Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Renal cell carcinoma, MiTF/TFE3 family translocation-associated subtype
Aaron Chow2; Nada Shaker1; Anil V Parwani1*
1 Department of Pathology, The Ohio State University Wexner Medical Center. Columbus, Ohio, USA.
2 Gulbransen Lab, Michigan State University College of Osteopathic Medicine, USA.
*Corresponding Author: Anil Parwani
Professor of Pathology and Biomedical Informatics, Vice-Chair of Anatomic Pathology, Director of
Pathology Informatics Director, Digital Pathology
Shared Resources, Principal Investigator, Cooperative Human Tissue Network (CHTN) Midwestern
Division Wexner Medical Center - Department of
Pathology, The Ohio State University, E409 Doan
Hall, 410 West 10th Ave, Columbus, OH, 43210, USA
Email: Anil.Parwani@osumc.edu
Received : Feb 02, 2022
Accepted : Feb 28, 2022
Published : Mar 07, 2022
Archived : www.jcimcr.org
Copyright : © Parwani A (2022).
Abstract
A 20-year old male presented with right flank pain and hematuria. Initial workup for nephrolithiasis by CT imaging revealed a right renal lower pole mass. Biopsy of the renal mass revealed “clusters of spindle cells and mature fat” with no malignant cells found. Subsequent MRI imaging confirmed the presence of the mass, with concern for an infiltrative process. Therefore, the patient underwent a robotic-assisted right nephrouretectomy that revealed a Micropthlamia Family of Transcription Factor (MiTF) translocation tumor involving the Xp11.2 TFE3 transcription factor, without lymph node involvement. The patient remains asymptomatic without evidence of abdominopelvic or intrathoracic metastatic disease or lymphadenopathy two years post nephrouretectomy
Keywords: renal cell carcinoma; 2016 who classification; MiTF; microphthalmia; TFE3; translocation; RCC subtyping; prognosis.
Citation: Chow A, Shaker N, Parwani A. Renal cell carcinoma, MiTF/TFE3 family translocation-associated subtype. J Clin Images Med Case Rep. 2022; 3(3): 1717.
Introduction
MiTF translocation tumors are a form of Renal Cell Carcinoma (RCC) that predominantly affect females and primarily manifests in the child to adolescent age group, despite the rare nature of RCC in childhood [1]. MiTF translocation RCC represents 20-75% of childhood RCC cases, while only accounting for 1-4% of adult RCC [2]. Despite typically presenting at a later tumor stage, pediatric onset of MiTF translocation RCC demonstrate a better prognosis than with adult onset of this disease [1,3-5]. This form of RCC has only been recently recognized as a distinct form of RCC in 2004 and incorporated into the World Health Organization classification as of 2016 [2,6-8]. MiTF translocation RCC arises due to the chromosomal translocation of MiTF family of transcription factors, leading to the overexpression of abnormal fusion proteins [2,7]. The two MiTF family transcription factors implicated in translocation RCC are TFE3 and TFEB, located on Xp11.2 and 6p21, respectively [2,9].
Here, we examine the clinical, histologic, immunohistochemical, and molecular features of MiTF translocation RCC. We report a case of TFE3 over expression variant of MiTF translocation RCC in a 20-year-old male who presented with flank pain and gross hematuria and treated by robotic-assisted nephroureterectomy without complications.
Case report
Patient history
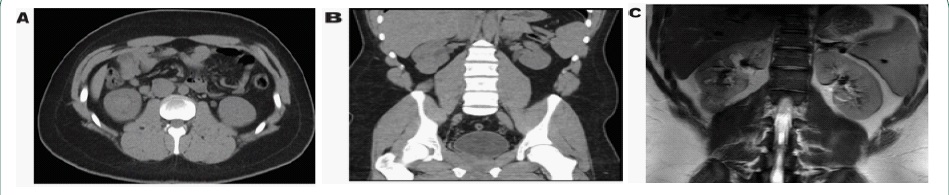
A 20-year-old male with a family history of metastatic renal cell carcinoma presents to his primary care physician with right flank pain and gross hematuria. CT workup for nephrolithiasis revealed a renal mass measuring up to 4.9 X 3.5 cm in the right lower pole (Figure 1A,B). Follow-up MRI confirms a 3.1 X 3.4 X 4.4 cm mass in the right lower pole that is concerning for malignancy (Figure 1C). CT-guided biopsy was performed which revealed clusters of spindle cells consisting of fibroblasts and smooth muscle cells with the presence of mature fat. Spindle cells were focally positive for smooth muscle actin, but negative for HMB-45 and MART-1. The patient underwent flexible cystoscopy which demonstrated no stones, lesions, or neoplasms in the bladder or ureters. Right retrograde pyeloureterogram showed no filling defects, strictures, or hydronephrosis. A large renal pelvis is visualized. The patient subsequently underwent right robotic-assisted nephroureterectomy and regional lymph node dissection.
Gross examination
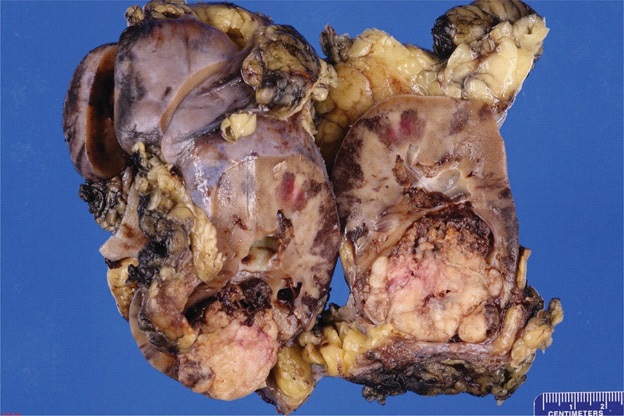
The renal specimen is designated “right kidney” and consists of a 13.2 X 7.8 X 6.3 cm radical nephrectomy specimen with a moderate amount of attached perinephric fat with a 2.3 cm in length and 0.4 cm in greatest diameter ureter, weighing 388.50 g in its entirety. A 0.8 cm in length and 0.3 cm in greatest diameter possible renal vein was identified which opened to reveal tan-pink, smooth vascular cut surfaces with no masses identified.
The specimen was bivalved through the hilum to reveal an 11.4 X 8.2 X 6.7 cm kidney with red-brown, unremarkable renal parenchyma with mildly dilated calyces. The corticomedullary junction was well-defined and measured 0.6 cm and 0.7 cm, respectively. Within the inferior pole, there was a 4.7 X 4.3 X 3.5 cm tan-white, firm, well-circumscribed, and partially (60%) necrotic mass that bulged into the perinephric fat. It remained confined to a possible capsule and remained 0.1 cm from the external surface. The mass bulged into the calyceal system but did not adhere to the urothelium. The scant amount of perinephric fat was also uninvolved. The mass remained greater than 3.0 cm from the ureter and hilar vascular margins. Indeterminate origin of mass from urothelium versus renal parenchyma. The ureter and renal pelvis were tan-pink and smooth with no masses identified (Figure 2).
Microscopic Examination
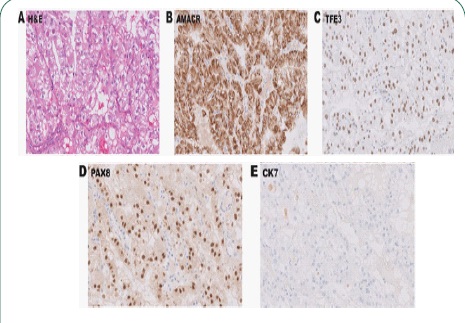
Specimen sections were initially stained via Hematoxylin and Eosin (H&E) (Figure 3A). Examination revealed renal parenchyma with mild chronic inflammation. Renal mass was well delineated from parenchyma and demonstrates a mixed papillary and solid nested growth with increased cytoplasmic clearing and prominent nucleoli. Coagulative necrosis is appreciated in about 60% of the mass. All examined lymph nodes were negative for malignancy
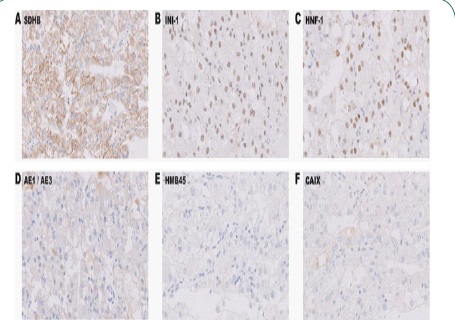
Immunohistochemistry (IHC) labeling revealed positive staining for Alpha-Methylacyl-Coa Racesmase (AMACR), and strong diffuse nuclear labeling for Transcription Factor Binding to IGHM Enhancer 3 (TFE3) and Paired Box 8 (PAX8) (Figure 3B-D). Tumor cells did not demonstrate any Cytokeratin 7 (CK7) labeling (Figure 3E). Additional stains for Succinate Dehydrogenase (SDHB), integrase interactor 1 (INI-1), and Hepatocyte Nuclear Factor 1β (HNF-1) all stained positively (Figure 4A-C). Cytokeratin AE1 / AE3 demonstrated weak, focal staining while Human Melanoma Black-45 (HMB-45) and Carbonic Anhydrase IX (CAIX) were negative (Figure 4D-F). IHC labeling is summarized in Table 1. Six months after surgery, renal function was roughly baseline and imaging studies showed no evidence of disease. The patient denied any symptoms. Eighteen months post-nephroureterectomy, the patient remains asymptomatic and presented with no signs of recurrence, progression, or metastasis.
Table 1: Summary of IHC labeling. IHC labels performed for this case are summarized as negative, focal positive, or positive.
Stain |
Result |
AE1/AE3 |
Weak, focal positive |
AMACR |
Positive |
CAIX |
Negative |
CK7 |
Negative |
HMB-45 |
Negative |
HNF-1 |
Positive |
INI-1 |
Positive |
PAX8 |
Positive |
SDH8 |
Positive |
TFE3 |
Positive |
Discussion
MiTF translocation RCC is a rare and distinct variant of nonclear cell carcinomas and are predominantly found in young patients. This form of RCC arises due to chromosomal translocation to create transcription factor fusion proteins which become over expressed. Translocation RCC is due to the over expression of MiTF family of transcription factors, consisting of TFE3, TFEB, TFC, and MiTF. The TFE3 transcription factor located on chromosomal region Xp11.2 is most commonly over expressed, due to forming one of 15 possible TFE3 fusion genes that have been previously described [2,10]. Description of new TFE3 translocations are being added to a growing list of TFE3 variants of MiTF translocation RCC, but majority of TFE3 gene fusion involves one of the ASPSCR1, PRCC, NONO, or SFPQ genes [11]. In addition to TFE3 transcription factor fusion protein over expression, the remainder of MiTF translocation RCC arise from theoverexpression of TFEB fusion proteins. These fusion proteins with TFEB occurs due to a t(6;11) translocation, as opposed to Xp11.2 translocation in TFE3-mediated translocation RCC [2]. TFEB translocation is less common compared to Xp11.2 TFE3 translocation and typically results in a less aggressive disease course [2].
Tumor development in MiTF translocation RCC often occurs unnoticed. It may present insidiously as a painless abdominal mass or painless hematuria and are generally detected incidentally upon abdominal imaging. As a result, patients may not present for evaluation until late in the clinical course at advanced tumor stages [12]. Gross examination of MiTF translocation RCC generally reveal a well circumscribed tan-yellow colored mass with or without a pseudo capsule, and are commonly necrotic and hemorrhagic [1].
Microscopically, MiTF translocation RCC can develop a mixed papillary, solid, and alveolar growth pattern. Furthermore, tumor cells are enlarged with voluminous cytoplasm, distinct cell borders, and have a clear or eosinophilic appearance. Hyalin nodules and psammoma bodies are seen in ASPSCR1-TFE3 carcinomas [13]. Due to their variable histological presentations, translocation RCC can resemble other forms of RCCs such as clear cell RCC, multilocular cystic RCC, urothelial carcinoma, collecting duct carcinoma, mucinous tubular carcinoma, or epithelioid angiomyolipoma [2,12]. While Fluorescent In Situ Hybridization (FISH) analysis is the current gold standard to identify Xp11.2 translocation [2,14], immunohistochemistry labeling for TFE3 can also be utilized as a marker for translocation RCC. Although TFE3 is normally constitutively expressed in tissue, it is expressed at a low level undetectable on IHC [12,15]. Therefore, TFE3 staining is observable via IHC only when overexpressed, conferring a strong nuclear stain. Translocation RCC also stains positively for PAX2, PAX8 and is generally positive for CD10, cathepsin-K and AMACR [13,16]. Vimentin, cytokeratin, and Epithelial Membrane Antigen (EMA) are typically under expressed, and TFEB (in TFE3 variant MiTF translocation RCC) and cytokeratin 7 stain negatively [12].
Data supports initial treatment of translocation RCCs with a VEGFR inhibitor such as sunitinib, and a potential therapeutic benefit in utilizing immune checkpoint inhibitors, but further study is warranted [17]. Alternatively, surgical intervention with partial nephrectomy is also an effective treatment [18].
Conclusions
In the present case, the patient experienced right flank pain and gross hematuria which prompted a workup for kidney stones, thereby incidentally revealing an intrarenal mass. Patient underwent robotic-assisted nephroureterectomy of the right kidney to remove the mass which was subsequently diagnosed as MiTF / TF3 family translocation RCC. Surgical removal resulted in resolution of patient’s symptoms without any signs of abdominopelvic metastatic disease or lymphadenopathy two years post nephrectomy.
While surgical resection is effective for removal of tumor mass, additional studies are warranted in the examination and development of targeted treatments to avoid nephrectomies that partially remove non-affected tissue.
Declarations
Conflict of interest: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Patient Consent: The authors attest that there is no patientidentifying information present in any of the figures or material presented.
Declarations of interest: None
Funding: This research did not receive any specific grant from funding agencies in the public, commercial, or not-forprofit sectors.
References
- Inamura K, Translocation Renal Cell Carcinoma: An Update on Clinicopathological and Molecular Features, Cancers (Basel). 2017; 9. https://doi.org/10.3390/CANCERS9090111.
- Caliò A, Segala D, Munari E, et al. MiT Family Translocation Renal Cell Carcinoma: From the Early Descriptions to the Current Knowledge, Cancers (Basel). 2019; 11. https://doi.org/10.3390/ CANCERS11081110.
- Craig KM, Poppas DP, Akhavan A, Pediatric renal cell carcinoma, Curr. Opin. Urol. 2019; 29: 500–504. https://doi.org/10.1097/ MOU.0000000000000656.
- Ma W, Liu N, Zhuang W, et al. Comparative Clinicopathologic Characteristics and Outcomes of Paediatric and Adult Xp11 Translocation Renal Cell Carcinomas: A Retrospective Multicentre Study in China, Sci. Rep. 2020; 10. https://doi.org/10.1038/ S41598-020-59162-5.
- Hodge JC, Pearce KE, Wang X, et al. Molecular cytogenetic analysis for TFE3 rearrangement in Xp11.2 renal cell carcinoma and alveolar soft part sarcoma: validation and clinical experience with 75 cases, Mod. Pathol. 2014; 27: 113–127. https://doi. org/10.1038/MODPATHOL.2013.83.
- Dijkhuizen T, van den Berg E, Wilbrink M, et al., Distinct Xp11.2 breakpoints in two renal cell carcinomas exhibiting X; autosome translocations, Genes. Chromosomes Cancer. 1995; 14: 43–50. https://doi.org/10.1002/GCC.2870140108.
- Argani P, MiT family translocation renal cell carcinoma, Semin. Diagn. Pathol. 2015; 32: 103–113. https://doi.org/10.1053/J. SEMDP.2015.02.003.
- Srigley JR, Delahunt B, Eble JN, et al. The International Society of Urological Pathology (ISUP) Vancouver Classification of Renal Neoplasia, Am. J. Surg. Pathol. 2013; 37: 1469–1489. https://doi.org/10.1097/PAS.0B013E318299F2D1.
- Gupta S, Argani P, Jungbluth AA, et al. TFEB Expression Profiling in Renal Cell Carcinomas: Clinicopathologic Correlations, Am. J. Surg. Pathol. 2019; 43: 1445–1461. https://doi.org/10.1097/ PAS.0000000000001307.
- Xie L, Zhang Y, Wu CL, Microphthalmia family of transcription factors associated renal cell carcinoma, Asian J. Urol. 2019; 6: 312–320. https://doi.org/10.1016/J.AJUR.2019.04.003.
- Green WM, Yonescu R, Morsberger L, et al. Utilization of a TFE3 break-apart FISH assay in a renal tumor consultation service, Am. J. Surg. Pathol. 2013; 37: 1150–1163. https://doi.org/10.1097/ PAS.0B013E31828A69AE.
- Armah HB, Parwani AV, Xp11.2 translocation renal cell carcinoma, Arch. Pathol. Lab. Med. 2010; 134: 124–129. https://doi. org/10.5858/2008-0391-RSR.1.
- Argani P, Zhong M, Reuter VE, et al. TFE3-Fusion Variant Analysis Defines Specific Clinicopathologic Associations Among Xp11 Translocation Cancers, Am. J. Surg. Pathol. 2016; 40: 723–737. https://doi.org/10.1097/PAS.0000000000000631.
- Rao Q, Williamson SR, Zhang S, et al. TFE3 break-apart FISH has a higher sensitivity for Xp11.2 translocation-associated renal cell carcinoma compared with TFE3 or cathepsin K immunohistochemical staining alone: Expanding the morphologic spectrum, Am. J. Surg. Pathol. 2013; 37: 804–815. https://doi.org/10.1097/ PAS.0B013E31827E17CB.
- Argani P, Lal P, Hutchinson B, et al. Aberrant nuclear immunoreactivity for TFE3 in neoplasms with TFE3 gene fusions: A sensitive and specific immunohistochemical assay, Am. J. Surg. Pathol. 2003; 27: 750–761. https://doi.org/10.1097/00000478- 200306000-00005.
- Komai Y, Fujiwara M, Fujii Y, et al. Adult Xp11 translocation renal cell carcinoma diagnosed by cytogenetics and immunohistochemistry, Clin. Cancer Res. 2009; 15: 1170–1176. https://doi. org/10.1158/1078-0432.CCR-08-1183.
- Boilève A, Carlo MI, Barthélémy P, et al. Immune checkpoint inhibitors in MITF family translocation renal cell carcinomas and genetic correlates of exceptional responders, J. Immunother. Cancer. 2018; 6. https://doi.org/10.1186/S40425-018-0482-Z.
- Gorin MA, Ball MW, Pierorazio PM, et al. Partial nephrectomy for the treatment of translocation renal cell carcinoma, Clin. Genitourin. Cancer. 2015; 13: e199–e201. https://doi.org/10.1016/J. CLGC.2014.12.008