Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Neurological relapse of acute lymphoblastic leukemia mimicking Guillain-Barré syndrome
Oumerzouk J1,4*; Klevor R3,4; Raissi A2; Yahyaoui H2; Louhab N3,4; Kissani N3,4
1 Neurology Department, Military Hospital Avicenne, Marrakech, Morocco.
2 Hematology Department, Military Hospital Avicenne, Marrakech, Morocco.
3 Neurology Department, Mohamed VI hospital, Marrakech, Morocco.
4 Neuroscience Research Laboratory, Marrakech Medical School, Cadi Ayyad University, Marrakesh, Morocco.
*Corresponding Author: Oumerzouk Jawad
Military Hospital Avicenne, Marrakech 10100,
Morocco.
Email: tamamro@yahoo.fr
Received : Mar 18, 2022
Accepted : Apr 07, 2022
Published : Apr 14, 2022
Archived : www.jcimcr.org
Copyright : © Jawad O (2022).
Abstract
Acute Lymphoblastic Leukemia (ALL) is the most common cancer of childhood. While some 5 to 8% of ALL cases will have neurological involvement at initial presentation, up to 30% of relapses have neurological manifestations. We present the case of a pediatric ALL with meningo-radiculoneuropathy revealing a relapse while on maintenance therapy, mimicking Guillain-Barré syndrome. Our patient presented the Philadelphia mutation, that is the t(9;22), and thus was at high risk of neurological involvement. The diagnosis of relapse was based on neuroimaging and CSF study. Treatment entailed modified Hyper CVAD chemotherapy with repeated intrathecal treatment until CSF clearance. Outcome was favorable with gradual regression of neurological symptoms.
Keywords: Lymphoblastic; Leukemia; Polyradiculoneuritis; CVAD.
Citation: Oumerzouk J, Klevor R, Raissi A, Yahyaoui H, Louhab N, et al. Neurological relapse of acute lymphoblastic leukemia mimicking Guillain-Barré syndrome. J Clin Images Med Case Rep. 2022; 3(4): 1792.
Introduction
Acute Lymphoblastic Leukemia (ALL) is the most common cancer of childhood and an important cause of death from cancer during the first two decades of life [1]. Recent advances in the fields of diagnostics and therapeutics have improved prognostic outcomes in patients [2,3]. Involvement of the nervous system remains, however, an issue of concern, and constitutes a negative prognostic factor [4,5]. While some 5 to 8% of ALL cases will have neurological involvement at initial presentation, up to 30% of relapses have neurological manifestations [4,6].
We present the case of a pediatric ALL with meningo-radiculoneuropathy revealing a relapse while on maintenance therapy and after contracting COVID-19 infection. This case study demonstrates how a thorough neurological examination and appropriate diagnostic workup are required to parse out the various differentials.
Case report
A 12-year-old girl, the third of 3 children born to a first degree consanguineous marriage, presented with acute motor impairment of the four limbs, predominant on lower extremities. The patient was followed up for acute B-cell Lymphoblastic Leukemia (B-cell ALL), treated with the FRALLE 2000 protocol and on the maintenance phase at the time of presentation. The patient had a female maternal cousin with a malignant hemopathy.
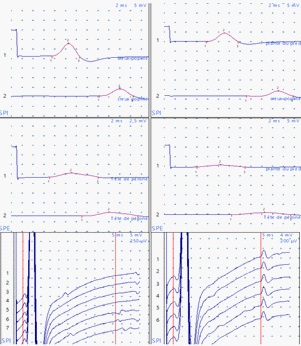
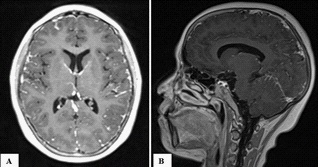
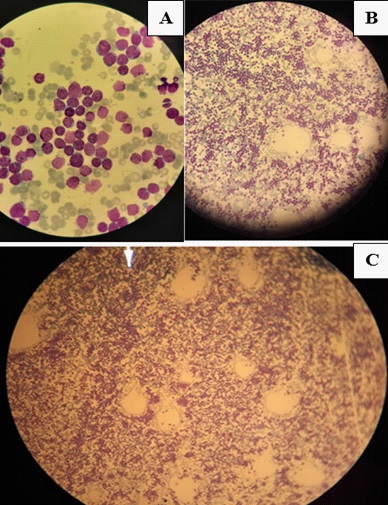
The clinical presentation goes back 20 days with the patient reporting a dry cough and fever. Covid-19 Polymerase Chain Reaction (PCR) was positive and the patient started on antibiotics and symptomatic treatment as well as respite from chemotherapy. The patient presented at the emergency department, with acute diplopia, urinary retention and weakness of the four limbs. On admission, the patient was conscious, stable on cardiorespiratory evaluation, afebrile without pallor or signs of hemorrhage or tumor. On neurological examination, the patient presented with tetraparesis with decreased muscle tone and absent deep tendon reflexes on all limbs (4/5 proximally and distally, bilaterally on upper extremities; 3-/5 proximally and distally, bilaterally on lower extremities on the Medical Research Council muscle strength grading). Plantar response was equivocal bilaterally. There was reduced pinprick sensation at the pulp of all fingers and in the lower extremities up to the knees with altered position sense of toes and reduced vibration perception up to the knees bilaterally. There was also urinary retention and constipation at presentation. Sensory perception was preserved in the perineal region. There was slight facial and abducens nerves involvement on the right side with slight extrinsic third nerve palsy on the left. The electroneuromyography revealed sensorimotor axonal polyradiculoneuropathy. The motor nerve conduction study showed a decrease in motor amplitudes in distal lower limbs, predominating on the common peroneal nerve, without signs of demyelination, motor conduction block or temporal dispersion. F wave latency was prolonged in the lower extremities with decreased sensory amplitudes for the sural and ulnar nerves. The needle ENMG showed a neurogenic tracing, with signs of active denervation and temporal summation, in the 4 limbs, according to a pluriradicular distribution (Figure 1). Brain and spinal Magnetic Resonance Imaging (MRI) revealed diffuse leptomeninges contrast enhancement of the whole central nervous system, including caudaequina roots (Figure 2). Whole body tomography was unremarkable. Cerebrospinal Fluid (CSF) study revealed 3590 atypical lymphocytes per mm3. Complete blood count (Hemoglobin: 15.2 g/dl; Platelets: 231 000/mm3; White blood cells: 9930/mm3) and peripheral blood smear were unremarkable. Bone marrow examination revealed 15% lymphoblasts (Figure 3). Prothrombin Time (PT) was 85%, Kaoline Clotting Time (KCT) was 35.3 seconds compared to a standard of 32 seconds. Fibrinogen was 3.11 g/l. Serum electrolytes, blood urea and nitrates, phosphates and calcium in the blood were unremarkable except for elevated uric acid (454 mg/l) and lactate dehydrogenase (737 u/l) levels. Immunophenotyping revealed B-cell ALL. Karyotype study of 27 cells using RHG technique revealed 46, XX, t(9;22) (q34:q11)[1]/52, idem, +X, +4, +6, +10, +mar1, mar2 [2]/46, XX [24]… a minority pseudodiploid cell clone of 46 chromosomes with a t(9;22) translocation indicating a positive Philadelphia chromosome (1 mitosis). Another hyperdiploidysubclone of 52 chromosomes with trisomies for chromosomes X, 4, 6, 10 and two chromosomes as markers (2 mitoses). A clone without anomalies found on examination (24 mitoses). Molecular biology using multiplex PCR did not find any transcript of BCR-ABL fusion. Human Leucocyte Antigen (HLA) typing using PCR and reverse dot-blot showed no HLA-A*29, HLA-B*27 or HLA-B*51.
Treatment entailed modified Hyper CVAD chemotherapy with repeated Intrathecal Treatment (IT) until CSF clearance, hyper hydration, allopurinol, proton inhibitor, prophylaxis with acyclovir and fluconazole. At the follow-up examination, diplopia resolved after the first cycle of HyperCVAD and CSF cleared after 4 IT treatments. Post induction bone marrow aspirate showed no excess of blasts. Consecutive HyperCVAD cycles were well tolerated. Human Leucocyte Antigen typing of the siblings failed to show compatibility, and the patient is planned to receive pheno-identical stem cell transplantation.
Discussion
ALL is a malignant transformation and proliferation of lymphoid progenitor cells in the bone marrow, blood and extramedullary sites [6]. This transformation is due to chromosomal abnormalities in precursors of B-cell or T-cell lineage. The majority of ALL occur in B-cell lineage as is the case of our patient [7]. Several genetic syndromes have been shown to present a higher incidence of the disease [8-10]. However, the majority of cases are de novo mutations [11]. Other risk factors have been identifies such as immunodepression, viral infection, toxic substances and ionizing radiation. Our patient’s history of being born to a first degree consanguineous marriage and a cousin with a history of unspecified malignant hemopathy are significant indicators of a possible transmissible genetic aberration. However, the patient’s clinical examination, did not reveal any dysmorphic, cutaneous or pre-diagnosis neurological issues suggestive of well-known syndromes such as Down’s syndrome, Fanconni’s anemia and ataxia-telangiectasia syndrome associated with ALL [8-10]. Global incidence rates have been estimated to be between 1.08 and 2.12 per 100,000 person-years, the condition being four times more frequent in childhood than in adulthood.
While neurological manifestations are relatively infrequent at the time of diagnosis, it is classic to find neurological involvement at presentation during relapses [4,6]. Our patient did not report neurological symptoms at presentation 2 months before, at the time the diagnosis of ALL was made. Instead, neurological involvement was the presenting feature of her relapse. The high frequency of these symptoms during relapse makes them important to recognize for early diagnosis and management. Central Nervous System (CNS) involvement is a major prognostic factor since it is a primary cause of mortality. CNS involvement entails a pleomorphic presentation. Cranial nerve palsy, signs and symptoms of meningeal irritation and increased intracranial pressure are important to recognize. Parenchymal involvement of the encephalic localization is seen in later stages of ALL [12]. Spinal cord involvement and hormonal deficit due to pituitary infiltration have also been described [13]. Importantly, peripheral involvement, aside from cranial nerves, could also be seen in these patients [14]. Management of these manifestations remains challenging with the risk of neurocognitive complications due to treatment [15].
Our patient presented, not only with cranial nerve involvement, but also polyradiculoneuritis. While it stands to reason that the context of neoplastic history makes the B-cell ALL a very likely culprit, it must be pointed out that in the case of our patient, other differentials are important to discuss [16], mainly post infectious acute polyradicluloneuropathy (Guillain-Barré syndrome), given the immunocompromised areas and the current pandemic context (Covid-19) [17]. Nevertheless, toxic neuropathies should also be considered. Performing Lumbar Puncture (LP) is an important procedure at the time of diagnosis and MRI is required if CNS involvement is suspected [18,19]. Furthermore, an important distinguishing criteria of neoplastic/ paraneoplasticpolyradiculoneuritis is the associated constitutional symptoms. In B-cell ALL, non specific ‘B symptoms’ such as fever, weight loss, fatigue, anorexia and night sweats are frequent but could also be seen in infectious diseases such as tuberculosis andLyme disease, as well as in vasculitis. Performing PCR in the CSF (GenXpert……) would also be contributory.
Only few cases of peripheral neuropathy associated with ALL relapse have been reported in the literature [20-22]. An important distinction must be made here. For example, nerve infiltration or neuroleukemiosis has been reported, in which mononeuropathy is the presenting picture [20]. A case of Guillain-Barré syndrome has been reported in which polyradiculoneuritis was an incidental diagnosis in a patient with ALL but was not related either to the neoplasm or to vincristine exposure [22]. Finally, only one case of ALL-related polyradiculoneuritis has been described in the literature to the best of our knowledge [21]. The possible presentation of ALL relapse with the clinical picture of Guillain-Barré syndrome therefore holds an important lesson for the practitioner. Polyradiculoneuritis in a patient in ALL remission should prompt the search for neurological relapse which in the case of the later example was followed by systemic relapse [21].
Only few cases of peripheral neuropathy associated with ALL relapse have been reported in the literature [20-22]. An important distinction must be made here. For example, nerve infiltration or neuroleukemiosis has been reported, in which mononeuropathy is the presenting picture [20]. A case of Guillain-Barré syndrome has been reported in which polyradiculoneuritis was an incidental diagnosis in a patient with ALL but was not related either to the neoplasm or to vincristine exposure [22]. Finally, only one case of ALL-related polyradiculoneuritis has been described in the literature to the best of our knowledge [21]. The possible presentation of ALL relapse with the clinical picture of Guillain-Barré syndrome therefore holds an important lesson for the practitioner. Polyradiculoneuritis in a patient in ALL remission should prompt the search for neurological relapse which in the case of the later example was followed by systemic relapse [21].
Only few cases of peripheral neuropathy associated with ALL relapse have been reported in the literature [20-22]. An important distinction must be made here. For example, nerve infiltration or neuroleukemiosis has been reported, in which mononeuropathy is the presenting picture [20]. A case of Guillain-Barré syndrome has been reported in which polyradiculoneuritis was an incidental diagnosis in a patient with ALL but was not related either to the neoplasm or to vincristine exposure [22]. Finally, only one case of ALL-related polyradiculoneuritis has been described in the literature to the best of our knowledge [21]. The possible presentation of ALL relapse with the clinical picture of Guillain-Barré syndrome therefore holds an important lesson for the practitioner. Polyradiculoneuritis in a patient in ALL remission should prompt the search for neurological relapse which in the case of the later example was followed by systemic relapse [21].
Declarations
Competing interests: The authors report no conflicts of interest.
Funding: No financial sources to declare.
References
- Smith MA, Seibel NL, Altekruse SF, Ries LAG, Melbert DL, O’Leary M, et al. Outcomes for Children and Adolescents With Cancer: Challenges for the Twenty-First Century. JCO 2010; 28: 2625– 2634. https://doi.org/10.1200/JCO.2009.27.0421.
- Tatar AS, Nagy Simon T, Tomuleasa C, Boca S, Astilean S. Nanomedicine approaches in acute lymphoblastic leukemia. J Control Release. 2016; 238: 123–138. https://doi.org/10.1016/j.jconrel.2016.07.035.
- Gafencu GA, Tomuleasa CI, Ghiaur G. PARP inhibitors in acute myeloid leukaemia therapy: How a synthetic lethality approach can be a valid therapeutic alternative. Med Hypotheses. 2017; 104: 30–34. https://doi.org/10.1016/j.mehy.2017.05.015.
- Baytan B, Evim MS, Güler S, Güneş AM, Okan M, et al. Acute Central Nervous System Complications in Pediatric Acute Lymphoblastic Leukemia. Pediatr Neurol. 2015; 53: 312–318. https://doi.org/10.1016/j.pediatrneurol.2015.03.006.
- Johnston DL, Alonzo TA, Gerbing RB, Aplenc R, Woods WG, Meshinchi S, et al. Central nervous system disease in pediatric acute myeloid leukemia: A report from the Children’s Oncology Group. Pediatr Blood Cancer. 2017; 64. https://doi.org/10.1002/ pbc.26612.
- Terwilliger T, Abdul Hay M. Acute lymphoblastic leukemia: A comprehensive review and 2017 update. Blood Cancer J. 2017; 7: e577–e577. https://doi.org/10.1038/bcj.2017.53.
- Hunger SP, Mullighan CG. Acute Lymphoblastic Leukemia in Children. New England Journal of Medicine. 2015; 373: 1541–1552. https://doi.org/10.1056/NEJMra1400972.
- Shah A, John BM, Sondhi V. Acute lymphoblastic leukemia with treatment--naive Fanconi anemia. Indian Pediatr. 2013; 50: 508– 510.
- Bielorai B, Fisher T, Waldman D, Lerenthal Y, Nissenkorn A, Tohami T, et al. Acute lymphoblastic leukemia in early childhood as the presenting sign of ataxia-telangiectasia variant. Pediatr Hematol Oncol. 2013; 30: 574–582. https://doi.org/10.3109/08 880018.2013.777949.
- Chessells J, Harrison G, Richards S, Bailey C, Hill F, Gibson B, et al. Down’s syndrome and acute lymphoblastic leukaemia: Clinical features and response to treatment. Arch Dis Child. 2001; 85: 321–325. https://doi.org/10.1136/adc.85.4.321.
- Mullighan CG, Collins-Underwood JR, Phillips LAA, Loudin ML, Liu W, Zhang J, et al. REARRANGEMENT OF CRLF2 IN B-PROGENITOR AND DOWN SYNDROME ASSOCIATED ACUTE LYMPHOBLASTIC LEUKEMIA. Nat Genet. 2009; 41: 1243–1246. https://doi. org/10.1038/ng.469.
- Ginsberg LE, Leeds NE. Neuroradiology of leukemia. AJR Am J Roentgenol 1995; 165: 525–534. https://doi.org/10.2214/ ajr.165.3.7645463.
- Zhang LD, Li YH, Ke ZY, Huang LB, Luo XQ, et al. Obesity as the initial manifestation of central nervous system relapse of acute lymphoblastic leukemia: Case report and literature review. J Cancer Res Ther. 2012; 8: 151–153. https://doi.org/10.4103/0973- 1482.95200.
- Anna Grisold WG. Leukemia and the Peripheral Nervous System: A review. J Leuk. 2014; 02. https://doi.org/10.4172/2329- 6917.1000162.
- Pui CH, Howard SC. Current management and challenges of malignant disease in the CNS in paediatric leukaemia. The Lancet Oncology. 2008; 9: 257–268. https://doi.org/10.1016/S1470- 2045(08)70070-6.
- Del Principe MI, Maurillo L, Buccisano F, Sconocchia G, Cefalo M, De Santis G, et al. Central nervous system involvement in adult acute lymphoblastic leukemia: Diagnostic tools, prophylaxis, and therapy. Mediterr J Hematol Infect Dis. 2014; 6: e2014075. https://doi.org/10.4084/MJHID.2014.075.
- Giometto B, Grisold W, Vitaliani R, Graus F, Honnorat J, Bertolini G, et al. Paraneoplastic neurologic syndrome in the PNS Euronetwork database: A European study from 20 centers. Arch Neurol. 2010; 67: 330–335. https://doi.org/10.1001/archneurol.2009.341.
- Rech A, de Carvalho GP, Meneses CF, Hankins J, Howard S, et al. The influence of traumatic lumbar puncture and timing of intrathecal therapy on outcome of pediatric acute lymphoblastic leukemia. Pediatr Hematol Oncol. 2005; 22: 483–488. https:// doi.org/10.1080/08880010591002242.
- Barranco Lampón G, Rozen Fuller E, Olarte Carrilo I, Martínez Tovar A, León González G, Castellanos Sinco H, et al. Association between traumatic lumbar puncture and the risk of central nervous system relapse in adults with acute lymphoblastic leukaemia. Rev Med Hosp Gen Mex. 2015; 78: 124–128. https:// doi.org/10.1016/j.hgmx.2015.08.002.
- Aregawi DG, Sherman JH, Douvas MG, Burns TM, Schiff D. Neuroleukemiosis: Case report of leukemic nerve infiltration in acute lymphoblastic leukemia. Muscle & Nerve. 2008; 38: 1196–1200. https://doi.org/10.1002/mus.21089.
- Boiron J-M, Ellie E, Vital A, Lagueny A, Vital C, Julien J, et al. Isolated Peripheral Nerve Relapse Masquerading as GuillainBarré Syndrome in a Patient with Acute Lymphoblastic Leukemia. Leukemia & Lymphoma. 1993; 10: 489–491. https://doi. org/10.3109/10428199309148207.
- Aral YZ, Gursel T, Ozturk G, Serdaroglu A. Guillain-Barré Syndrome in a Child with Acute Lymphoblastic Leukemia. Pediatric Hematology and Oncology. 2001; 18: 343–346. https://doi. org/10.1080/088800101300312618.
- Frishman Levy L, Izraeli S. Advances in understanding the pathogenesis of CNS acute lymphoblastic leukaemia and potential for therapy. Br J Haematol. 2017; 176: 157–167. https://doi. org/10.1111/bjh.14411.
- Alsadeq A, Schewe DM. Acute lymphoblastic leukemia of the central nervous system: on the role of PBX1. Haematologica. 2017; 102: 611–613. https://doi.org/10.3324/haematol.2017.165142.
- Sanchez R, Ayala R, Alonso RA, Martínez MP, Ribera J, García O, et al. Clinical characteristics of patients with central nervous system relapse in BCR-ABL1-positive acute lymphoblastic leukemia: the importance of characterizing ABL1 mutations in cerebrospinal fluid. Ann Hematol. 2017; 96: 1069–1075. https://doi. org/10.1007/s00277-017-3002-1.
- Thomas DA, O’Brien S, Faderl S, Garcia-Manero G, Ferrajoli A, Wierda W, et al. Chemoimmunotherapy With a Modified HyperCVAD and Rituximab Regimen Improves Outcome in De Novo Philadelphia Chromosome–Negative Precursor B-Lineage Acute Lymphoblastic Leukemia. JCO. 2010; 28: 3880–3889. https:// doi.org/10.1200/JCO.2009.26.9456.