Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Research Article - Open Access, Volume 3
Intestinal infections due to various protozoan and helminth parasites and bacteria in a tertiary care institute
Sheetal Goenka; Monika Matlani*; Vinita Dogra
Vardhman Mahavir Medical College & Safdarjung Hospital, India.
*Corresponding Author: Monika Matlani
Associate Professor in Vardhman Mahavir Medical
College & Safdarjung Hospital, India.
Email: monikamatlani@yahoo.com
Received : Dec 30, 2021
Accepted : Feb 15, 2022
Published : Feb 22, 2022
Archived : www.jcimcr.org
Copyright : © Matlani M (2022).
Abstract
Diarrhoea due to intestinal parasites and bacteria are prevalent in individuals belonging to the lower socio-economic strata. Infectious intestinal diseases are often self-limiting. They are usually treated without proper diagnosis or information about the etiology. Important bacteria causing diarrhoea are diarrhoeagenic Escherichia coli, Shigella spp, Salmonella spp, and Vibrio cholerae. The common enteric parasites like Giardia lamblia, Entamoeba histolytica, Ascaris lumbricoides, Ancylostoma duodenale, Trichuris trichiura, Enterobius vermicularis, Hymenolepis nana and Strongyloides stercoralis are responsible for considerable morbidity and mortality in the extremes of age. As these parasitic infections are often overlooked, the identification of these pathogenic organisms is of great importance for proper treatment and institution of control measures.Improving the quality of the water, sanitation, and overall hygiene can help prevent diarrhoeal diseases. The accurate diagnosis of the causative agents, an institution of proper treatment and regular monitoring of antimicrobial resistance profiles will help in preventing the spread of multidrug-resistant strains in the community.
Keywords: enteric parasite; diarrhoea; bacteria; hygiene; sanitation.
Citation: Goenka S, Matlani M, Dogra V. Intestinal infections due to various protozoan and helminth parasites and bacteria in a tertiary care institute. J Clin Images Med Case Rep. 2022; 3(2): 1683.
Introduction
Enteric parasitic infections and bacterial diarrhoea are very common in developing countries, affecting almost everyone at some stage in their lives. In a country like India, enteric infections persist and flourish due to inadequate personal hygiene, malnourishment, unsanitary environmental conditions, improper waste disposal, lack of potable water supply, low socioeconomic status, lack of proper toilet facilities and low literacy rates leading to an increased rate of intestinal parasitic infections [1]. The incidence and frequency of intestinal parasitic infections also varies with age, sex and geography [2]. Prompt diagnosis, accurate treatment and timely management can significantly reduce the morbidity and mortality rate associated with infectious diarrhoea.
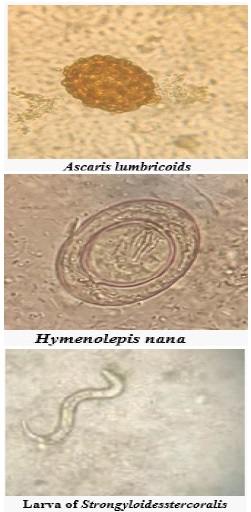
Escherichia coli, Shigella spp, Salmonella spp, and Vibrio cholera are important enteric bacteria causing diarrhoea and dysentery. Giardia lamblia, Entamoeba histolytica, Ascaris lumbricoides, Ancylostoma duodenale, Trichuris trichiura, Enterobius vermicularis, Hymenolepis nana and Strongyloides stercoralis are important parasites responsible for considerable morbidity in children and adults [3,4]. In developing countries children with Giardiasis, often present with acute or chronic diarrhoea, weight loss, flatulence and malabsorption syndrome. According to World Health Organisation reports, Entamoeba histolytica is considered to be the third most common protozoa causing intestinal manifestations.
Many times, parasitic infections are overlooked as important causes of diarrhea, which leads to significant morbidity and mortality. The World Health Organization has reported that approximately 21% of India’s population may be infected with intestinal parasites namely soil-transmitted helminths [5]. The global disease burden attributed to intestinal helminths is 39 million disability-adjusted life years [6]. In India, chemical and biological factors such as the soil consistency, temperature and humidity of the environment have a lot of impact on the degree of endemicity of helminths in a particular area. The development and growth of Ancylostoma require a temperature of 23- 27o C while moisture is important for the survival of the larva. Hence parasitic infections must be looked for judiciously in our setup.
Most of the times clinicians use antibiotics inadvertently without looking for parasitic causes. Overuse/misuse of antibiotics in cases of diarrheas is leading to problems of resistant bacteria like CRE (Carbapenemase Resistant Enterobacteriaceae), hence it is important to identify bacterial and parasitic diarrhea correctly and give antibiotics only if required. Hence this retrospective study was planned to evaluate the prevalence of bacterial and parasitic diarrhea in our settings.
Aim: To study the prevalence of various parasitic and bacterial intestinal pathogens in patients attending a tertiary care institute in New Delhi, India.
Materials & methods
A hospital-based retrospective study was carried out over 5 years (January 2015–March 2021). A total of 2619 stool samples were analyzed for the presence of intestinal parasites and bacteria in the Microbiology laboratory in New Delhi, India. Fresh stool samples were received in laboratory-prescribed leakproof, sterile plastic containers without preservatives. On receipt of the sample in the laboratory, naked eye and microscopic examination of the stool samples was done within a period of 1-2 hours. On naked eye examination of stools, the color, consistency, presence of mucous, blood and the presence of intestinal worms or the segments of tapeworm were looked for. The microscopic examination of the wet mount of stool sample was included saline and iodine preparations which were observed at10x and 40x magnification for the presence of ova, cyst and trophozoites. Stool microscopy for darting motility of Vibrio cholerae was noted. Samples were inoculated directly on the culture media and also after enrichment in Selenite F broth and Alkaline Peptone Water (APW) onto MacConkey’s agar, Xylose Lysine Deoxycholate Agar (XLD) and Bile Salt Agar. Processing of the samples was done as per standard protocols. A battery of biochemical tests was put for identification. Serotyping of the isolates was done wherever required. Antibiotic susceptibility testing of the bacterial isolate was done by the Kirby Bauer disc diffusion technique and its interpretation was done as per CLSI guidelines.
Results
Over a period of 5 years, a total of 2619 stool samples were analyzed for the presence of intestinal parasites and bacteria. Out of these, 448 (17.10%) showed the presence of different species of intestinal protozoan, helminth, and non-lactose fermenting intestinal bacteria. The prevalence rate was found to be 52.80% in males and 47.2% in females. The most common intestinal parasites in the different age groups are summarized in Table 1. Giardia intestinalis and E. histolytica were the commonest parasites found in all age groups. Out of 150 infant patients, intestinal parasitic infection was found in 15 (4.77%) patients. Giardia intestinalis 9 (60%) was the most common parasite followed by Entamoeba histolytica/ Entamoeba dispar 4 (26.66%) and Hymenolepis nana 2 (13.33 %). In the pediatric age group, out of 535 patients, intestinal infection was found in 72 (14.01%) of the patients. Giardia intestinalis 42 (68.18%) was the most commonly identified parasite followed by Entamoeba histolytica/Entamoeba dispar 14 (20.45%), Ascaris lumbricoides 5 (2.27%), Hymenolepis nana 2 (2.27%). In the adult patients, of the 1934 stool samples analyzed, 256 (13.18%) were positive for intestinal parasites as given in Table 1.
Table 1: Parasitic and bacterial infections in patients of different ages.
Pathogenic organisms |
Infants |
Paediatric
|
Adult |
Total |
Parasites |
|
|
|
|
Protozoan parasites |
|
|
|
|
Giardia intestinalis |
9 |
42 |
105 |
156 |
E. histolytica /dispar |
4 |
14 |
156 |
174 |
Balantidium coli |
|
|
15 |
15 |
Trichomonas hominis |
|
|
2 |
2 |
Helminths - ova |
|
|
|
|
Tapeworm |
|
|
1 |
1 |
Trichuris trichura |
|
|
5 |
5 |
Ancylostoma duodenale |
|
|
8 |
8 |
Hymenolepis nana |
2 |
2 |
5 |
9 |
Ascaris lumbricoides |
|
5 |
15 |
20 |
Strogyloides stercoralis |
|
|
2 |
2 |
Bacterial pathogens |
|
|
|
|
Vibrio cholerae |
|
|
30 |
30 |
Shigella spp |
|
2 |
11 |
13 |
Mixed - > than one pathogen |
|
|
|
|
E histolytica/ E dispar, Giardia |
|
5 |
- |
5 |
E histolytica/ E dispar and Hymenolepis nana ova |
|
4 |
2 |
6 |
E histolytica/ E dispar, Giardia intestinalis and Vibrio cholerae |
|
|
2 |
2 |
TOTAL |
15 |
74 |
359 |
448 |
The bacterial pathogens isolated were Vibrio cholerae and Shigella spp. in the samples from the adult population. Thirty Vibrio cholerae isolates were obtained from the samples of the adults, Two samples from adult were positive for mixed infections i.e. Vibrio cholerae and parasitic infections (Entamoeba histolytica/ Entamoeba dispar and Giardia intestinalis respectively). All the Vibrio cholerae isolates belonged to serogroup O1, serotype Ogawa. There were a total of 13 samples (11 from adults and 2 from pediatric patients) that were positive for Shigella species and all the isolates were identified as Shigella flexneri. In addition to the parasites found in the stool samples, we obtained 3 worms in the samples of the patients. 2 were expelled from the oral cavity of the patients while one was obtained during exploratory laparotomy of the patient. All three worms were identified as Ascaris lumbricoides. Stool samples were also obtained from these patients which demonstrated the eggs of Ascaris lumbricoides.
On analysis of the drug susceptibility of the bacterial isolates, Vibrio cholerae were susceptible to Cefotaxime (70%), Imipenem (68%), Ciprofloxacin (50%), Amikacin (30%) whereas highly resistant to Nalidixic acid (93.33%) and Trimethoprimsulfamethoxazole (100%). Shigella isolates demonstrated a high percentage of resistance (100%) to Nalidixic acid while resistance to Trimethoprim-Sulfamethoxazole was demonstrated by 85% of the strains. The percentage resistance seen in the isolates against Ciprofloxacin, Ceftriaxone, and Gentamicin was 53.84%, 27.07%, and 7.69% respectively.
In addition to the parasites found in the stool samples, we obtained 3 worms in the samples of the patients. 2 were expelled from the oral cavity of the patients while one was obtained during exploratory laparotomy of the patient. All three worms were identified as Ascaris lumbricoides. Stool samples were also obtained from these patients which demonstrated the eggs of Ascaris lumbricoides.
Discussion and conclusion
Due to lack of education, poverty, low socioeconomic status, poor sanitation, and personal hygiene, the type of toilet and water facilities available there is a high incidence of recurrent intestinal infections in the developing countries. Stool examination for intestinal parasitic ova, cysts, trophozoite, and larvae remains the gold standard for the laboratory diagnosis of intestinal parasitic infections. A paucity of knowledge about the parasites prevalent in a particular geographic area may lead to the misdiagnosis of intestinal parasitic infections as inflammatory bowel diseases [2].
Human Intestinal helminthic infections pose a major public health problem in developing countries like India and Bangladesh.
Ascariasis (roundworm infestation) in a community is a reflection of the socioeconomic status, poor environmental conditions, lack of education and is a mirror of poverty [3]. Ancylostoma duodenale is a major cause of enteric blood loss and iron deficiency anemia in many tropical countries [7]. As per a World Health Organisation Report, 44 million pregnancies face complications because of Ancylostoma infections [8]. So it is important that Regular deworming programs should be adopted to check the surge of intestinal parasites and the subsequent co-morbidities associated with it.
The prevalence of parasitic infections in our study was seen to be 17.10%, which is higher than Kotian et al 11% [4] lower when we compared with the study from Dashwa et al [5] (30%). In India, a wide range (11-90%) of the prevalence of these intestinal parasites has been reported from different geographical provinces and populations [3]. In our study, the prevalence rate of Entamoeba histolytica/dispar is high in adult patients which is in concordance with a Chennai study by Dhanabal et al [6] (21.8%). In children, Giardia intestinalis was the most common pathogen isolated. It is transmitted by the feco oral route after drinking contaminated water as it is a common environmental contaminant of the water supply [4]. This observation is in concordance with other studies. Approximately 15% of children aged 0-24 months in the developing world have been reported to have giardia infection. Intestinal parasitic infections remain a significant health problem in a developing country like India, especially among children [9,10]. Recurrent intestinal infections can have a detrimental effect on their health and can lead to malnutrition in children [11].
Cholera, a severe public health problem in underdeveloped countries and poses grave consequences if not addressed immediately. Cholera is one of the diarrheal conditions which requires treatment with antibiotic medication, particularly tetracycline, in addition to maintaining hydration. An efficient antibiotic aids in the reduction of fluid loss as well as the duration, severity, transmission, morbidity, and death associated with the condition [12]. So, a timely diagnosis and appropriate treatment can significantly reduce the mortality rate associated with infectious diarrhoea. Apart from Vibrio cholerae, 13 samples were positive for Shigella flexneri. 2 in the paediatric age group and 11 in adults.
On analyzing the sensitivity patterns of Vibrio cholerae and Shigella flexneri, we observed a high degree of resistance to amoxicillin, cotrimoxazole and nalidixic acid. However, resistance to gentamicin, amikacin and third-generation cephalosporins was low. These findings are consistent with other studies. The drugs showing sensitivity should be in kept reserve and their use should be restricted especially in cases of diarrhea to prevent the development of resistance to these therapeutic agents [13].
Improving the quality of the water, sanitation, and overall hygiene can help prevent diarrhoea. The accurate diagnosis of the causative agents like bacteria and parasites and treatment based on the findings and regular monitoring of antimicrobial resistance profiles will help in preventing the spread of multidrug-resistant strains.
Acknowledgment: I would like to express special thanks and gratitude to All the patients who took part in the study
References
- Kulkarni SV, Kairon R, Sane SS, Padmawar PS, Kale VA, Thakar MR, et al. Opportunistic parasitic infections in HIV/AIDS patients presenting with diarrhoea by the level of immunesuppression. Indian Journal of Medical Research. 2009; 130(1): 63.
- Mathuria YP., & Singh, A. Prevalence of Intestinal Parasites at a Tertiary Care Centre at Moradabad, Western Uttar Pradesh, India: A One-Year Observational Study. Annals of International Medical and Dental Research. 2017; 3(5): 7.
- Choubisa SL, Jaroli VJ, Choubisa P, Mogra N. (2012). Intestinal parasitic infection in Bhil tribe of Rajasthan, India. Journal of parasitic diseases. 2012; 36(2): 143-148.
- Kotian S, Sharma M, Juyal D, Sharma N. Intestinal parasitic infection-intensity, prevalence and associated risk factors, a study in the general population from the Uttarakhand hills. International Journal of Medicine and public health. 2014; 4(4).
- Chowdhury S. (2017). Chakraborty P pratim. Universal health coverage-There is more to it than meets the eye. J Fam Med Prim Care. 2017; 6(2): 169-70.
- Dhanabal J, Selvadoss PP, Muthuswamy K. Comparative study of the prevalence of intestinal parasites in low socioeconomic areas from South Chennai, India. Journal of parasitology research. 2014.
- Pullan RL, Smith JL, Jasrasaria R, Brooker SJ. Global numbers of infection and disease burden of soil transmitted helminth infections in 2010. Parasites & vectors. 2014; 7(1): 1-19.
- Stephenson LS, Latham MC, Ottesen EA. (2000). Malnutrition and parasitic helminth infections. Parasitology. 2000; 121(S1): S23-S38.
- Brooker S, Hotez PJ, Bundy DA. Hookworm-related anaemia among pregnant women: a systematic review. PLoS neglected tropical diseases. 2008; 2(9): e291.
- Praharaj I, Sarkar R, Ajjampur SSR, Roy S, Kang G. Temporal trends of intestinal parasites in patients attending a tertiary care hospital in south India: A seven-year retrospective analysis. The Indian journal of medical research. 2017; 146(1): 111.
- Khurram Baqai FI, Laique N. Frequency and antimicrobial pattern resistance of vibrio cholera in stool samples.
- Chiyangi H, Muma JB, Malama S, Manyahi J, Abade A, Kwenda G, Matee MI. Identification and antimicrobial resistance patterns of bacterial enteropathogens from children aged 0–59 months at the University Teaching Hospital, Lusaka, Zambia: a prospective cross sectional study. BMC infectious diseases. 2017 ;17(1): 1-9.