Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Case Report - Open Access, Volume 3
Limping as the first manifestation of peripheral primitive neuroectodermal tumor (PNET) in children
Masoumeh Rashidpour Aghamahali1; Reza Shiari2*; Azadeh Zeinab Mirzaee2; Mehrnoush Hassas Yeganeh2; Ahmad Reza Reihani3; Yalda Nilipour4
1 Shahid Beheshti University of Medical Sciences, Tehran, Iran.
2 Shahid Beheshti University of Medical Sciences, Department of Pediatric Rheumatology, Mofid Children Hospital, Tehran, Iran.
3 Shahid Beheshti University of Medical Sciences, Department of Orthopedic, Mofid Children Hospital, Tehran, Iran.
4 Shahid Beheshti University of Medical Sciences, Department of Pathology, Mofid Children Hospital, Tehran, Iran.
*Corresponding Author: Reza Shiari, MD, PhD
Pediatric Rheumatologist, Associate Professor,
Mofid Children’s Hospital, Shariati Ave, Hosseinieh
Ershad, Tehran, Iran.
Email: shiareza@yahoo.com
Received : Apr 01, 2022
Accepted : Apr 26, 2022
Published : May 03, 2022
Archived : www.jcimcr.org
Copyright : © Shiari R (2022).
Abstract
Background: Ewing sarcoma (ES) and peripheral primitive neuroectodermal tumor (PNET) comprise the same spectrum of neoplastic diseases known as the Ewing sarcoma family of tumors (EFT). Ewing sarcoma is the most common bone sarcoma typically arising from bone and is one of the most common pediatric solid tumors. It mostly affects the bones of the lower parts of the body.
Case presentation: Herein we have introduced an 8.5-year-old girl who presented with symptoms of limping and nocturnal pain of the limb which had been exacerbated in the last three months. Diagnosis of PNET was made according to core needle biopsy and immunohistochemical staining.
Conclusion: Although A-traumatic limping occurs post infections, most malignancies should be considered as one of the main differential diagnoses in all children presenting with hip pain.
Keywords: Ewing sarcoma; Limping; PNET; Malignant bone tumor; Nocturnal pain; Bone scan; Core needle biopsy
Citation: Masoumeh RA, Reza S, Azadeh ZM, Mehrnoush HY, Ahmad RR, et al. Limping as the first manifestation of peripheral primitive neuroectodermal tumor (PNET) in children. J Clin Images Med Case Rep. 2022; 3(5): 1817.
Introduction
Malignant bone and soft tissue tumors are rare in childhood. However, Osteosarcoma and Ewing sarcoma are the two most common malignant bone tumors in children [1-5]. Ewing sarcoma typically arising from bone [6,7] and is one of the most common pediatric solid tumors with a highly aggressive behavior, with a survival of 70-80% for patients with standard-risk and localized disease and almost 30% for those with metastatic disease [8,9].
Herein, we described an 8.5-year-old girl with limping and bone pain, which finally lead to the final diagnosis of Ewing sarcoma.
Case report
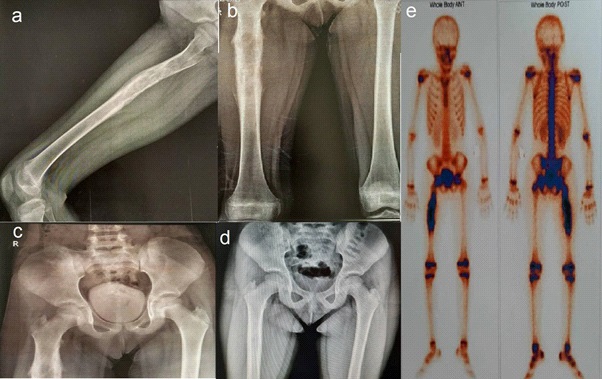
An 8.5-year-old girl was referred to the out-patient clinic of pediatric rheumatology in Mofid Children’s Hospital with limping that was exacerbated during the last three months. In fact, the symptom was started about a year ago as pain in the right leg, which increased at night and caused her to wake up. During this time, pain has spread to the lumbar and has been associated with decreased appetite. About 6 months ago, her physician was performed the first X-ray (Figure 1d), and blood tests. In a simple X-ray, due to not finding a specific problem and due to the spread of pain in the lumbar region, she underwent lumbosacral MRI, which was normal.
Her laboratory data on that time were as followings:
Complete blood count (CBC) showed WBC = 6800/mm3 (50% Neutrophils), Hgb = 13 g/dl, PLT = 291000/micL, ESR = 49 mm/ hr, CRP = 3+, Alkaline phosphatase = 43 IU/L, U/A = within normal limits.
On that time, she was diagnosed as having complex regional pain syndrome and was treated with naproxen, nefidipine and imipramine. However, she returns again due to lack of recovery and aggravation of symptoms and limping. The new laboratory data showed, WBC = 9000/mm3 (48 Neutrophils), Hgb = 13 g/dl, PLT = 315000 mic/L, ESR = 48 mm/hr, Wright & 2ME = negative, Calcium = 9 mg/dl, Phosphor = 3.3 mg/dl, Creatine phosphokinase = 31 IU/L, lactate dehydrogenase = 566 U/L.
Her new bone Rontgen radiography shows a suspicious view of the onion skin (Figures 1a, 1c), which is subjected to Bone Scan with injection of 20 MCI tc-99m mdp, that showed hyperemic and active bone lesion in the right shaft of Femora with no hip and knee involvement and no active bone lesion or metastasis in the rest of her skeleton (Figure 1e).
CT imaging guideline core needle biopsy was performed to detect pathology which showed a malignant neoplasm composed of sheets of uniform small to medium-sized round cells containing hyperchromatic nuclei with compact nuclei and occasional small nucleoli, as well as a scant eosinophilic cytoplasm with indistinct borders and a few scattered mitotic figures in fibrovascular stroma with Foci of necrosis.
Chest, abdomen and pelvic CT-Scan with and without contrast showed hypodence nodule diameter 4 mm in spleen and few lymphadenopathy in external iliac 15 mm size in short axis.
According to her clinical findings, Imaging and result of immunohistochemical staining in pathology, she was diagnosed as having Ewing sarcoma / PNET.
Discussion
Ewing sarcoma (ES) and peripheral primitive neuroectodermal tumor (PNET) comprise the same spectrum of neoplastic diseases known as the Ewing sarcoma family of tumors (EFT). These diseases are associated with malignant small-cell tumors that can be found both in the bone and soft tissues of the extremities, but can also present in the axial skeleton. This family of tumors has the same chromosomal translocation t(11;22). Osteosarcoma and Ewing sarcoma are the two most common malignant bone tumors in children [1-5].
Treatment comprises local surgery, radiotherapy and chemotherapy, which are associated with acute and chronic adverse effects that may compromise quality of life in survivors. Histologically, Ewing sarcomas are composed of small round cells expressing high levels of CD99 and Genetically, they are characterized by balanced chromosomal translocations in which a member of the FET gene family is fused with an ETS transcription factor, with the most common fusion being EWSR1–FLI1 (85% of cases) [8,9].
At the time of diagnosis, 20-25% of the patients present with pulmonary (70-80%) and/or osseous (40-50%) metastases [10- 12].
Anatomic location preference is also different between these tumors [5,13,14], about 33% of Ewing sarcoma occur in the lower extremities, 24% in the pelvis, and 12% in the thorax [15]. Our patient also presented Ewing in right shaft of her Femora.
Staging investigations following the diagnosis of Ewing sarcoma may include chest computerized tomography (CT), technetium bone scintigraphy (bone scan), [F-18]-fluorodeoxy-D-glucose positron emission tomography (FDG-PET) scan, and bone marrow aspiration and biopsy (BMA/Bx). Each of these staging investigations provides complementary prognostic information, however the optimal combination of staging investigations is not clear [16].
In non-metastatic Ewing sarcoma 10-year overall survival is currently 55-65%, but survival in metastatic Ewing sarcoma is still dismal, with a 5-year overall survival of only 20-35% [10- 12]. Principles of treatment consist of neo-adjuvant both, and adjuvant chemotherapy [8,11].
Limping is a challenging symptom in the pediatric patient due to the variety of possible differential diagnoses [17]. Because limping is not normal, many parents will seek medical care for a child who refuses to walk, or complains of limb pain, but fortunately only a few conditions require urgent treatment. The differential diagnosis for a child with limping is quite broad, includes trauma, infection, neoplasia and inflammatory, congenital, neuromuscular or developmental disorders. The patient’s age can further narrow the differential diagnosis, because certain disease entities are age-specific [18,19].
However, limping can be a sign of a serious or even life-threatening condition. Postponements in diagnosis and treatment can result in significant morbidity and mortality [20]. Examination of a limping child should begin with a thorough history, focusing on the presence of pain, any history of trauma, and any associated systemic symptoms (fever, night sweats, weight loss, and anorexia) suggests the possibility of infection, inflammation, or malignancy. Physical examination should focus on identifying the type of limp and localizing the site of pathology by direct palpation and by examining the range of motion of individual joints. Localized tenderness may indicate contusions, fractures, osteomyelitis, or malignancy. Based on the most probable diagnoses suggested by the history and physical examination, the appropriate use of laboratory tests and imaging studies can help confirm the diagnosis [20].
Many imaging studies can be used to evaluate the bones and soft tissues, but conventional radiography is the primary imaging modality. More sophisticated imaging modalities including radionuclide scintigraphy (bone scan), computed tomography (CT), ultrasonography, and magnetic resonance (MR) imaging are reserved for specific clinical situations. Each of these imaging modalities has specific advantages and disadvantages [21].
References
- Stiller C, Parkin D. Geographic and ethnic variations in the incidence of childhood cancer. British medical bulletin. 1996; 52(4): 682-703.
- Canale ST, Beaty JH. Campbell’s operative orthopaedics e-book: Elsevier Health Sciences. 2012.
- Arndt CA, Rose PS, Folpe AL, Laack NN, editors. Common musculoskeletal tumors of childhood and adolescence. Mayo Clinic Proceedings. 2012. Elsevier.
- Mirabello L, Troisi RJ, Savage SA. Osteosarcoma incidence and survival rates from 1973 to 2004: data from the Surveillance, Epidemiology, and End Results Program. Cancer: Interdisciplinary International Journal of the American Cancer Society. 2009; 115(7): 1531-43.
- Laitinen M, Parry M, Albergo JI, Jeys L, Sumathi V, Grimer R. Outcome of pelvic bone sarcomas in children. Journal of Pediatric Orthopaedics. 2018; 38(10): 537-42.
- Delattre O, Zucman J, Plougastel B, Desmaze C, Melot T, Peter M, et al. Gene fusion with an ETS DNA-binding domain caused by chromosome translocation in human tumours. Nature. 1992; 359(6391): 162-5.
- Sorensen PH, Lessnick SL, Lopez-Terrada D, Liu XF, Triche TJ, Denny CT. A second Ewing’s sarcoma translocation, t (21; 22), fuses the EWS gene to another ETS–family transcription factor, ERG. Nature genetics. 1994; 6(2): 146-51.
- Grünewald TG, Cidre-Aranaz F, Surdez D, Tomazou EM, de Álava E, Kovar H, et al. Ewing sarcoma. Nature reviews Disease primers. 2018; 4(1): 1-22.
- Albano D, Dondi F, Schumacher RF, D’Ippolito C, Porta F, Giubbini R, et al. Clinical and Prognostic Role of 18F-FDG PET/CT in Pediatric Ewing Sarcoma. Journal of Pediatric Hematology/Oncology. 2020; 42(2): e79-e86.
- Ladenstein R, Pötschger U, Le Deley MC, Whelan J, Paulussen M, Oberlin O, et al. Primary disseminated multifocal Ewing sarcoma: results of the Euro-EWING 99 trial. J Clin Oncol. 2010; 28(20): 3284-91.
- Gaspar N, Hawkins DS, Dirksen U, Lewis IJ, Ferrari S, Le Deley M-C, et al. Ewing sarcoma: current management and future approaches through collaboration. J Clin Oncol. 2015; 33(27): 3036-46.
- Pappo AS, Dirksen U. Rhabdomyosarcoma, Ewing sarcoma, and other round cell sarcomas. Journal of Clinical Oncology. 2018; 36(2): 168-79.
- Whelan J, McTiernan A, Cooper N, Wong YK, Francis M, Vernon S, et al. Incidence and survival of malignant bone sarcomas in England 1979–2007. International journal of cancer. 2012; 131(4): E508-E17.
- Ognjanovic S, Linabery AM, Charbonneau B, Ross JA. Trends in childhood rhabdomyosarcoma incidence and survival in the United States, 1975‐2005. Cancer: Interdisciplinary International Journal of the American Cancer Society. 2009; 115(18): 4218- 26.
- Hamilton SN, Carlson R, Hasan H, Rassekh SR, Goddard K. Longterm outcomes and complications in pediatric Ewing sarcoma. American journal of clinical oncology. 2017; 40(4): 423-8.
- Newman EN, Jones RL, Hawkins DS. An evaluation of [F‐18]‐fluorodeoxy‐D‐glucose positron emission tomography, bone scan, and bone marrow aspiration/biopsy as staging investigations in Ewing sarcoma. Pediatric Blood & Cancer. 2013; 60(7): 1113-7.
- Bartoloni A, Gómez MPA, Cirillo M, Allen G, Battista G, Guglielmi G, et al. Imaging of the limping child. European Journal of Radiology. 2018; 109: 155-70.
- Leet AI, Skaggs DL. Evaluation of the acutely limping child. American Family Physician. 2000; 61(4): 1011-8.
- Lawrence LL. The limping child. Emergency medicine clinics of North America. 1998; 16(4): 911-29.
- Sawyer JR, Kapoor M. The limping child: a systematic approach to diagnosis. American family physician. 2009; 79(3): 215.
- Chaturvedi A, Rupasov A, editors. The acutely limping preschool and school-age child: an imaging perspective. Seminars in musculoskeletal radiology; 2018. Thieme Medical Publishers.