Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Could the association of sunitinib and stereotactic ablative radiotherapy lead to abscopal effect in metastatic renal cell carcinoma?
Chiara L Deantoni1*; Simone Baroni1; Silvia Foti2; Andrei Fodor1; Gianluca Del Conte2; Nadia G Di Muzio1,3
1 Department of Radiation Oncology, IRCCS San Raffaele Scientific Institute, Milan, Italy.
2 Department Unit of Oncology, Medical Oncology Department, IRCCS-Ospedale San Raffaele; Università Vita-Salute, Milano, Italy.
3 Vita-Salute San Raffaele University, Milan, Italy.
*Corresponding Author: Chiara L Deantoni
Department of Radiation Oncology, IRCCS San Raffaele Scientific Institute, Milan, Italy.
Email: deantoni.chiaralucrezia@hsr.it
Received : Mar 04, 2022
Accepted : Mar 28, 2022
Published : Apr 04, 2022
Archived : www.jcimcr.org
Copyright : © Deantoni CL (2022).
Abstract
Simultaneous prescription of systemic targeted therapy and Stereotactic Ablative Radiotherapy (SABR) is being used increasingly in various histological types of oligometastatic/oligoprogressive tumors. The underlying mechanisms of the synergistic effect are not yet well known. This case report concerns the interaction between a tyrosine kinase inhibitor and SABR in a patient affected by oligoprogressive metastatic renal cell carcinoma.
Keywords: Radiotherapy; Genitourinary oncology; Thoracic oncology.
Citation: Deantoni CL, Baroni S, Foti S, Fodor A, Del Conte G, et al. Could the association of sunitinib and stereotactic ablative radiotherapy lead to abscopal effect in metastatic renal cell carcinoma?. J Clin Images Med Case Rep. 2022; 3(4): 1774.
Introduction
The interaction of systemic targeted therapy and Radiation Therapy (RT) is currently the subject of great interest, due to the fact that their combination may lead to a significant immunestimulatory effect, boosting the natural anti-tumor immune response. A possible consequence is the Abscopal Effect (AE), a rare phenomenon but with high therapeutic potential for tumor regression in synchronous metastases not treated with RT.
The biological mechanisms underlying this effect are still unclear. The most accredited hypothesis is that RT determines immunogenic cell death, causing cryptic exposure of tumor antigens, tumor-antigen presentation and tumor microenvironment alteration. All these effects promote a new awakening of immune surveillance.
Unlike conventional fractionated radiotherapy, SABR induces direct tumor vascular-endothelial damage that may synergize with tumor targeted therapy [1,2]. The rationale in combining SABR with targeted agents could be to improve the therapeutic ratio, increasing tumor cell killing with a stable or minimal increase in toxicity.
Concerning the afore mentioned association, a field of interest is represented by the oligoprogressive disease during targeted therapy. In fact it is common to observe isolated disease progression in a few sites, in a scenario of stable disease, because of inter- and intra-tumoral mutational heterogeneity. In this setting, the main aim of SABR is to prolong the efficacy of the current target therapy, to delay the switch to other systemic therapies and to improve patient outcome, modifying the natural history of the disease.
Renal Cell Carcinoma (RCC) is a rapidly progressive tumor, not infrequently metastatic at diagnosis. Metastatic RCC is notoriously resistant to conventional RT and chemotherapy. However, considering its immunogenicity, promising results were obtained using targeted therapy and immunotherapy, which are nowadays the standard treatments in this setting [3]. Patients with metastatic RCC, in any case, continue to represent a therapeutic challenge.
Case report
In March 2017, a 56-year old (male) patient, during right lower limb paresthesia assessment, was diagnosed with a right kidney cancer, with bilateral multiple lung metastases measuring up to 1 cm in diameter.
In April 26th he underwent radical right nephrectomy with cavotomy and thrombectomy. Pathological examination showed a clear cell carcinoma, 7 cm of diameter, Fuhrman 3 of 4. The pathological TNM stage was pT3b pN0 M1 with angioinvasion, necrosis and invasion of kidney vein (thrombus).
In June 2017, after a CT scan showing a lung metastasis progression, the patient started targeted therapy with Sunitinib. The patient presented a long period of lung disease stability, until January 2020, when, according to RECIST criteria, he experienced a dimensional progression of two metastases located in the left medium and lower lobe.
Due to oligoprogressive pattern, the case was discussed in a multidisciplinary context and a salvage radiotherapy approach was considered appropriate.
The Radiation Oncologist decided to refer the patient to a SABR, with M6 Cyberknife® (CK) system [Accuray, Inc, Sunnyvale, California], directed to the only two progressive lesions. The treatment was performed with Xsight Spine tracking, a static tracking method using the alignment of the spinal skeleton structure and relying entirely on an Internal Gross Target Volume (IGTV) based approach. Full‐inhale and full‐exhale phase Computed Tomography (CT) data with a 1,25 mm thickness were acquired and an “inhale GTV “ and “exhale GTV” were delineated. The IGTV encompassed the whole respiratory tumor motion area, using the two GTVs, and an isotropic margin of 5 mm was added to create the PTV.
The dose prescription for both lesions was 50 Gy in 5 fractions to the 77% isodose.
The treatment was performed from February 24th to February 28th 2020. Concomitant Sunitinib (50 mg/day, 4 weeks on and 2 weeks off) was maintained. SABR was well tolerated and no toxicity or change inquality of life has been reported.
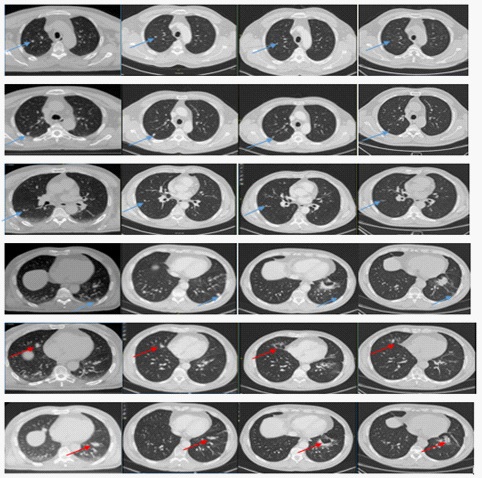
At first follow-up evaluation, three months later, the CT scan demonstrated stable disease with millimetric progression of all lung lesions.
Surprisingly, at the six-month CT scan there was evidence of dimensional reduction of all lung metastases (treated and not treated). The response was confirmed with evidence of further dimensional reduction at the nine-month and twelve-month CT scans.
Due to this unexpected result, the treatment plan was reviewed to verify that none of the untreated lesions was close to the two treated metastases and had received an indirect dose of radiation. A direct effect of radiation therapy in inducing the response of untreated lesions was excluded.
At the last follow up, 12 months after the SABR, the patient was in excellent general condition, and the Medical Oncologist maintained the Sunitinib prescription (37.5 mg/day, 4 weeks on and 2 weeks off).
Discussion
Sunitinib, a multitarget tyrosine kinase inhibitor of VEGFR1, VEGFR2, VEGFR3, PDGFR, c-kit, FLT3 and ret, is a well-studied angiogenesis inhibitor with an acceptable single agent toxicity profile. Preclinical data suggest that Sunitinib and other angiogenesis inhibitors enhance response to radiotherapy [4]. In addition to its effects on angiogenesis, several research groups have demonstrated robust effects of Sunitinib on inhibition of immune-suppressive myeloid-derived suppressor cells [5]. Acting on multiple targets, Sunitinib could enhance apoptosis and reduce clonogenic survival when given together with RT.
In metastatic RCC, concurrent Sunitinib and SABR is an active, frequently used regimen.
We are aware of the possibility that in our patient the radiation treatment stopped the increasing of the two lesions, which were outside the control of Sunitinib, while Sunitinib continued to control the other lesions, all leading to the excellent overall response. However, taking into account the evolution of the response, the possibility of an Abscopal Effect (AE) is an unavoidable hypothesis.
Few cases report AE in metastatic RCC after local radiotherapy [6,7]. In a small case series of 4 patients, published by Wersäll and coworkers [6], all patients who exhibited an AE lived more than 50 years, demonstrating the long-lasting anti-tumor effect in these cases. Interestingly, our case highlights the potential synergy between SABR and antiangiogenic therapies. The patient was treated with ablative radiotherapy instead of lower doses in order to maximize local control, not considering, at least in the first instance, the possibility of an AE. Perhaps the irradiation of two targets, and not just one, contributed to the good result [8].
The AE is extremely rare. Only 46 cases were reported from 1969 to 2014 (45 years!), more precisely only 7 cases of AE in RCC were reported in this long period of time [9]. Very few authors have described AE and among these, Wersäll described as many as four cases. Our hypothesis is that this phenomenon was not systematically sought and may therefore be more frequent than we are apt to think. Hence our idea to bring this case to the attention of other clinicians, in order to increase the cases described. We are also aware that recent randomized trials failed to observe an AE [10].
Pseudo progression, defined as an initial increase in tumor burden followed by a response to therapy, has been observed in 7% to 8% of RCC patients, especially those treated with immuno-checkpoint inhibitors. The root cause of pseudo progression is unclear, and may reflect various factors such as a brisk inflammatory response as well as with drawal from the effects of prior therapy, which despite overall tumor progression may still have inhibited tumor growth to some extent.
Differentiating between pseudo progression and true progression is important. Maintaining the same systemic therapy in the setting of true progression exposes patients to adverse side effects and delays potentially effective treatments. Prematurely terminating an effective therapy may reduce its benefit. Our case report demonstrates that long-term benefit can be obtained, despite an apparent initial radiological progression, highlighting the need for biomarkers and imaging techniques that can help differentiate between pseudo progression and true progression in a clinically significant time frame.
Strategies that aim to maintain a systemic treatment in patients with oligoprogressive disease, adding a local treatment only to the site of progression so as not to use all the systemic therapies available in a short period, leaving the patient without alternatives, are being increasingly adopted. Results beyond expectations, like this one, may be a sign that this strategy deserves more in-depth systematic research.
Conflicts of interest/Competing interests: no conflicts of interest to declare.
Patient consent: obtained.
References
- Park HJ, Griffin RJ, Hui S, et al, radiation-induced vascular damage in tumors: implications of vascular damage in ablative hypofractionated radiotherapy (SBRT and SRS). Rad Res. 2012; 177: 311-327.
- Hellevik T, Martinez-Zubiaurre I, Radiotherapy and the tumor stroma: The importance of dose and fractionation. Front Oncol; 2014; 4: 1.
- Ljungberg B, Albiges L, Bensalah K, et al. EAU Guidelines. Edn. presented at the EAU Annual Congress Milan 2021. ISBN 978-94- 92671-13-4.EAU Guidelines Office, Arnhem, The Netherlands. http://uroweb.org/guidelines/compilations-of-all-guidelines/
- Mauceri HJ, Nanna NN, Beckett MA, et al. Combined effects of angiostatin and ionizing radiation in antitumour therapy. Nature; 1998; 394: 287–291.
- Ozao-Choy J, Ma G, Kao J, et al. The novel role of tyrosine kinase inhibitor in the reversal of immune suppression and modulation of tumor microenvironment for immune-based cancer therapies. Cancer Res; 696: 2514–2522.
- Hiromichi I, Bin ST, Hua R, Spontaneous regression of thoracic metastases while progression of brain metastases after stereotactic radiosurgery and stereotactic body radiotherapy for metastatic renal cell carcinoma: abscopal effect prevented by the blood-brain barrier? Clin Genitourin Cancer. 2012; 10: 196-198.
- Wersall PJ, Blomgren H, Pisa P, et al, Regression of non-irradiated metastases after extracranial stereotactic radiotherapy in metastatic renal cell carcinoma. Acta Oncol. 2006; 45: 493-497.
- Brooks ED, Chang JY. Time to abandon single-site irradiation for inducing abscopal effects. Nat Rev Clin Oncol. 16: 123-135.
- Buodeh Y, Venkat P, Kim S. Systematic review of case reports on the abscopal effect. Current Problems in Cancer. 2016; 40: 25- 37.
- McBride S, Sherman E, Jillian Tsai C, et al. Randomized Phase II Trial of Nivolumab with Stereotactic Body Radiotherapy versus Nivolumab alone in metastatic head and neck squamos cell carcinoma. J Clin Oncol. 2021; 39: 30-37. https:// doi.org/10.1200/ JCO.20.00290