Journal of Clinical Images and Medical Case Reports
ISSN 2766-7820
Research Article - Open Access, Volume 3
Serum Mir-34A and Mir-122 as an early non-invasive diagnostic marker for simple steatosis and NASH in comparison to FIB-4
Farag NM 1 ; Hassanin TM 2 ; Abouelhassan MMA 3 ; Alyaa E 4 ; Latief AE 5 ; Nomeir HM 6 ; Abdullah NM 1 *
1 Associate Professor of Clinical and Chemical Pathology Department, Minia University, Egypt.
2 Associate Professor of Endemic Medicine and Gastroenterology, Minia University, Egypt.
3 Associate Professor of Clinical and Chemical Pathology Department, Cairo University, Egypt.
4 MD, Endemic and Infectious Diseases, Suez Canal University, Egypt.
5 Lecturer of Public Health and Occupational Medicine, Minia University, Egypt.
6 Lecturer of Medical Biochemistry Department, Alexandria University, Egypt.
*Corresponding Author: Noha M Abdullah
Department of Clinical and Chemical Pathology,
Minia University Minia, Egypt.
Email: ahmedmohamedmostafa1971@yahoo.com
Received : Mar 29, 2022
Accepted : May 10, 2022
Published : May 17, 2022
Archived : www.jcimcr.org
Copyright : © Abdullah NM (2022).
Abstract
Early detection of simple steatosis and differentiating it from NASH is an important turning point for evaluation and management. Liver biopsy is the gold standard method, but it is unpractical for many reasons and not accepted by most patients. So, many noninvasive methods have been used including microRNAs and fibrosis score (Fib-4). Our study aimed to validate the usefulness of these biomarkers for the early diagnosis of simple steatosis, differentiating it from NASH and evaluating their performance in comparison to FIB-4.Sixty two patients who met the inclusion criteria and thirty healthy controls were evaluated by Routine laboratory tests, miRNA (122,34a), Fib-4 and Liver Stiffness Measurement (LSM). The diagnostic value of miRNAs and its correlations to clinical and laboratory data in NASH and simple steatosis were analyzed and compared with FIB-4. A simple model was developed, combining microRNA levels and hs-CRP. miR-122 and -34a levels showed differential values between simple steatosis and NASH patients (p<0.01) and correlated with hepatic histology. Combined microRNA expression profiles with hs-CRP had a higher potential of simple steatosis and NASH compared to other risk biomarkers (AUC > 0,990). We concluded that circulating miR-122 and -34a may act as a disease-specific noninvasive biomarker for NASH diagnosis. miRNA along with hs-CRP assigning a prediction panel with improved diagnostic value superior to FIB-4 in early recognition of NASH to tailor efficient therapeutic interventions.
Keywords: NASH; HsCRP; miR-122; MiR34a.
Citation: Abouelhassan MMA, Alyaa E, Latief AE, Nomeir HM, Abdullah NM et al. Serum Mir-34A and Mir-122 as an early non-invasive diagnostic marker for simple steatosis and NASH in comparison to FIB-4. J Clin Images Med Case Rep. 2022; 3(5): 1837.
Introduction
Cases of Non-Alcoholic Fatty Liver Disease (NAFLD) are increasing due to alteration of food habits, obesity, metabolic syndrome, lack of exercise and poor health care system [1]. Chronic liver disease is considered as the third most common cause in the world and it’s a main cause of severe liver complications such as cirrhosis and hepatocellular carcinoma so, it becomes the most frequent indication for liver transplantation by 2030 [2,3]. However, earlier studies had suggested that simple steatosis is benign and can be treated by life-style modifications; meanwhile 25% of NASH may develop fibrosis [1,3]. For these reasons, NASH is a disease requiring early and accurate detection, routine follow-up and comprehensive medical intervention that must be distinguished from simple steatosis [4]. Liver biopsy is the gold standard test for NASH detection [5,6] but the use of liver biopsy routinely in clinical practices is difficult for many reasons including invasiveness, misdiagnosis, sampling error, cost and inter-observer variability [7]. So measurement and validation of non-invasive biomarkers that accurately diagnose, predict and monitor clinical outcomes are critically needed [7,8]. Researchers have found many serological and molecular markers that can be used instead of liver biopsy; however none have been validated [9,10]. Nowadays, FIB-4 is used as scoring system for assessing fibrosis because it is simple, accurate, and inexpensive [11,12]. But it was found that a change in fibrosis was unclear and this was detected by changes in FIB-4 index during the follow-up period. Sumida et al. reported that the FIB-4 index had a high negative predictive value (NPV) for excluding advanced fibrosis [13] and the EASLALEH clinical practice guidelines reported that a FIB-4 may had low sensitivity in detecting intermediate phases of fibrosis [14]. One of the reasons is the components of FIB-4 as serum AST and ALT levels change with diet and exercise therapy. Many treatments for NAFLD complications as T2DM and hyperlipidemia decrease AST and ALT levels [6,14]. It is also unknown if the FIB-4 index can become a marker for histological changes, because longitudinal studies about the correlation between them are rarely performed [15,16]. Recent Studies have focused on the involvement of miRNA dysregulation in the pathogenesis and progression of NAFLD, regulating many cellular pathways, mainly inflammation, apoptosis, fibrosis, lipid metabolism, and insulin resistance [17]. Also, miRNA are suitable for evaluation of liver fibrosis as they are protected from RNases in the body fluids and extremely stable [2,18]. Previous studies have reported that miR-122 is the most expressed miRNA in the human liver, consisting of 70% of total miRNAs and being confidently detected in serum [7]. Moreover, miR-34a has been implicated in a signaling pathway which is responsible for exacerbation of inflammation and triggering in turn hepatocyte apoptosis [8,19]. For these reasons, higher circulating levels of miR-34a and miR-122 are matched with the degree of hepatic fibrosis in NAFLD [20,21].
Patients and methods
This study was performed on 62 biopsy-proven patients recruited from the outpatient and inpatient Gastroenterology and Hepatology Department at El Minia University Hospital and 30 matched healthy controls recruited from blood bank donors (group I). Patients were divided into two groups, group II including 34 simple steatosis patients and group III including 28 NASH patients. The exclusion criteria were: 1) Other causes of chronic liver disease; 2) alcohol intake more than 100 g ethanol/ week; 3) Absence of clinical and laboratory data necessary for calculation of the score.
All patients were subjected to detailed history and examination, laboratory investigations, liver fibrosis severity determined by FIB-4 score calculated as follows: [age (year) ×AST (U/L)]/ [(platelet count (PLT)(109/L)) × (ALT (U/L))1/2] [8] and Liver Stiffness Measurements (LSM) were performed using two-dimensional shear wave elastography. Histological diagnosis of NASH is defined as “the occurrence of hepatic steatosis and inflammation with hepatocyte damage (ballooning) associated with or without fibrosis”. While, nonalcoholic fatty liver or simple steatosis was defined as “the occurrence of hepatic steatosis with no evidence of hepatocellular damage in the form of hepatocytes ballooning or no evidence of fibrosis” According to the latest AASLD guidelines [2]. Approximately 8 mL of blood were withdrawn, chemical profile analyzed on Selectra proM, ELITech Group, France, CBC using Sysmex diagnostic, high sensitive C reactive protein determined by ELISA Kit supplied by DRG International Inc. USA. Serum was separated, stored at −80 ◦C for further measurement of miRNA expression on RTPCR. Serum miRs were extracted by a miRNA purification kit (Norgen) according to the manufacturer’s instructions. Next, reverse transcription with a universal anchor primer (CGACTCGATCCAGTCTCAGGGTCCGAGGTATTCGATCCTAACCCTCTCCTCGGTATCGAGTCGCACTTTTTTTTTTTTVN) (Invitrogen, Barcelona, Spain). Quantitation of miRNA was conducted using a universal reverse primer (CCAGTCTCAGGGTCCGAGGTATTC), a specific forward primer for each miRNA 122: 50AACGCCAUUAUCACACUAAAUA; miR-34a: 50CAAUCAGCAAGUAUACUGCCCU and a universal TaqMan probe (FAMTCTCCTCGGTATCGAGTCGCACT-TAMRA). The expression levels of miR-34a and miR-122 in serum were determined by comparing the Ct values of each sample with the standard curve.
Statistical analysis
The analysis of the data was done by IBM SPSS 20.0 statistical package software. Normality of the data was tested via the Shapiro-Wilk or Kolmogorov-Smirnov tests. Data were expressed as mean ± Standard Deviation (SD), minimum and maximum range, median and range for quantitative non-parametric measures in addition to both number and percentage for categorized data. For comparison between more than two independent groups, Analysis Of Variance (ANOVA) was performed for comparison for parametric data followed by LSD post-hoc test to assess intergroup differences, and Kruskal-Wallis (KW) statistical test followed by Dunn’s post-hoc test for non-parametric data. Chi-square test or Fisher’s exact test were done to compare categorical variables. Correlations between the parameters were analyzed by Pearson’s correlation analysis. The accuracy of different biomarkers for diagnosis of steatosis and NASH was assessed using a ROC curve, which plots the sensitivity (true-positive rate) to the false- positive rate (1 -specificity). A p-value of 0.05 or less was considered significant.
Results
An increasing trend in severity of liver injury was observed when progressing from simple steatosis to NASH, as evidenced by the significantly higher values of AST, ALT, GGT and FIB-4, lower platelet and albumin levels in group (III) (p < 0.01). They were significantly higher not only when compared to healthy subjects but also between the NASH and simple steatosis groups. Also, miR-122 and miR-34 were significantly up regulated in group III and group II as compared to control and when compared to each other (p < 0.05) (Table 1).
Table 1: Demographic and routine investigations of study participants
Mean±SD |
Control (I) |
Simple steatosis (II) |
NASH (III) |
p value |
||
(n=30) |
(n=34) |
(n=28) |
I vs II |
I vs III |
II vs III |
|
Age (y) |
36.9±9.4 (21-53) |
34.7±5.5 (27-46) |
33.8±8 (31-58) |
0.26 |
0.29 |
0.31 |
Sex Male Female |
10 (33.3%) (66.7%) |
10 (29.4%) (70.6%) |
8 (28.6%) (71.4%) |
0.929 |
0.519 |
0.576 |
PLT (x109 /L) |
249.1±35.2 (180-310) |
242±29.5 (198-3510 |
222.8±22.4 (188-264) |
0.351 |
0.001* |
0.012* |
ALT U/L |
15.2±8 (4.9-33.8) |
56.8±15.2 (37.2-97.8) |
89.1±27.6 (50.6-162.4) |
<0.001* |
<0.001* |
<0.001* |
AST (U/L) |
14.7±9.5 (4.1-36.4) |
49.2±13.9 (30.3-83.6) |
76.3±25.3 (39.7-151.7) |
<0.001* |
<0.001* |
<0.001* |
Alb- (g/dl) |
4.1±0.5 (3.4-5) |
3.7±0.4 (3-4.3) |
3.5±0.5 (2.9-4.5) |
<0.001* |
<0.001* |
0.282 |
GGT U/L |
28.4±13.4 (9-59) |
44.9±11.8 (29-67) |
52±14.7 (29-79) |
<0.001* |
<0.001* |
0.039* |
FIB-4 |
0.56±0.26 (0.18-1.24) |
0.93±0.18 (0.61-1.24) |
1.59±0.31 (1.15-2.12) |
<0.001* |
<0.001* |
<0.001* |
hsCRP (mg/ L) |
0.4±0.4 (0.2-1.9) |
(0.2-1.9) (0.5-5.1) |
3.9±1.9 (0.7-7) |
<0.001* |
<0.001* |
<0.003* |
miR 122 |
0.8±0.8 (0.4-3.7) |
4.2±1.5 (0.9-6.7) |
5.1±1.6 (1.1-7.6) |
<0.001* |
<0.001* |
<0.017* |
miR 122 |
1.3±0.5 (0.5-2.4) |
2.35±1.04 (0.97-4.32) |
3.47±1.77 (0.96-6.62) |
0.004* |
<0.001* |
0.001* |
Although miR-122 and miR-34a (Table 2) were positively correlated with FIB-4, the observations did not reach statistical significance for miR34 a. Additionally, There were statistically significant positive association among AST, ALT with mir34a and mir122 and negative correlation with albumin and platelets. Also, There were Significant positive correlation among hs-CRP and (AST, ALT, GGT, mir122, miR 34a) but hs-CRP did not reach statistical significance with FIB 4.
Table 2: Correlation between specific markers with other variables
|
FIB-4 |
Mir 34a |
Mir 122 |
hsCRP |
||||
|
r |
p |
r |
p |
r |
p |
r |
p |
hsCRP |
0.328 |
0.460 |
0.548 |
0.031* |
0.547 |
0.019* |
- |
- |
PLT |
-0.546 |
0.015* |
-0.214 |
0.248 |
-0.253 |
0.169 |
0.278 |
0.13 |
ALT |
0.428 |
0.016* |
0.517 |
0.006* |
0.659 |
0.009* |
0.515 |
0.010* |
AST |
0.453 |
0.010* |
0.010* |
0.019* |
0.576 |
0.005* |
0.505 |
0.012* |
GGT |
0.274 |
0.135 |
0.750 |
<0.001* |
0.575 |
0.001* |
0.651 |
0.004* |
ALB |
-0.317 |
0.082 |
-0.635 |
<0.001* |
-0.581 |
0.001* |
-0.558 |
0.001 |
miR 34 |
0.254 |
0.168 |
- |
- |
- |
- |
- |
- |
miR 122 |
0.504 |
0.004* |
0.511 |
0.009* |
- |
- |
- |
- |
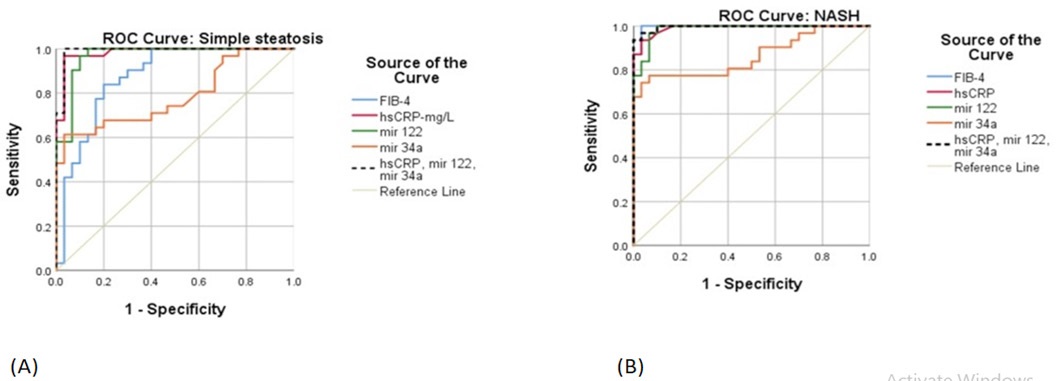
ROC curve analysis shows the diagnostic performances predicting the presence or absence of simple steatosis. Hs- CRP and miR122 had the highest sensitivity while miR34a had the lowest sensitivity but the highest specificity for simple steatosis diagnosis. Areas under the Curve (AUC) values for these specific markers were >0.78 which displayed statistically significant predictive values (p<0.001). Fortunately, these three markers if used together yields better sensitivity and specificity results giving better prediction of simple steatosis than using it individually or using FIB-4 indicating that this panel is a more sensitive indicator for simple steatosis than NASH (Table 3 and Figure1A).
Table 3: ROC curves analysis and Predictive value of different biomarkers
|
NASH |
Simple Steatosis |
||||||||||
|
Cutoff |
AUC |
PPV |
NPV |
Sensitivity |
Specificity |
Cutoff |
AUC |
PPV |
NPV |
Sensitivity |
Specificity |
FIB-4 |
>1.14 |
0.996 |
96.7 |
96.8 |
96.8 |
96.7 |
>0.73 |
0.873 |
81.2 |
82.8 |
83.9 |
80 |
hsCRP |
>1.2 |
0.991 |
96.7 |
93.5 |
93.5 |
96.7 |
>1.2 |
0.983 |
96.8 |
96.7 |
96.8 |
96.7 |
miR 122 |
>2.4 |
0.986 |
93.5 |
96.7 |
96.8 |
93.3 |
>1 |
0.983 |
90.9 |
96.4 |
96.8 |
90 |
miR 34a |
>1.97 |
0.863 |
95.7 |
78.9 |
74.2 |
96.7 |
>1.97 |
0.785 |
95.0 |
70.7 |
61.3 |
96.7 |
HsCRP, miR 122, miR 34a |
|
0.996 |
100.0 |
93.7 |
93.5 |
100 |
|
0.990 |
96.9 |
100 |
100 |
96.7 |
We further estimated the predictive value of circulating miRNAs and FIB -4 in our patients (Table 3 and Figure 1B). As expected, FIB-4 provided high predictive value for NASH with Area under the Curve (AUC) of 0.996, but its predictive value for simple steatosis is suboptimal (AUC: 0.873). In comparison, the predictive value of miR122 performs similarly to Fib -4 in NASH with AUC 0.986 but is superior to it in simple steatosis with AUC 0.986 means it is better for early detection. The combined model derived from miRNAs and hs-CRP does not significantly improve the NPV from miR34a for simple steatosis alone but also moderately increases the predictive value to predict NASH. This may inform the ability of development of miRNA-based diagnostic strategies.
A correlation analysis between serum miRNAs and hepatic histology was performed in patients (Table 4). miR - 34a and -122 were correlated with hepatic steatosis (r= 0.317 and 0.257 respectively, P <0.01, P < 0.05) and miR- 34a had the strongest correlation with lobular inflammation (r = 0.473) while, miR-122 had the strongest correlation with hepatocellular ballooning (r = 0.348).
Table 4: Correlations between miRNAs and histological features
|
miR-34a |
miR-34a |
||
|
r |
p |
r |
p |
Steatosis |
0.317 |
< 0.01 |
0.257 |
< 0.05 |
Lobular inflammation |
0.473 |
< 0.01 |
0.195 |
< 0.05 |
Hepatocellular ballooning |
0.289 |
< 0.05 |
0.348 |
< 0.01 |
Discussion
Diagnosis of the NAFLD among population is routinely evaluated by ultrasonography or other imaging methods, but recently, liver biopsy is the only method available used to validate and differentiate between NAFL and NASH. Therefore, trials for searching a suitable serum biomarkers is pursued with high priority. Currently published studies have focused on efficiency of several plasma markers that significantly predict NAFLD progression independently but all of them have limitations, both intrinsic and related to liver damage etiology [20,21]. However, this study represents a panel of biomarkers with unique combination which has not been established before.
Despite the expanding body of literature on miRNA in NAFLD, most studies are small and comparing groups of patients with NAFLD or NASH to healthy controls [22]. Herein, focused on two of the most important NAFLD-related miRNAs: miR-34a, miR-122. In order to increase accuracy of these tests we investigated them individually or if their combination could be useful. In addition, many scoring systems are present, but it has a low positive PPV and NASH tends to be overlooked. One of these systems is called BARD score that include BMI and reported to be less reliable for Japanese people with a low BMI. Among the various scoring systems, FIB-4 has been reported as the liver fibrosis system with the most valid AUC [23]. Therefore, we used it in our study as it is considered the most reliable [24]. Liver enzymes commonly used as a marker for liver inflammation but has low predictive value for diagnosis of NASH. Serum ALT value more than two times the upper limit of normal has a sensitivity of 50% and specificity of 61% for NASH detection as 80% of patients with fatty liver have ALT levels within normal and do not associate with the degree of hepatic fibrosis. Also, the majority of previous studies reported that AST/ALT ratio and GGT are not helpful or may have little predictive value of liver fibrosis. So, NASH can’t be excluded in patients with normal aminotransferases [25]. In agree with our study, Tada et al found significant increase of ALT and AST in NAFLD indicating that these miRNAs are secreted from hepatocytes during pathophysiological states associated with damage of cell membrane [26]. It can be proved that miRNAs generally show an earlier increase than ALT in NASH patients. This hypothesis is strengthened by founding of 25% of NASH patients with predictive miRNA had no abnormalities in ALT level [27]. Hs-CRP is used as a marker of lowgrade systemic inflammation and similar to our study, two studies found that hs-CRP levels were significantly higher in patients with NASH compared to those with simple steatosis and associated with advanced fibrosis which means the risk for NAFLD was increased as increased the level of hs-CRP [28,29]. Yilmaz et al supported our study which showed absence of significant correlation between FIB-4 and hsCRP in NAFLD [29]. However, Hui JM et al. showed that measuring of hs-CRP was not useful in predicting the histological severity of NAFLD, because there was no correlation between hs-CRP levels and the grades of fibrosis [30]. Recently, Zimmermann E et al. recorded that hsCRP may be a marker of steatosis, but not of NAFLD severity [31]. Our study in agreement with Ghideon et al and Salvoza et al shows higher miR122 and miR34a in patient groups compared to healthy controls and in patients with NASH relative to those with simple steatosis so more beneficial for detection and strongly correlate with severity in NAFLD [22,32]. This also in agreement of Liu et al who reported that miR-122, and miR-34a could differentiate NAFLD from healthy controls and could distinguish NASH from simple steatosis [25].
Also, Pirola et al. detected high level of mir122 as 7.2-fold in NASH patients and 3.1-fold in simple steatosis, so it can be used as a fingerprint of NASH [33]. Similar to our findings, Yamada et al. reported that miR-122 level was correlated with the severity of steatosis. However, no significant correlation was established between the expression level of serum miR-34a and severity [34]. Also, Ghideon Ezaz et al found miR 122 correlates significantly with FIB -4 in NASH only, and we can say, this study is the first one that demonstrate the relations between mir34a with AST [22].
In agreement with our study, miRNAs provide good predictive value to determine early stages of fibrosis and miR- 34a was the most micro RNA associated with all histological features of NAFLD [22,35]. Also, Liu et al found a great correlation between hepatic steatosis and serum miR-122 and -34a. Meanwhile, serum miR-34a levels showed stronger correlation with hepatic inflammation severity, including lobular inflammation and hepatocellular ballooning [25]. MiR-34a can down-Regulate Sirtuin1 (SIRT1) leading to accumulation cholesterol and activation of hepatocytes apoptosis. Also, it regulates lipoprotein metabolism and promotes liver steatosis, miR-122 has inhibitory effect on activation of the stellate cell and deposition of collagen inside the liver [36-38]. Previous studies indicated an increase of miR-122 in serum with hepatocytes apoptosis caused by inflammatory damage to the liver [39].
Interestingly, combining markers with 34a in our study increase its predictive value, specificity and sensitivity making a well predictive panel superior to FIB-4 especially for diagnosing simple steatosis so preventing unnecessary liver biopsy. This is of a clinical important utility as a noninvasive method for bloodbased fibrosis scoring tool. Finally, in larger sample number new fibrosis score can be calculated and using these markers in routine clinical practice will make it of low cost so, analysis can be valuable at the primary care levels.
Conclusion
We concluded that a combination of miR122, miR34a, and hs-CRP can improve their diagnostic value in recognition of simple steatosis and NASH. So, in larger sample size a new fibrosis score can be calculated and its use in routine clinical practice will make it low cost suitable for use in primary care levels
Declarations
Author contributions: Conceptualization, Nagla Farag and Noha Abdullah; Data curation, Nagla Farag, Taha Hassanin and Abd Latief; Formal analysis, Nagla Farag, Mona elhassan, Abd Latief, Hanan Nomeir and Noha Abdullah; Investigation, Nagla Farag, Mona elhassan, Hanan Nomeir and Noha Abdullah; Methodology, Nagla Farag, Mona elhassan and Noha Abdullah; Supervision, Nagla Farag; Writing – original draft, Nagla Farag; Writing - review & editing, Noha Abdullah
Funding: This research had no external funding.
Institutional review board statement: The study was conducted according to the guidelines of the Declaration of Helsinki and was approved by the Institutional Review Board (or Ethics Committee) of faculty of medicine, Minia University (Approval No. 133-2021).
Informed consent statement: Informed consent was obtained from all patients involved in the study.
Data availability statement: The data are not publicly available due to privacy issues.
Acknowledgements: All authors had grateful with many thanks to all patients who included in this study.
Conflicts of interest: The authors declare no conflict of interest
References
- Lonardo A, Byrne C, Caldwell S, Cortez‐Pinto H, Targher G. Global epidemiology of nonalcoholic fatty liver disease: Meta‐analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016; 64: 1388-1389.
- Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018; 67: 328-357.
- Paul J. Recent advances in non-invasive diagnosis and medical management of non-alcoholic fatty liver disease in adult. Egyptian Liver Journal. 2020; 10: 1-18.
- Kobayashi N, Kumada T, Toyoda H, Tada T, Ito T, et al. Ability of cytokeratin-18 fragments and FIB-4 index to diagnose overall and mild fibrosis nonalcoholic steatohepatitis in Japanese nonalcoholic fatty liver disease patients. Digestive Diseases. 2017; 35: 521-530.
- Albitar AR, Emad N, Yamany A. Pentraxin 3 and Non-Alcoholic Fatty Liver Disease in Egyptian Patients: Merits and Flaws. Indian Journal of Public Health Research & Development. 2019: 10.
- Maher M, Abdelaziz H, yossif T, Ossama M. Cytokeratin 18 as a noninvasive marker in diagnosis of NASH and its usefulness in correlation with disease severity in Egyptian patients. QJM: An International Journal of Medicine. 2020; 113: 10.
- Egyptian Patients: Merits and Flaws. Indian Journal of Public Health Research & Development. 2019; 10.
- Satapathy SK, Kuwajima V, Nadelson J, Atiq O, Sanyal AJ. Druginduced fatty liver disease: An overview of pathogenesis and management. Annals of hepatology. 2015; 14: 789-806.
- Ezaz G, Trivedi HD, Connelly MA, Filozof C, Howard KL,et al. Differential associations of circulating MicroRNAs with pathogenic factors in NAFLD. Hepatology communications. 2020; 4: 670- 680.
- Jampoka K, Muangpaisarn P, Khongnomnan K, Treeprasertsuk S, Tangkijvanich P, et al. Serum miR- 29a and miR-122 as potential biomarkers for non-alcoholic fatty liver disease (NAFLD). Microrna. 2018; 7: 215- 222.
- Mohammed MA, Omar NM, Mohammed SA, Amin AM, Gad DF. FICK-3 Score Combining Fibrosis-4, Insulin Resistance and Cytokeratin-18 in Predicting Non-alcoholic Steatohepatitis in NAFLD Egyptian Patients. Pakistan journal of biological sciences: PJBS. 2019; 22: 457-466.
- Seko Y, Sumida Y, Sasaki K, Itoh Y, Iijima H, et al. Effects of canagliflozin, an SGLT2 inhibitor, on hepatic function in Japanese patients with type 2 diabetes mellitus: pooled and subgroup analyses of clinical trials. Journal of gastroenterology. 2018; 53: 140-151.
- Sumida Y, Yoneda M, Hyogo H, Itoh Y, Ono M, et al. Validation of the FIB4 index in a Japanese nonalcoholic fatty liver disease population. BMC Gastroenterol. 2012; 5: 12.
- European Association for Study of Liver; Asociacion Latinoamericana para el Estudio del Higado. EASL-ALEH Clinical Practice Guidelines: Non-invasive tests for evaluation of liver disease severity and prognosis. J Hepatol. 2015; 63: 237-264.
- Wong VWS. Nonalcoholic fatty liver disease in A sia: A story of growth. Journal of gastroenterology and hepatology. 2013; 28: 18-23.
- Paola D, Salvatore P, Ville M, Mancina RM, Rosaria P, et al. Statin use and non-alcoholic steatohepatitis in at risk individuals. 2015.
- Zhiqiang Fang , Guorui Dou , Lin Wang . MicroRNAs in the Pathogenesis of Nonalcoholic Fatty Liver Disease . Int. J. Biol. Sci. 2021; 17: 1851-1863.
- Soronen J, Yki‐Järvinen H, Zhou Y, Sädevirta S, Sarin AP, et al. Novel hepatic microRNAs upregulated in human nonalcoholic fatty liver disease. Physiological reports. 2016; 4: e12661.
- Sobolewski C, Calo N, Portius D, Foti M, editors. MicroRNAs in fatty liver disease. Seminars in liver disease, Thieme Medical Publishers. 2015.
- Zambrano-Huailla R, Guedes L, Stefano JT, de Souza AAA, Marciano S, et al. Diagnostic performance of three non-invasive fibrosis scores (Hepamet, FIB-4, NAFLD fibrosis score) in NAFLD patients from a mixed Latin American population. Annals of Hepatology. 2020; 19: 622-626.
- Sun W, Cui H, Li N, Wei Y, Lai S, et al. Comparison of FIB‐4 index, NAFLD fibrosis score and BARD score for prediction of advanced fibrosis in adult patients with non‐alcoholic fatty liver disease: a meta‐ analysis study. Hepatology research. 2016; 46: 862-870.
- Ghideon Ezaz, Hirsh D Trivedi, Margery A Connelly, Claudia Filozof, Kellie Howard, et al. Differential Associations of Circulating MicroRNAs With Pathogenic Factors in NAFLD. Hepatology CommuniCations. 2020; 4: 5.
- Keisuke Okamura, Tetsu Okuda, Yosuke Takamiya, Kazuyuki Shirai, Hidenori Urata. High Fib4 index in patients with suspected NASH is associated with elevation of chymase-dependent angiotensin II-forming activity in circulating mononuclear leucocytes. Heart and Vessels. 2019; 34: 1559-1569.
- Darweesh SK, AbdElAziz RA, Abd-ElFatah DS, AbdElazim NA, Fathi SA, et al. Serum cytokeratin-18 and its relation to liver fibrosis and steatosis diagnosed by FibroScan and controlled attenuation parameter in nonalcoholic fatty liver disease and hepatitis C virus patients. European journal of gastroenterology & hepatology. 2019; 31: 633-641.
- Liu X-L, Pan Q, Zhang R-N, Shen F, Yan S-Y, et al. Disease-specific miR-34a as diagnostic marker of non-alcoholic steatohepatitis in a Chinese population. World journal of gastroenterology. 2016; 22: 9844.
- Tada T, Kumada T, Toyoda H, Saibara T, Ono M . New scoring system combining the FIB-4 index and cytokeratin-18 fragments for predicting steatohepatitis and liver fibrosis in patients with nonalcoholic fatty liver disease. Biomarkers. 2018; 23: 328-334.
- Becker PP, Rau M, Schmitt J, Malsch C, Hammer C, et al. Performance of Serum microRNAs-122, -192 and -21 as Biomarkers in Patients with Non-Alcoholic Steatohepatitis. PLoS ONE. 2015; 10: e0142661.
- Yoneda M, Mawatari H, Fujita K, Iida H, Yonemitsu K, et al. Highsensitivity C-reactiveprotein is an independent clinical feature of nonalcoholic steatohepatitis (NASH) and also of the severity of fibrosis in NASH. J Gastroenterol. 2007; 42: 573-582.
- Yalmaz, et al. Neutrophil-Lymphocyte Ratio (NLR) Could Be Better Predictor than C-reactive Protein (CRP) for Liver Fibrosis in Non-alcoholic Steatohepatitis (NASH). Annals of Clinical & Laboratory Science. 2015; 45: 3.
- Hui JM, Farrell GC, Kench JG, George J. High sensitivity C-reactive protein values do not reliably predict the severity of histological changes in NAFLD. Hepatology. 2004; 39: 1458-1459.
- Zimmermann E, Anty R, Tordjman J, Verrijken A, Gual P, et al. C-reactive protein levels in relation to various features of nonalcoholic fatty liver disease among obese patients. J Hepatol. 2011; 55: 660-665.
- Salvoza NC, Klinzing DC, Gopez-Cervantes J, Baclig MO. Association of circulating serum miR-34a and miR- 122 with dyslipidemia among patients with non-alcoholic fatty liver disease. PloS one. 2016; 11: e0153497.
- Pirola CJ, Gianotti TF, Castaño GO, Mallardi P, San Martino J, et al. Circulating microRNA signature in non-alcoholic fatty liver disease: from serum non-coding RNAs to liver histology and disease pathogenesis. Gut. 2015; 64: 800-812.
- Yamada H, Suzuki K, Ichino N, Ando Y, Sawada A, et al. Associations between circulating microRNAs (miR-21, miR-34a, miR122 and miR-451) and non-alcoholic fatty liver. Clin him Acta. 2013; 424: 99-103.
- Kumar, et al , Association of high-sensitivity C-reactive protein (hs-CRP) with non-alcoholic fatty liver disease (NAFLD) in Asian Indians: A cross-sectional study. 2021; IP: 156.195.138.103.
- Min HK, Kapoor A, Fuchs M, Mirshahi F, Zhou H, et al. Increased hepatic synthesis and dysregulation of cholesterol metabolism is associated with the severity of nonalcoholic fatty liver disease. Cell Metab. 2012; 15: 665-674.
- Ding J, Li M, Wan X, Jin X, Chen S, et al. Effect of miR-34a in regulating steatosis by targeting PPARα expression in nonalcoholic fatty liver disease. Sci Rep. 2015; 5: 13729.
- Castro RE, Ferreira DM, Afonso MB, Borralho PM, Machado MV, et al. Rodrigues CMmiR-34a/SIRT1/p53 is suppressed by ursodeoxycholic acid in the rat liver and activated by disease severity in human non-alcoholic fatty liver disease. J Hepatol. 2013; 58: 119-125.
- Roderburg C, Benz F, Vargas Cardenas D, Koch A, Janssen J, et al. Elevated miR-122 serum levels are an independent marker of liver injury in inflammatory diseases. Liver international: official journal of the International Association for the Study of the Liver. 2015; 35:1172-1184.