Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Acute motor weakness of lower extremity after a novel balloon-inflatable catheter for percutaneous epidural adhesiolysis and decompression: A case report
Chun Woo Yang; Na Eun Kim; Hye Rim Kwon; Sunhee Lee; Ji Sung Park; Byung Gun Kim*
Department of Anesthesiology and Pain Medicine, Inha University Hospital, Inha university School of Medicine, Inhang-ro 27, Jung-gu, Incheon, 22332, Korea
*Corresponding Author: Byung-Gun Kim
Department of Anesthesiology and Pain Medicine,
Inha University School of Medicine, Inha University
Hospital, Sinheung-dong 3-ga, Jung-gu, Incheon,
South Korea, 400-711
Email: mirrorcastle@naver.com
Received : Feb 05, 2022
Accepted : Mar 04, 2022
Published : Mar 11, 2022
Archived : www.jcimcr.org
Copyright : © Kim BG (2022).
Abstract
Background: As our population ages and the rate of spine surgery continues to rise, the use of epidural lysis of adhesions has emerged as a popular treatment for spinal stenosis and disc herniation. Percutaneous Epidural Adhesiolysis (PEA) causes pain by preventing the release of increased spinal nerve movement and nerve sensitivity due to nerve compression. PEA and decompression with a newly developed inflatable balloon catheter have been widely used to treat radicular pain caused by spinal stenosis or a herniated intervertebral disc.
Case presentation: A 64-year-old female patient who had lower back pain and right buttock pain radiating to the leg caused by an L4- L5 severe spinal stenosis underwent PEA and decompression using a Zigzag-motion Inflatable Neuroplasty (ZiNeu®, JUVENUI, Seoul, Korea) catheter. After the procedure, the patient complained of acute motor weakness in the right leg. After conservative treatment, motor weakness and paresthesia gradually improved over time. At the follow-up visit after 3 weeks, there were no signs of motor or sensory abnormalities.
Conclusions: Practitioners should be aware that these complications can occur at any time, and a skilled practitioner should undertake the procedure.
Keywords: cute motor weakness; percutaneous epidural adhesiolysis and decompression; spinal stenosis.
Abbreviations: HIVD: Herniated Intervertebral Disc; MRI: Magnetic Resonance Imaging; NRS: Numeric Rating Scale; ODI: Oswestry Disability Index; PEA: Percutaneous Epidural Adhesiolysis.
Citation: Yang CW, Kim NE, Kwon HR, Lee S, Kim BG, et al. Acute motor weakness of lower extremity after a novel ballooninflatable catheter for percutaneous epidural adhesiolysis and decompression: A case report. J Clin Images Med Case Rep. 2022; 3(3): 1731.
Background
PEA and decompression with a newly developed inflatable balloon catheter are being increasingly performed to treat radicular pain caused by spinal stenosis, Herniated Intervertebral Disc (HIVD), or failed back surgery [1-3]. Although PEA is known to be an effective method, it has the potential risks of complications. Complications of PEA include infection, urinary and fecal incontinence, sexual dysfunction, paresthesia, nerve injury, epidural abscess, and meningitis [1-3]. Here, we report a case of a patient who suffered from monoplegia of the rightside lower extremity following PEA and decompression with a newly developed inflatableballoon catheter on the right side and recovered after conservative treatment.
Case report
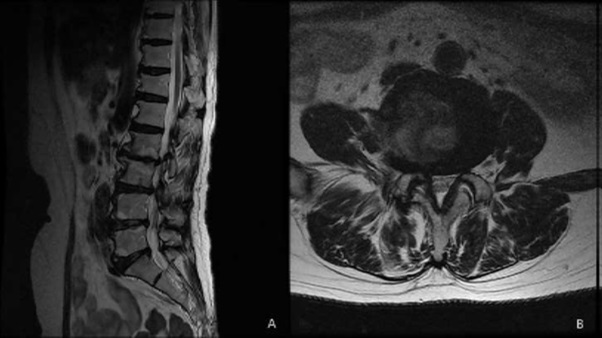
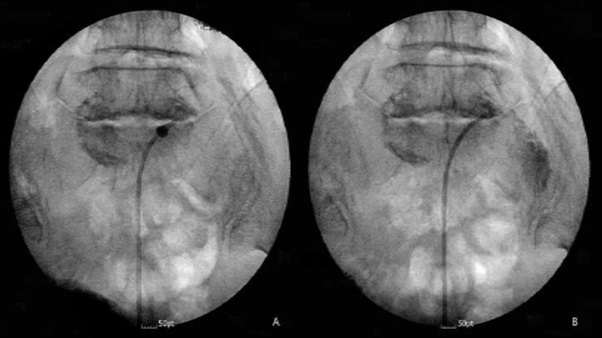
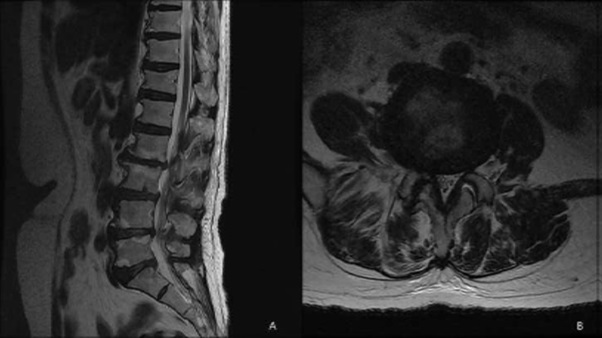
A 64-year-old patient visited the outpatient clinic for lower back pain and right buttock pain radiating to the leg 2 months prior. The patient’s pain score was 7/10 on the numeric rating scale (NRS; 0 = no pain, 10 = worst pain imaginable), and her Oswestry Disability Index (ODI; ranging from 0-100; 0 = no disability) score was 58. There were no sensory-motor abnormalities, and reflexes showed normal responses. Lumbar Spine Magnetic Resonance Imaging (MRI) revealed central stenosis at the L3-4 and L4-5 levels due to a bulging disc, facet arthrosis, and thickening of the ligamentum flavum (Figure 1). The patient was treated with nonsteroidal anti-inflammatory drugs, physical therapy, and five sets of interlaminar epidural steroid injections. However, there were no changes in her symptoms. The patient mainly complained of pain in her right leg. Lumbar decompression surgery was suggested by orthopedic surgeons as the patient complained of consistent and severe pain. The MRI findings showed moderate-to-severe spinal stenosis, and there was no response to conservative treatment. However, the patient refused surgery and wanted interventional treatment. Therefore, we decided to treat her with PEA and decompression with a newly developed inflatable balloon to reduce pain and extend the duration of pain relief. The patient was transferred to the operating room after informed consent was obtained. Prophylactic antibiotics (ceftezole, 1 g) were administered intravenously after negative skin test confirmation. Routine monitoring was done using electrocardiogram, blood pressure monitor, and pulse oximeter, and the patient was placed in a prone position with a pillow under her abdomen to reduce lumbar lordosis, the legs were restrained, and the feet were inverted. Following sterile preparation and draping, local infiltration was undertaken with a 1% lidocaine in the sacral hiatus. After sterile preparation for the procedure, a 10 G guide needle, specially designed to prevent cutting and skiving of the catheter, was inserted into the epidural space through the sacral hiatus. The epidural space was identified by injection of contrast medium (Omnipaque, Nycomed Imaging AS, Oslo, Norway) under fluoroscopy. A caudal epidurogram showed a filling defect from the central epidural space at the L3-4 levels in both L5 intervertebral foramina. After expanding the entrance with a guide wire and dilator, we inserted the ASAN-ZINEU balloon catheter, and the stylet was removed. We performed mechanical adhesiolysis and decompression by moving the catheter side-to-side and back and forth and removed the adhesions more widely using a ballooning catheter. The balloon was filled with 0.13 ml of contrast agent, and each ballooning process was limited to 5 seconds (Figure 2A). The extent of balloon inflation was adjusted by the degree of pain; if moderate-to-severe pain was noted during balloon inflation, no further treatment attempts were made for safety reasons. The catheter was moved only in the deflated state. After PEA and decompression, the contrast agent in the anterior epidural space spread upward above the level of L5-S1, suggesting that successful adhesiolysis had been achieved (Figure 2B). Before removal of the catheter, 1,500 units of hyaluronidase in 10 ml of normal saline was injected. Additional dye (2 ml) was injected to confirm the contours of the nerve root, and 10 ml of a mixture of 0.1% ropivacaine and 20 mg triamcinolone was administered via the catheter. There were no complications during the procedure, such as bleeding or damage to the dura. The patient did not complain of severe pain, paresthesia, or unusual responses during the procedure. The needle was removed, and a sterile dressing was applied. After 2 h, the patient complained of sudden motor weakness in the right lower limb. The great toe dorsiflexion was marked as motor grade I/V, and ankle dorsiflexion was marked as motor grade II/V. Paresthesia and a dull sensation in the right L4-L5 dermatome developed, but the patient did not complain of pain. At first, we suspected temporary motor weakness caused by ropivacaine, and the symptoms were observed for 1h. However, the patient’s symptoms and signs did not change. An urgent MRI was performed, but no epidural hematoma or suspicious lesions were observed (Figure 3). We hypothesized that edema in the right L5 root was caused by mechanical adhesiolysis and decompression. After consulting with the orthopedic surgeons and neurosurgeons, 1000 mg of methylprednisolone was injected intravenously daily for 3 days, but this injection was discontinued due to adverse effects. Pain was managed with 150 mg pregabalin twice daily (Lyrica, Pfizer, Groton, CT), 75 mg extended-release tramadol HCl, and 650 mg acetaminophen fixed-combination tablets. During the 10-day follow-up period, motor weakness and paresthesia improved gradually over time, and the patient was discharged and followed up. At the follow-up visit after 3 weeks, there were no signs of motor or sensory abnormalities. On follow-up monitoring after 1 month, the patient’s pain had been reduced from an NRS of 7 to 4 and the ODI score decreased to 38.
Discussion
Most patients suffering from low back pain generally have pain that is refractory to most commonly used treatment modalities, such as pharmacologic modalities, physical therapy, and epidural steroid injections. PEA has become increasingly common as a treatment choice for the management of chronic resistant spinal pain [4]. It is effective in removing fibrous tissue in the epidural space for various reasons. PEA is commonly performed with a Racz catheter or a more steerable navigation catheter (Navi Cath®, Myelotec, Inc., Roswell, GA, USA) and has proven effective [5,6]. However, the approach and correct placement of these catheters can be difficult in patients with severe adhesions or stenosis, leading to incomplete removal of the adhesions [7]. Moreover, the long-term effects (i.e., over more than 6 months) of this treatment are uncertain and controversial [8]. Importantly, no treatment has been developed to relieve stenosis itself through a nonsurgical method. Birkenmaier et al. reported that transforaminal balloon treatment resulted in significant pain relief and functional improvement in patients with chronic refractory lumbar foraminal stenosis [9]. Based on the study by Birkenmaier et al., a novel balloon catheter for more effective PEA and decompression was developed: The Zigzag motion Inflatable Neuroplasty catheter, which can be adjusted side-to-side and has an inflatable balloon attached to the end of the catheter tip [9]. However, this procedure may be associated with various complications. These complications could be due to not only the procedure itself but also the status of patients undergoing the procedure. The most common complications of PEA are dural puncture, spinal cord compression, catheter shearing, infection, unintended subarachnoid, subdural puncture, and unintended injection of injected solutions during the procedure [10]. Bleeding, infection, or nerve damage can generally develop after epidural neuroplasty, but motor and sensory weaknesses are rare complications [10]. Similar to our case, there are several reasons for unilateral motor weakness after PEA [10,11]. First, motor weakness can be caused by direct nerve injury from the catheter during the procedure. However, there were no complaints of severe pain during the procedure. Second, when the patient complained of right leg motor weakness, we considered the possibility of an epidural hematoma. However, the patient’s blood test results were normal, and MRI findings showed no evidence of hematoma. Third, it can be caused by intrathecal or subdural injections of local anesthetics. However, symptoms were observed for 2 hours, and the symptoms and signs did not change. Fourth, although nerve damage from hypertonics can be a reason, we did not use hypertonic saline [12]. Finally, injection of a large volume of fluid into the epidural space may cause a transient neurological deficit [12- 14]. Rocco et al [12], reported that epidural injections after spinal surgery may cause complications, such as cauda equina syndrome or nerve root damage, because the epidural space after surgery is limited, and the injected drugs may increase the pressure and cause nerve damage either directly or by ischemia, due to decreased blood flow. We think that, especially for older patients (>60 years) or for patients with poor epidural hygiene or lower volume of distribution in the epidural space, it is recommended to inject a smaller amount slowly, considering the summation effect of the injections and pressure effect of volume. Ho et al [13], have reported a case that presented with acute right monoplegia that had occurred after epidural adhesiolysis of the bilateral L5 nerve root in a patient with failed back surgery syndrome, with spontaneous recovery after 5 weeks. They suggested that the large volume of fluid injection might have been the reason for the transient neurological deficit, as a compartment loculated by the fluid might have compressed the nerve. They also suggested careful injection under the observation of the excretion of the contrast dye through the neural foramen to prevent nerve injuries caused by compression from a large volume of injected drugs. In particular, if the spinal stenosis is severe, an epidural injection may increase the pressure in the epidural space, and even mild edema around the nerve root may cause a neurologic deficit by compression of the nerve root [14]. We assume that the large volume injected during the procedure and the severe spinal stenosis could have caused the motor weakness in this case, as in the previously reported case [14]. Moreover, injecting a large volume during neuroplasty might have led to swelling of the L5 nerve root. However, further evaluation is required to confirm our hypothesis. Methylprednisolone has been reported to be effective in excessive swelling of nerve roots, helping with ambulation and overcoming functional limitations [15].
Conclusion
We presented a rare case of unilateral motor weakness following with PEA and decompression with a newly developed inflatable balloon. The injection of a large amount of fluid during procedure in a patient with spinal stenosis may cause compressive nerve damage. We should be aware that these complications can occur at any time, and a skilled practitioner should undertake he procedure.
Declarations
Acknowledgements: The authors wish to thank ChunWoo Yang, M.D., for her contributions in drafting the manuscript and revising it for important intellectual content
Availability of data and materials: The authors agree to make the raw data and materials described in our manuscript freely available.
Author contributions: Conceptualization: Byung-Gun Kim Writing – original draft: Chunwoo Yang, Byung-Gun Kim. Writing – review & editing: Na Eun Kim,Hye Rim Kwon, Sunhee Lee, Ji Sung Park.
Declaration of conflicting interest: The authors declare that there is no conflict of interest.
Ethics approval and consent to participate: Not applicable.
Funding: This work was supported by Inha University Research Grant (number 57848-01). Since this is a case report, institutional review board approval is not sought; however, written consent for publication was obtained from the patient.
Consent for publication: Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor in-Chief of this journal.
References
- Kim TH, Shin JJ, Lee WY. Surgical treatment of a broken neuroplasty catheter in the epiduralspace: A case report. J Med Case Rep. 2016; 10: 277.
- Anderson SR, Racz GB, Heavner J. Evolution of epidural lysis of adhesions. Pain Physician. 2000; 3: 262-270.
- Kim SH, Choi SS. Epidural neuroplasty/epidural adhesiolysis. Anesth Pain Med. 2016; 11: 14-22.
- Lim YS, Jung KT, Park CH, Wee SW, Sin SS, Kim J. Acute motor weakness of opposite lower extremity after percutaneous epidural neuroplasty. Korean J Pain. 2015; 28: 144-147.
- Manchikanti L, Singh V, Bakhit CE, Fellows B. Interventional techniques in the management of chronic pain: Part 1.0. Pain Physician. 2000; 3: 7-42.
- Lee JH, Lee SH. Clinical effectiveness of percutaneous adhesiolysis using Navicath for the management of chronic pain due to lumbosacral disc herniation. Pain Physician. 2012; 15: 213-221.
- Helm Ii S, Benyamin RM, Chopra P, Deer TR, Justiz R, et al. Percutaneous adhesiolysis in the management of chronic low back pain in post lumbar surgery syndrome and spinal stenosis: A systematic review. Pain Physician. 2012; 15: E435-62.
- Manchikanti L, Bakhit CE. Percutaneous lysis of epidural adhesions. Pain Physician. 2000; 3: 46-64.
- Birkenmaier C, Baumert S, Schroeder C, Jansson V, Wegener B, et al. A biomechanical evaluation of the epidural neurolysis procedure. Pain Physician. 2012; 15: E89-97.
- Bromage PR. Subarachnoid catheters and the cauda equina syndrome: Hypotheses in need of trial. Anesthesiology. 1994; 80: 711-712.
- Lee F, Jamison DE, Hurley RW, Cohen SP. Epidural lysis of adhesions. Korean J Pain. 2014; 27: 3-15.
- Rocco AG, Philip JH, Boas RA, Scott D. Epidural space as a Starling resistor and elevation of inflow resistance in a diseased epidural space. Reg Anesth. 1997; 22:167-177.
- Ho KY, Manghnani P. Acute monoplegia after lysis of epidural adhesions: A case report. PainPract. 2008; 8: 404-407.
- Chaudhari LS, Kop BR, Dhruva AJ. Paraplegia and epidural analgesia. Anaesthesia. 1978; 33: 722-725.
- Feng Y, Cai F, Chen L, Gu Y, Geng DC, Yang HL, et al. Excessive swelling of nerve roots: Important factor for recurring sciatica after lumbar surgery. Orthopade. 2020; 49: 502-509.