Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
PD-1/PD-L1 inhibitors combined with chemotherapy in the treatment of thymic carcinoma metastasis to the liver
Weiling Chen1,2#; Zhihang Huang3#; Yongqu Zhang1,2; Qun Chen5; Ronghui Li1,2,4*
1 Xiamen Key Laboratory of Endocrine-Related Cancer Precision Medicine, Xiamen University, Xiamen 361101, China.
2 Cancer Research Center, School of Medicine, Xiamen University, Xiamen, China.
3 Department of Cardiology Department, Xiang’an Hospital of Xiamen University, 2000 East Xiang’an Rd., Xiamen, China.
4 Department of Medical Oncology, Xiang’an Hospital of Xiamen University, 2000 East Xiang’an Rd., Xiamen, China.
5 Department of Clinical Research, Sun Yat-Sen University Cancer Center, 651 East Dongfeng Rd., Guangzhou 510060, China.
*Corresponding Author: Ronghui Li
Department of Medical Oncology, Xiang’an Hospital of Xiamen University, 2000 East Xiang’an Rd.,
Xiang’an, Xiamen, China.
Email: lironghui881155@126.com
Received : Mar 17, 2022
Accepted : Apr 06, 2022
Published : Apr 13, 2022
Archived : www.jcimcr.org
Copyright : © Ronghui Li (2022).
Abstract
Epithelial neoplasms of the thymus is a relatively rare malignancy. In this report, we present a case of thymic carcinoma of a 68-yearold man with hepatic metastasis, which with the severe hypokalemia, hypoproteinemia, and fatigue of concomitant symptom. After positive symptomatic treatment such as correction of electrolyte disorder, supplementation of albumin and nutritional support, the patient’s general condition recovered and improved. Then, we reviewed the literature on the treatment of thymic carcinoma, and decided to use immunotherapy combined with chemotherapy to treat this case of advanced thymic squamous carcinoma, expecting to have a good efficacy and improve the progression-free and overall survival.
Keywords: Thymic carcinoma; Chemotherapy; Immunotherapy.
Citation: Chen W, Huang Z, Zhang Y, Chen Q, Li R, et al. PD-1/PD-L1inhibitors combined with chemotherapy in the treatment of thymic carcinoma metastasis to the liver. J Clin Images Med Case Rep. 2022; 3(4): 1791.
Background
Thymic epithelial neoplasms include thymoma, thymic carcinoma and neuro-endocrine. Rare, they nevertheless represent 20% of all mediastinal tumors and 50% of those located in the anterior mediastinum, with very few extrathoracic spread [1]. Thymic carcinoma is more aggressive than thymoma, and accounts for about 15% of the incidence of thymic tumors [2]. The most common histological types of thymic carcinoma are squamous cell carcinoma, lymphoepithelial carcinoma, undifferentiated carcinoma and so on. Complete surgical resection with postoperative adjuvant radiotherapy and chemotherapy is preferred for surgically resectable thymic epithelial neoplasms. Radiotherapy and chemotherapy are the main treatments for advanced unresectable thymic epithelial neoplasms, but there is a lack of standard treatment. In recent years, targeted therapy drugs and immune checkpoint inhibitors have shown certain therapeutic prospects in thymic carcinoma.
For advanced or metastatic thymic carcinoma, platinumbased combination chemotherapy should be used to shrink the tumor and alleviate the associated symptoms. At present, the standard chemotherapy regimens are still uncertain, but paclitaxel + carboplatin is the first choice for the first-line chemotherapy of thymic carcinoma, and fluorouracil/tetrahydrofolate, gemcitabine/capecitabine, paclitaxel, pemetreuse, etoposide, isophoramide and so on, can be selected for the second-line chemotherapy [3]. There is a lack of effective evidence-based medical evidence for targeted therapy of thymic carcinoma, but the new drugs in the study include everolimus, sunitinib and others [4]. Further, some studies have shown that patients with higher PD-L1 expression in thymic carcinoma have better efficacy after receiving immunotherapy [5,6].
In this case report, we present a patient with liver metastasis who had no indication for surgery. Then, he was treated with Camrelizumab that is the PD-1 immunotherapy combined with Paclitaxel for Injection and carboplatin, which showed a good therapeutic effect.
Case report
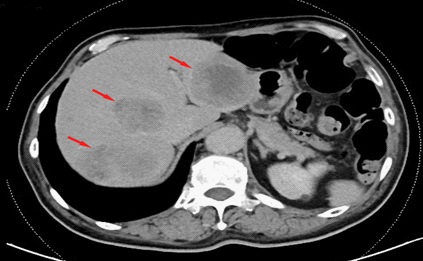
A 68-year-old man who developed poor, fatigue, and emaciation for 4 months, was not attention to treatment. He was admitted to the emergency department because his symptoms gradually worsened and he developed nausea, vomiting, and difficulty swallowing. Emergency whole-abdominal CT examination revealed multiple liver lesions with a diameter of about 67 mm for the larger one, which enhanced scan showed ring enhancement, so it was considered as a metastatic tumor (Figure 1).
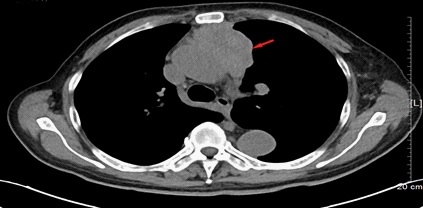
After admission, the patient was further examined by chest CT, and then found a soft tissue mass in the anterior mediastinum which uneven density and fuzzy edge of mass, disappearance of local and perivascular space. The size of the tumour was about 37 mm X 69 mm, which accompanied by multiple lymph node enlargement in the anterior mediastinum, left neck root, clavicular and left axilla (Figure 2).
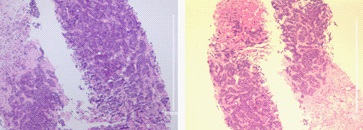
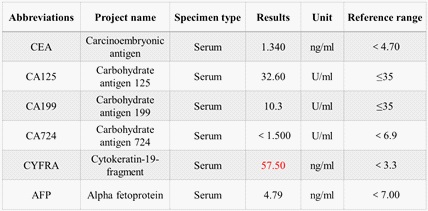
Subsequently, a coarse needle biopsy of the liver mass was performed, which indicated that the liver tissue was infiltrated by malignant tumor. Combined with immunohistochemical results, the liver metastasis of squamous cell carcinoma was consistent with thymic carcinoma (Figure 3). Immunohistochemistry investigations demonstrated the carcinoma cells were positive for CD5, CD117, P40, CK5/6, P53 and Ki-67 (50%), and negative for other immunomarkers (CK20, CK7, CEA, GPC3, Villin, PSAP, NapsinA, CgA, Syn) in the panel, which supported the diagnosis of a metastasis from a primary thymic carcinoma rather. Associated tumor marker tests suggested that only the level of CYFRA was higher, while the other markers were within the normal range (Figure 4).
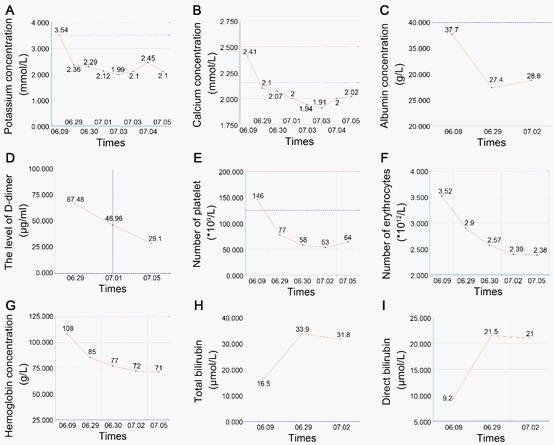
After diagnosis, the patient’s physical condition was always poor, with persistent hypokalemia, hypocalcium and hypoproteinemia, and then gradually hapened the anemia, elevated bilirubin and hypercoagulability (Figure 5). We corrected the electrolyte disturbances (more than 10 g of potassium every day), and supplemented albumin and blood transfusions boosted the immune function. Furthermore, low-molecular-weight heparin sodium injection was given as anticoagulant, and bilirubin was reduced, and more hepatocyte function was protected. Then, the patient was stabilized and started on albumin-bound paclitaxel and carboplatin combined with the PD-1 inhibitor Camrelizumab for therapy.
Discussion
Thymic carcinoma is a rare mediastinal malignancy caused by thymic epithelial cells, with an incidence of 0.13 to 0.32 per million [7]. Thymic carcinoma tends to occur in adult males over the age of 50 [8]. The most commonhistopathological types are squamous and undifferentiated carcinoma. Many thymic carcinoma patients were initially diagnosed with invasion or metastasis, usually involving surrounding organs or metastasized to mediastinal lymph nodes, pleura, lung, pericardium and so on [9]. Few thymic carcinomas are found only on physical examination without any clinical symptoms.
The onset of thymic tumor is insidious, and the first symptoms of patients are often local mediastinal compression symptoms, such as chest tightness, shortness of breath, head and face swelling, dyspnea, severe cough and so on. In addition, when the tumor compresses the sympathetic nerve, it can cause ipsilateral blepharoptosis, pupil narrowing, enophthalmos, frontal apperspiration, and Horner syndrome. Hoarseness can be caused when the tumor compresses the recurrent laryngeal nerve. And superior vena cava obstruction syndrome can be occurred when it is compressed. One third of thymomas are associated with autoimmune diseases [10], most of which are related to anti-acetylcholine receptor antibodies, including myasthenia, aplastic anemia and hypoproteinemia [11].
This patient’s initial symptoms were abdominal discomfort and digestive tract symptoms, mainly due to thymic carcinoma metastasize to the liver. Afterward, he developed severe electrolyte disturbances, hypoproteinemia and anemia, which is consistent with the autoimmune symptoms associated with thymoma.
Study reported that about one-third of patients with thymic carcinoma were initially diagnosed with lymph node or distant metastasis [12], which often metastasize to liver, bones, lung, pleura or lymph nodes.
P53 is one of the most important tumor suppressor genes discovered so far, whose mutation can lead to dysregulation of cell apoptosis, uncontrolled cell proliferation and tumgenesis, and suggest a poor prognosis [13,14]. Ki-67 is a nuclear protein that can be used as a marker of cell proliferation [15,16], and its index is closely related to the degree of differentiation, invasion, metastasis and prognosis of many tumors. In this case, p53 expression was positive and the positive Ki-67 staining was more than 50%, which suggest that this patient’s thymic carcinoma is more aggressive and capable of distant metastasis.
The standard treatment for thymic carcinoma has not been established and its prognosis is poor. Clinical experience suggests that paclitaxel and carboplatin are the first choice for chemotherapy. At the same time, PD-1/L1 immunotherapy for thymic carcinoma was found to have significant clinical benefits [17-19]. Katsuya et al. reported a phase II study of Nivolumab in 15 patients with unresectable or relapsed thymic carcinoma, including 13 patients with squamous cell carcinoma, which subgroup analysis showed a median PFS of 3.8 months and an OS of 14.1 months [20]. Studies have found that the expression level of PD-L1 are reported to be higher in thymic carcinoma than in thymoma, but it remains unclear whether the expression level of PD-L1 predicts the prognosis of thymic carcinoma [21]. Albumin-paclitaxel is based on paclitaxel and added albumin as a carrier, which has better efficacy and fewer adverse reactions. Therefore, this patient was treated with albumin-paclitaxel and carboplatin for chemotherapy, and combined with Camrelizumab for immunotherapy.
Declarations
Acknowledgments: This research was supported by the Science and Technology Projects of Xiamen municipal Bureau of Science and Technology (Grant No. 3502Z20194042 and No.3502Z20199047) and the Youth Funding project of Xiang ‘an Hospital (Grant No. PM202103050005).
Conflict of interest: The authors declare that they have no conflict of interest.
References
- Thomas PA, Payan-Defais MJ. Epithelial tumours of the thymus. Rev Pneumol Clin. 2010; 66: 41-51.
- Bansal N, Roychoudhury AK, Dhingra H. Thymic carcinoma - a rare case report. Clujul Med, 2018; 91: 238-241.
- Berghmans T, et al. Systemic treatments for thymoma and thymic carcinoma: A systematic review. Lung Cancer. 2018; 126: 25- 31.
- Krishnan M, Ganti AK. The role of targeted therapy in thymic carcinoma. J Oncol Pharm Pract. 2019; 25: 1712-1718.
- Giaccone G, et al. Pembrolizumab in patients with thymic carcinoma: A single-arm, single-centre, phase 2 study. Lancet Oncol. 2018; 19: 347-355.
- Jakopovic M, et al. Immunotherapy for thymoma. J Thorac Dis. 2020; 12: 7635-7641.
- Okuma Y, et al. Phase II trial of S-1 treatment as palliative-intent chemotherapy for previously treated advanced thymic carcinoma. Cancer Med. 2020; 9: 7418-7427.
- Girard N, et al. Thymic epithelial tumours: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015; 26 Suppl 5: v40-55.
- Wei ML, et al. Chemotherapy for thymic carcinoma and advanced thymoma in adults. Cochrane Database Syst Rev. 2013; CD008588.
- Alexakou Z, et al. Thymic Carcinoma With Multiple Paraneoplastic Disorders. Am J Med Sci. 2021.
- Robinson SP, Akhondi H. Thymoma, in Stat Pearls. 2021: Treasure Island (FL).
- Yang Y, et al. Stage IVb thymic carcinoma: Patients with lymph node metastases have better prognoses than those with hematogenous metastases. BMC Cancer. 2017; 17: 217.
- Kanapathipillai M. Treating p53 Mutant Aggregation-Associated Cancer. Cancers (Basel). 2018; 10.
- Tang Q, et al. Mutant p53 on the Path to Metastasis. Trends Cancer. 2020; 6: 62-73.
- Sun X, Kaufman PD. Ki-67: More than a proliferation marker. Chromosoma. 2018; 127: 175-186.
- Jabbarzadeh M, et al. Ki-67 expression as a diagnostic biomarker in odontogenic cysts and tumors: A systematic review and metaanalysis. J Dent Res Dent Clin Dent Prospects. 2021; 15: 66-75.
- Yokoyama S, Miyoshi H. Comparison of PD-L1 immunohistochemical assays and the significance of PD-L1 expression in thymoma. J Thorac Dis. 2020; 12: 7553-7560.
- Turna A, Sarbay I. Multimodality approach in treatment of thymic tumors. J Thorac Dis. 2020; 12: 7626-7634.
- Kaira K, Imai H, Kagamu H. Perspective of Immune Checkpoint Inhibitors in Thymic Carcinoma. Cancers (Basel). 2021; 13.
- Katsuya Y, et al. Single-arm, multicentre, phase II trial of nivolumab for unresectable or recurrent thymic carcinoma: PRIMER study. Eur J Cancer. 2019; 113: 78-86.
- Arbour KC, et al. Expression of PD-L1 and other immunotherapeutic targets in thymic epithelial tumors. PLoS One. 2017; 12: e0182665.