Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Trastuzumab-related sarcoidosis in patient with breast adenocarcinoma: Case report and review of the literature
Oliveira PO1; Vitorino RC2, Da Cruz LA3, Silva RC4, Dos Santos-Junior EG5, Figueiredo RG6*
1 Departamento de Saúde, Universidade Estadual de Feira de Santana (UEFS), Feira de Santana, Bahia, Brazil.
2 Acadêmico de Medicina, Departamento de Saúde, Escola Bahiana de Medicina e Saúde Publica, Salvador, Bahia, Brazil.
3 Oncologista, Grupo Baiano de Oncologia (GBO), Feira de Santana, Brazil.
4 Membro, Colégio Brasileiro de Radiologia e Diagnóstico por Imagem, Salvador, Bahia, Brazil.
5 Professor de Cardiologia, Departamento de Saúde, Universidade Estadual de Feira de Santana (UEFS), Feira de Santana, Bahia, Brazil.
6 Professor de Pneumologia, Departamento de Saúde, Programa de Pós-Graduação em Saúde Coletiva (PPGSC-UEFS), Universidade Estadual de Feira de Santana (UEFS), Feira de Santana, Bahia, Brazil.
*Corresponding Author: Ricardo Gassmann
Figueiredo
Professor de Pneumologia, Departamento de Saúde,
Programa de Pós-Graduação em Saúde Coletiva
(PPGSC-UEFS), Universidade Estadual de Feira de
Santana (UEFS), Feira de Santana, Bahia, Brazil.
Email: rgfigueiredo@uefs.br
Received : Jan 06, 2022
Accepted : Mar 02, 2022
Published : Mar 09, 2022
Archived : www.jcimcr.org
Copyright : © Figueiredo RG (2022).
Abstract
Introduction: Sarcoidosis is a complex systemic granulomatous disease of unknown etiology, characterized by the formation of non-caseous granulomas. It has a biphasic age pattern, with higher incidence in young adults as well as in older people. Trastuzumab is a monoclonal antibody that inhibits the Human Epidermal Growth Factor Receptor (HER2) and there are few reports in the literature concerning sarcoidosis or sarcoidosis-like reactions induced by cancer immunotherapy.
Case report: We reported a peculiar association between sarcoidosis and trastuzumab immunotherapy in a 38-year-old woman with breast cancer. Multiple well-defined lung nodules in a perilymphatic distribution were seen in association with opacities that tended to merge to form nodular images with irregular outlines, the so-called galaxy sign, in the posterior part of the upper left lobe.
Conclusion: Herein, we also briefly review the role of neoplasic factors in sarcoidosis and immunopathological triggers to the development of granulomas in patients under trastuzumab therapy.
Keywords: sarcoidosis; breast cancer; neoplasis; monoclonal antibody; trastuzumab
Citation: Oliveira PO, Vitorino RC, Da Cruz LA, Silva RC, Figueiredo RG, et al. Trastuzumab-related sarcoidosis in patient with breast adenocarcinoma: Case report and review of the literature. J Clin Images Med Case Rep. 2022; 3(3): 1727.
Introduction
Sarcoidosis is a systemic granulomatous disease of unknown etiology, characterized by the formation of non-caseous granulomas, consisting of epithelioid cells and aggregated giant cells [1]. Although affecting all racial and ethnic groups, its prevalence is higher among those with black skin color and lower among Asians [2-4], as well as being predominant in women [1,5,6]. It has a biphasic age pattern, with higher occurrence between 20 and 40 years as well as greater than 60 [1,7,8]. Trastuzumab is a monoclonal antibody that inhibits the Human Epidermal Growth Factor Receptor (HER2), a receptor with overexpression in some subgroups of individuals with breast cancer and other solid tumors [9,10]. Its most common collateral effects are hypersensitivity reactions (less than 10% of individuals), which can be avoided with the use of antihistamines and corticosteroids. Myelosuppression, nausea and vomiting are rare [11]. In recent years, reports of cardiotoxicity have increased; however, there continue to be few reports in the literature concerning sarcoidosis or sarcoidosis-like reactions caused by Trastuzumab. Herein, we report a case of a trastuzumab-related sarcoidosis in a woman with breast adenocarcinoma.
Case report
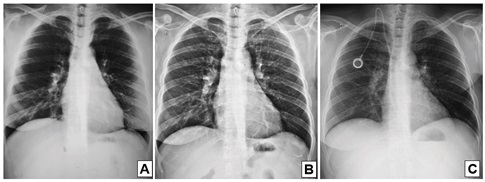
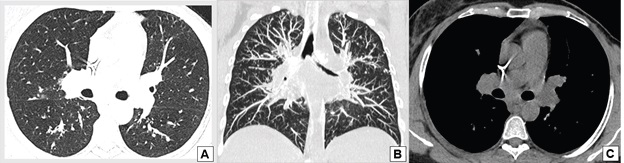
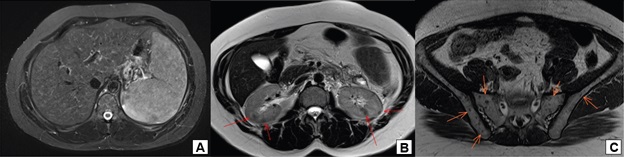
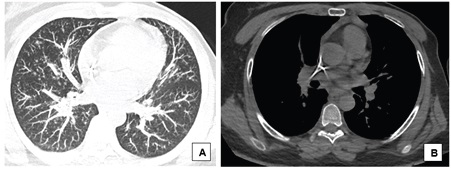
A 38-year-old woman with breast cancer had been using trastuzumab for 3 months when she presented painful hepatomegaly, 10 kg of weight loss and arthralgia in the knees, without clinical signs of uveitis. She was referred for a pneumological evaluation to check for lymph node enlargement and perilymphatic pulmonary nodules (Figure 1A). A chest x-ray conducted 12 months before the symptoms appeared did not show abnormalities (Figure 1B). The tuberculin test was less than 5 mm. A subsequent investigation with a chest computed tomography in June 2020 revealed multiple welldefined opacities in the axial, septal and subpleural interstice of both lungs, seen mainly in the upper lobes, and sparse small well-defined nodules with peribronchovascular distribution. Opacities that tended to merge to form nodular images with irregular outlines, the so-called galaxy sign, were observed in the posterior part of the upper left lobe (Figure 2). Magnetic resonance imaging of the abdomen in June 2020 showed hepatoesplenomegaly, multiple small indeterminate hepatic nodules, splenic nodules, renal nodules, and nodules in the pelvic bone (Figure 3). A magnetic resonance scan performed in January 2019 did not show any of these findings, except for focal nodular hyperplasia, which remained unchanged.
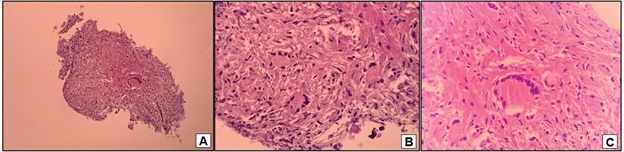
The correlation between radiological findings and the clinical status reinforced the suspicion of sarcoidosis, with a differential diagnosis of secondary metastatic lesions secondary to the base disease and tuberculosis. The patient underwent a bronchoscopy with transbronchial biopsies that showed a non-caseating chronic granulomatous inflammation (Figure 4). GROCOTT and Ziel-Neelsen stains were negative for infectious agents, as did a direct search for microbacteria and fungi. The anatomopathological findings favored the diagnosis of sarcoidosis. Due to technical limitations, measure serum ACE and soluble interleukin-2 receptor levels were not available. Given the symptoms and systemic involvement of multiple organs, we decided to interrupt the immunobiological treatment and administered prednisone, 30 mg/day (0,5 mg/ kg/day). Subsequent radiological control after 4 months showed a significant improvement with resolution of abdominal pain and reduced profusion of interstitial nodules and mediatinal and hilar lymph node enlargement (Figure 1C). The patient had undergone a gradual taper in prednisone dosage for six months to prevent sarcoidosis relapse with mild weight gain and cushingoid features.
Discussion
Sarcoidosis is a systemic granulomatous disease whose etiology is not completely understood. There is an association with exposure to unknown antigens that function as immunopathological triggers for the formation of granulomas. Exposure to bacteria, fungi, viruses, medication, and nanoparticles is potentially related to its pathogenesis; however, no causal nexus has been established yet [12,13]. Genetic factors may have a considerable influence in sarcoidosis. It has been shown that members of the same family have an increased risk of developing the disease, but the genetic loci involved have not been fully identified [13]. The involvement of TNF-alpha has been documented as being central to the immune response, which also involves lymphocytes, macrophages, and other antigen-presenting cells [14].
We reported an infrequent association between sarcoidosis and immunotherapy with Trastuzumab in patients with breast cancer. A well-established temporal nexus can be noted between the introduction of the drug and the development of symptoms. However, the causality is uncertain, given that cancer and other antigens are also mentioned in the literature as being potential triggers for sarcoidosis. After suspending the immunobiological treatment, it was decided that observation without the use of corticoids was not an option due to the extensive systemic symptoms present.
The role of neoplastic factors in the development of granulomas is controversial. Some cohort studies and case reports have shown an association between cancer and sarcoidosis during all phases of neoplasia. However, these associations are not fully understood, with majority of the reports describing the appearance of sarcoidosis in the active phase of neoplasia or after its treatment. Brincker and Wilbek were the first to show a relationship between cancer and sarcoidosis. From a group of 2544 patients with respiratory sarcoidosis, 48 developed a malignant cancer, when only 33.8 cases would be expected in the general population. The occurrence of malignant lymphoma was 11 times and lung cancer 3 times greater than expected. The authors concluded that a possible immunological dysfunction commonly encountered in patients with sarcoidosis may have influenced the considerable rate of malignant lymphomas found in the analysis [15]. Nonetheless, this study was later contested by Romers, who identified a misdiagnosis of sarcoidosis in 14 of the 48 cases reported [16]. Sarcoidosis after malignancy has been reported most often in cases of hematologic neoplasia [17]. Shigemitsu and collaborators first described the term “sarcoidosis cancer syndrome” to define sarcoidosislike reactions and multisystem sarcoidosis in patients with solid malignant neoplasia and lymphomas [18].
Among solid tumors, breast cancer has been highlighted in two small cohort studies that point to a possible causal relationship with sarcoidosis [19,20]. Patients with breast cancer showed initial signs of sarcoidosis at a much older age than general population, as well as having higher prevalence of sarcoidosis and sarcoidosis-like reactions. More robust prospective studies are needed to better understand this association. A recent literary review analyzed 104 individuals with breast neoplasia who were also diagnosed with sarcoidosis or sarcoidosis-like reaction. The breast cancer diagnosis preceded the identification of sarcoidosis in 22% of cases, with an average interval of 4.8 years. In approximately one-tenth of patients, the diseases occurred simultaneously. According to the authors, the long temporal hiatus between the appearance of the two conditions makes the hypothesis of a causal association a fragile one [21].
Trastuzumab was interpreted as being the probable agent related to the sarcoidosis in this individual due to the temporal nexus between its introduction and the development of the disease. It is worth noting that radiological studies of the chest and abdomen carried out 8 months prior to the clinical signs did not show alterations compatible with granulomatous disease. The patient also did not report any change in their basic health status before beginning the immunobiological treatment. The rapid clinical and radiological response to treatment can be attributed to the use of corticoids and the suspension of the immunobiological treatment.
The connection between the epidermal growth factor and HER receptors activates signaling pathways that provoke cell division, death, motility, and adhesion responses. Cancer causes a deregulation of these pathways and trastuzumab blocks this receptor, provoking a reduction in HER2 expression levels in a dosedependent way, as well as inducing an immune response in the cells with HER2 overexpression and opsonization induced by monoclonal antibodies [22,23].
The cardiotoxicity of trastuzumab is broadly confirmed by the existing literature; however, reports concerning granulomatous reactions provoked by this agent are scarce. Cardiac toxicity is thought to be associated with oxidative stress induced by the inhibition of cardiomyocyte intracellular mechanisms, because of reduced HER2 activity [24].
The development of granulomas after the introduction of immunotherapeutic treatments has been described and should be considered when suspected. The so-called drug induced sarcoidosis reaction (DISR) and sarcoidosis have been principally associated with anti-TNFs, immune checkpoint inhibitors and interferons[25]. Some cohort studies identified a granulomatous reaction in individuals with melanoma and rheumatic diseases undergoing treatment with the abovementioned therapeutic groups [26-28].
An analysis of the WHO pharmacovigilance database (WHO) (VigiBase) identified 2,425 cases of druginduced sarcoidosis-like reactions. The main therapies involved in this reaction included interferon or peginterferon, TNF-alpha antagonists, and checkpoint immunological inhibitors, 55 drugs in total. In 62.1% (1,505 cases), the drug was the only possible causal factor for sarcoidosis, and the temporal relationship was consistent with medication-induced sarcoidosis [29]. Despite numerous reports of sarcoidosis and DISR following medical therapies, the immunopathological triggers involved have not, as yet, been characterized. As in this case report, other drugs have been described in isolated cases of sarcoidosis or sarcoidosis-like reactions, generally based on a causal relationship based on the short period between the appearance of granulomas and the introduction of the drugs, followed by a partial or complete improvement upon suspension of the exposure[30].
Conclusion
Some authors have recommended that DISR and sarcoidosis should be suspected in patients using monoclonal antibodies and who develop symptoms suggestive of the disease. It is therefore prudent to consider the potential role of immunobiological treatments in clinical findings and radiological characteristics suggestive of granulomas in individuals undergoing treatment with trastuzumab.
Declarations
Conflict of interest: We declare that there is no financial interest or any conflict of interest.
Author’s contributions
Paloma França de Oliveira GROUP 1: Conception of the work AND acquisition of data. GROUP 2: Drafting the work AND revising the work critically for important intellectual content. GROUP 3: Final approval of the version to be published. GROUP 4: Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Rafael Campos Vitorino GROUP 1: Conception of the work AND acquisition of data. GROUP 2: Drafting the work AND revising the work critically for important intellectual content. GROUP 3: Final approval of the version to be published. GROUP 4: Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Livia Almeida da Cruz GROUP 1: Acquisition of data AND clinical management of the patient. GROUP 3: Final approval of the version to be published. GROUP 4: Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Rubia Mara Correia Campos Silva GROUP 1: Interpretation AND analysis of medical imaging GROUP 2: Drafting the work AND revising the work critically for important intellectual content GROUP 3: Final approval of the version to be published. GROUP 4: Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Edval Gomes dos Santos Junior GROUP 2: Drafting the work AND revising the work critically for important intellectual content. GROUP 3: Final approval of the version to be published. GROUP 4: Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved
Ricardo Gassmann Figueiredo GROUP 1: Conception of the work AND design of the work. GROUP 2: Drafting the work AND revising the work critically for important intellectual content GROUP 3: Final approval of the version to be published. GROUP 4: Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Salah S, Abad S, Brézin AP, et al. Sarcoïdose [Sarcoidosis]. J Fr Ophtalmol. 2019; 42: 303-321. doi:10.1016/j.jfo.2018.06.015. PMID:30850198.
- Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007; 357: 2153-65. doi:10.1056/NEJMra071714. PMID:18032765.
- Ungprasert P, Crowson CS, Matteson EL. Influence of Gender on Epidemiology and Clinical Manifestations of Sarcoidosis: A Population-Based Retrospective Cohort Study 1976-2013. Lung, 2017; 195: 87-91. doi:10.1007/s00408-016-9952-6. PMID:27709291
- Ungprasert P, Crowson CS, Matteson EL. Epidemiology and clinical characteristics of sarcoidosis: an update from a population-based cohort study from Olmsted County, Minnesota. Reumatismo. 2017; 69: 16-22. doi:10.4081/reumatismo.2017.965. PMID:28535617
- Ungprasert P, Carmona EM, Utz JP, et al. Epidemiology of Sarcoidosis 1946-2013: A Population-Based Study. Mayo Clin Proc. 2016; 91: 183-8. doi:10.1016/j.mayocp.2015.10.024. PMID: 26727158
- Costabel U, Wessendorf TE, Bonella F. Epidemiologie und klinisches Erscheinungsbild der Sarkoidose [Epidemiology and Clinical Presentation of Sarcoidosis]. Klin Monbl Augenheilkd. 2017; 234: 790-795. doi:10.1055/s-0042-105569. PMID: 27454307.
- Gerke AK, Judson MA, Cozier YC, et al. Disease Burden and Variability in Sarcoidosis. Ann Am Thorac Soc. 2017; 14: S421-S428. doi: 10.1513/AnnalsATS.201707-564OT. PMID: 29087725
- Arkema EV, Grunewald J, Kullberg S, et al. Sarcoidosis incidence and prevalence: a nationwide register-based assessment in Sweden. Eur Respir J, 2016; 48:1690-1699.doi:10.1183/13993003.00477- 2016.PMID:27471207.
- Sarosiek T, Morawski P. Trastuzumab and its biosimilars [Trastuzumab and its biosimilars]. Pol Merkur Lekarski. 2018; 44:253-257.PMID:29813044.
- Oh DY, Bang YJ. HER2-targeted therapies - a role beyond breast cancer. Nat Rev Clin Oncol. 2020; 17:33-48.doi:10.1038/s41571- 019-0268-3.PMID:31548601.
- Hudis CA. Trastuzumab--mechanism of action and use in clinical practice. N Engl J Med. 2007; 357:39-51.doi:10.1056/ NEJMra043186.PMID:17611206.
- El Jammal T, Jamilloux Y, Gerfaud-Valentin M, et al. Refractory Sarcoidosis: A Review. Ther Clin Risk Manag. 2020; 16:323-345. doi:10.2147/TCRM.S192922.PMID:32368072
- Smith L. Sarcoidosis. Workplace Health Saf, 2015; 63:372. doi:10.1177/2165079915592748.PMID:26240121.
- Yee AM. Sarcoidosis: Rheumatology perspective. Best Pract Res Clin Rheumatol. 2016; 30:334-356. doi: 10.1016/j. berh.2016.07.001.PMID:27886804.
- Brincker H, Wilbek E. A incidência de tumores malignos em pacientes com sarcoidose respiratória. Br J Cancer. 1974; 29:247- 51.doi:10.1038/bjc.1974.64.PMID:4830144
- Pandha HS, Griffiths H, Waxman J. Sarcoidosis and cancer. Clin Oncol (R Coll Radiol), 1995; 7:277-8.doi: 10.1016/s0936- 6555(05)80533-3.PMID:8580051.
- Blank N, Lorenz HM, Ho AD, et al. Sarcoidosis and the occurrence of malignant diseases. Rheumatol Int. 2014; 34:1433-9. doi: 10.1007/s00296-014-2983-5.PMID:24658811.
- Shigemitsu H. Is sarcoidosis frequent in patients with cancer? Curr Opin Pulm Med. 2008; 14:478-80.doi:10.1097/ MCP.0b013e328305bf7d.PMID:18664979.
- Schweitzer MD, Salamo O, Holt G, et al. Sarcoidosis onset after breast cancer; a potential association. Eur J Intern Med. 2017; 44:11-12.doi: 10.1016/j.ejim.2017.08.006.PMID:28797536
- Arish N, Kuint R, Sapir E, et al. Characteristics of Sarcoidosis in Patients with Previous Malignancy: Causality or Coincidence? Respiration. 2017; 93:247-252.doi:10.1159/000455877. PMID:28231584.
- Chen J, Carter R 3rd, Maoz D, et al. Breast Cancer and Sarcoidosis: Case Series and Review of the Literature. Breast Care (Basel). 2015; 10:137-40.doi:10.1159/000381324.PMID:26195943
- Yarden Y, Sliwkowski MX. Untangling the ErbB signalling network. Nat Rev Mol Cell Biol. 2001; 2:127-37.doi:10.1038/35052073. PMID:11252954
- Baselga J, Albanell J, Molina MA, et al. Mechanism of action of trastuzumab and scientific update. Semin Oncol. 2001; 28:4-11. doi:10.1016/s0093-7754(01)90276-3.PMID:11706390
- Nicolazzi MA, Carnicelli A, Fuorlo M, et al. Anthracycline and trastuzumab-induced cardiotoxicity in breast cancer. Eur Rev Med Pharmacol Sci. 2018; 22:2175-2185.doi: 10.26355/ eurrev_201804_14752.PMID:29687878
- Chopra A, Nautiyal A, Kalkanis A, et al. Drug-Induced SarcoidosisLike Reactions. Chest. 2018; 154:664-677.doi:10.1016/j. chest.2018.03.056.PMID:29698718
- Chorti E, Kanaki T, Zimmer L, et al. Drug-induced sarcoidosis-like reaction in adjuvant immunotherapy: Increased rate and mimicker of metastasis. Eur J Cancer. 2020; 131:18-26.doi:10.1016/j. ejca.2020.02.024.PMID:32248071
- Lomax AJ, McGuire HM, McNeil C, et al. Immunotherapyinduced sarcoidosis in patients with melanoma treated with PD-1 checkpoint inhibitors: Case series and immunophenotypic analysis. Int J Rheum Dis. 2017; 20:1277-1285.doi:10.1111/1756- 185X.13076.PMID:28480561
- Cohen Aubart F, Lhote R, Amoura A, et al. Drug-induced sarcoidosis: an overview of the WHO pharmacovigilance database. J Intern Med. 2020; 288:356-362.doi:10.1111/joim.12991. PMID:31612562.
- Sanderson E, Wimaleswaran H, Senko C, et al. Durvalumab induced sarcoid-like pulmonary lymphadenopathy. Respirol Case Rep. 2020; 8:e00542.doi:10.1002/rcr2.542.PMID:32110415
- Rubio-Rivas M, Moreira C, Marcoval J. Sarcoidosis related to checkpoint and BRAF/MEK inhibitors in melanoma. Autoimmun Ver. 2020; 19:102587.doi:10.1016/j.autrev.2020.102587. PMID:32553612.