduction in the risk of death! (HR 0.54). But the survival benefit is also seen in all patients treated with immunotherapy, regardless of PDL-1 status. In fact, in all randomized patients, the median of OS was 13.2 for nivolumab plus chemotherapy compared to 10.7 for chemotherapy alone, with 54% of patients alive at 12 months, when treated with the chemo-immune combination, compared to 44% of patients treated with chemotherapy alone, for a 26% reduction in the risk of death (HR 0.74). In patients with PDL-1> 1%, the median PFS was 6.9 months for the combination chemotherapy versus 4.4 months for chemotherapy alone (Figure 9). At 12 months, 25% of patients treated with nivolumab plus chemotherapy is progression-free and maintain response over the following months, compared with 10% of patients treated with chemotherapy. This rate of patients, however, differently than those treated with immunotherapy, drastically reduces in the following months. The combination nivolumab plus chemotherapy gives an advantage also in terms of objective responses with 53% compared to 20% of chemotherapy alone.
The combination ipilimumab plus nivolumab, compared in the same trial with chemotherapy proved to be superior. In the population with PDL-1> 1%, the median OS was 13.7 for combo immune versus 9.1 months of chemotherapy, and 12-month survival was 57% for nivolumab plus ipilimumab and 37% for chemotherapy alone. At 24 months, survival was tripled with more than 30% of patients alive when treated with the combo immune compared to 10% of those treated with chemotherapy. In terms of PFS, the median is practically comparable between combo immuno and chemotherapy arms, while at 12 months 26% of patients treated with ipilimumab plus nivolumab are progression-free and remain constant in the following months, compared to 10% of patients treated with chemotherapy, which however drastically reduces in the following months. The combination ipilimumab plus nivolumab gives an advantage over chemotherapy alone with 35% responses compared to 20% for chemotherapy, and with long-lasting responses [23].
Even in the treatment of a malignancy with a particularly poor prognosis and complex management such as esophageal carcinoma, immunotherapy is radically changing the prognosis of patients.
Conclusions
The goal of cancer treatments is to increase the survival of patients as much as possible. What immunotherapy has shown is to give the long-term benefit, first in metastatic melanoma, then in other solid tumors. The possibility of having patients alive after years of starting a treatment has also allowed to revolutionize doctor-patient communication, being able to elicit objective hopes with respect to the long-term prognosis even in the case of particularly aggressive tumors, such as lung cancers. All this revolution has taken place in the recent years, in which it has been understood that, by adequately stimulating the immune system, it can attack and destroy solid tumors of different origins. Furthermore, as demonstrated by the adjuvant therapy of melanoma, immunotherapy can definitively cure radically resected stage III and IV patients. The same strategy can also be applied to other neoplasms, in order to increase the number of cured patients more and more.
References
- Korn EL et al. Meta-analysis of phase II cooperative group trials in metastatic stage IV melanoma to determine progression-free and overall survival benchmarks for future phase II trials. col. 2008; 26(4): 527-34. doi: 10.1200/JCO.2007.12.7837.
- Wolchock JD et al. Long-Term Outcomes With Nivolumab Plus Ipilimumab or Nivolumab Alone Versus Ipilimumab in Patients With Advanced Melanoma J Clin Oncol. 2021; JCO2102229. doi: 10.1200/JCO.21.02229.
- Tawbi HA et al. Long-term outcomes of patients with active melanoma brain metastases treated with combination nivolumab plus ipilimumab (CheckMate 204): final results of an open-label, multicentre, phase 2 study. Lancet Oncol. 2021; 22(12): 1692- 1704. doi: 10.1016/S1470-2045(21)00545-3. Epub 2021 Nov 10.
- Long GV et al. Five-year overall survival from the anti-PD1 brain collaboration (ABC Study): Randomized phase 2 study of nivolumab (nivo) or nivo+ipilimumab (ipi) in patients (pts) with melanoma brain metastases (mets). Abstr 9508. J Clin Oncol Meeting Abstract | 2021 ASCO Annual Meeting.
- Atkins MB, et al: DREAMseq: A phase III trial—ECOG-ACRIN EA6134. ASCO Plenary Series. Abstract 356154. Presented November 16, 2021.
- Ascierto PA et al. SECOMBIT: The best sequential approach with combo immunotherapy [ipilimumab (I) /nivolumab (N)] and combo target therapy [encorafenib (E)/binimetinib (B)] in patients with BRAF mutated metastatic melanoma: A phase II randomized study. LBA40 ESMO 2021 Abstract book.
- Gettinger S et al. Five-Year Follow-Up of Nivolumab in Previously Treated Advanced Non-Small-Cell Lung Cancer: Results From the CA209-003 Study J Clin Oncol. 2018; 36(17): 1675-1684. doi: 10.1200/JCO.2017.77.0412. Epub 2018 Mar 23.
- Reck M et al Pembrolizumab versus Chemotherapy for PDL1–Positive Non–Small-Cell Lung Cancer. N Engl J Med. 2016; 375(19): 1823-1833. doi: 10.1056/NEJMoa1606774. Epub 2016 Oct 8.
- Herbst RS et al. Atezolizumab for First-Line Treatment of PDL1–Selected Patients with NSCLC. N Engl J Med. 2020; 383(14): 1328-1339.doi:10.1056/NEJMoa1917346.
- Sezer A et al. Cemiplimab monotherapy for first-line treatment of advanced non-small-cell lung cancer with PD-L1 of at least 50%: a multicentre, open-label, global, phase 3, randomised, controlled trial. Lancet. 2021; 397(10274): 592-604. doi: 10.1016/S0140-6736(21)00228-2.
- Gadgeel S et al. Updated Analysis From KEYNOTE-189: Pembrolizumab or Placebo Plus Pemetrexed and Platinum for Previously Untreated Metastatic Nonsquamous Non-Small-Cell Lung Cancer. J Clin Oncol. 2020; 38(14): 1505-1517. doi: 10.1200/ JCO.19.03136. Epub 2020 Mar 9.
- Socinski MA et al. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. Engl J Med. 2018; 378(24): 2288-2301. doi: 10.1056/NEJMoa1716948. Epub 2018 Jun 4.
- Paz-Ares L et al. Pembrolizumab plus Chemotherapy for Squamous Non–Small-Cell Lung Cancer. N Engl J Med. 2018; 379(21): 2040-2051. doi: 10.1056/NEJMoa1810865. Epub 2018 Sep 25.
- Hellman M et al. Nivolumab plus Ipilimumab in Advanced Non– Small-Cell Lung Cancer. N Engl J Med. 2019 Nov 21;381(21):2020- 2031. doi: 10.1056/NEJMoa1910231. Epub 2019 Sep 28.
- Paz-Ares L et al First-line nivolumab plus ipilimumab combined with two cycles of chemotherapy in patients with non-small-cell lung cancer (CheckMate 9LA): an international, randomised, open-label, phase 3 trial Lancet Oncol. 2021; 22(2): 198-211. doi: 10.1016/S1470-2045(20)30641-0. Epub 2021 Jan 18.
- Johnson ML, et al: Durvalumab ± tremelimumab + chemotherapy as first-line treatment for mNSCLC: Results from the phase 3 POSEIDON study. IASLC 2021 World Conference on Lung Cancer. Abstract PL02.01. Presented September 9, 2021.
- Spigel DR et al. Five-year survival outcomes with durvalumab after chemoradiotherapy in unresectable stage III NSCLC: An update from the PACIFIC trial. J Clin Oncol. Vol 30, Issue 15 suppl.
- Forde PM, Spicer J, Lu S, et al: Nivolumab + platinum-doublet chemotherapy vs chemotherapy as neoadjuvant treatment for resectable (IB-IIIA) non-small cell lung cancer in the phase 3 CheckMate 816 trial. AACR Annual Meeting 2021. Abstract CT003. Presented April 10, 2021.
- Horn L et al. First-Line Atezolizumab plus Chemotherapy in Extensive-Stage Small-Cell Lung Cancer. Engl J Med. 2018 Dec 6;379(23):2220-2229. doi: 10.1056/NEJMoa1809064. Epub 2018 Sep 25.
- Scherpereel BP et al. First-line nivolumab plus ipilimumab in unresectable malignant pleural mesothelioma (CheckMate 743): a multicentre, randomised, open-label, phase 3 trial. Lancet. 2021; 397(10272): 375-386. doi: 10.1016/S0140-6736(20)32714-8. Epub 2021 Jan 21.
- Albiges L et al. Nivolumab plus ipilimumab versus sunitinib for first-line treatment of advanced renal cell carcinoma: extended 4-year follow-up of the phase III CheckMate 214 trial. ESMO Open. 2020; 5(6): e001079. doi: 10.1136/esmoopen-2020-001079.
- Kelly RJ et al. Adjuvant Nivolumab in Resected Esophageal or Gastroesophageal Junction Cancer. N Engl J Med. 2021. PMID: 33789008 Clinical Trial.
- Chau I, Doki Y, Ajani JA, et al: Nivolumab plus ipilimumab or nivolumab plus chemotherapy versus chemotherapy as first-line treatment for advanced esophageal squamous cell carcinoma: First results of the CheckMate 648 study. 2021 ASCO Annual Meeting. Abstract 4001. Presented June 5, 2021.